Cardio Diabetes Master Class Asian chapter January 28

Cardio Diabetes Master Class Asian chapter January 28 -30 2011, Shanghai Presentation topic TZD’s in the management of diabetes & CV disease: All about benefits and safety Slide lecture prepared and held by: Prof. John Betteridge, MD University College London, United Kingdom PHYSICIANS’ ACADEMY FOR CARDIOVASCULAR EDUCATION

Diabetes and Cardiovascular Disease: Time To Act! “With the rising tide of diabetes around the globe, the double jeopardy of diabetes and cardiovascular disease is set to result in an explosion of these and other complications- unless preventive action is taken. ” Prof Sir George Alberti, IDF President. International Diabetes Federation
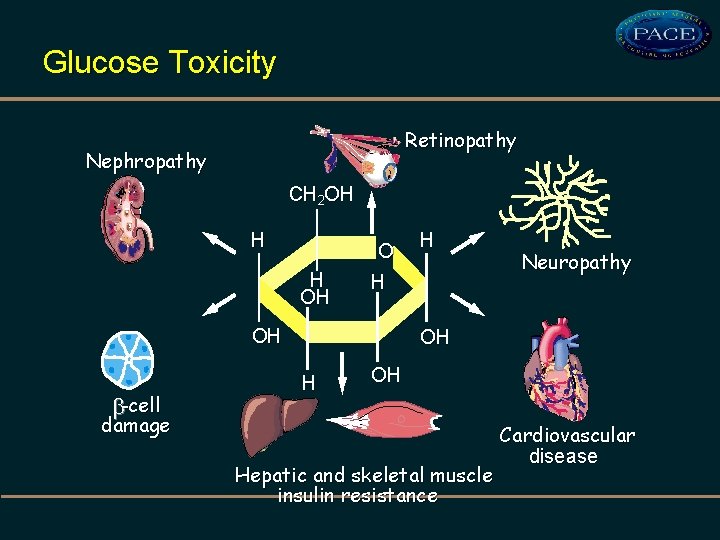
Glucose Toxicity Retinopathy Nephropathy CH 2 OH H H OH O H OH -cell H Neuropathy OH H OH damage Hepatic and skeletal muscle insulin resistance Cardiovascular disease
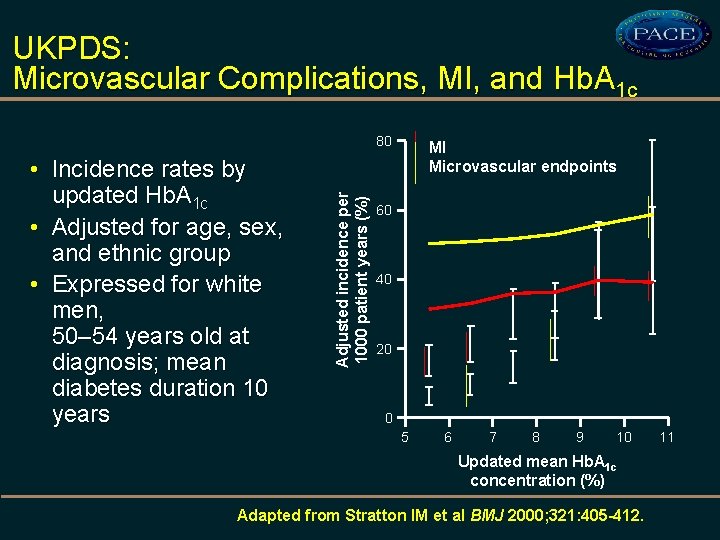
UKPDS: Microvascular Complications, MI, and Hb. A 1 c • Incidence rates by updated Hb. A 1 c • Adjusted for age, sex, and ethnic group • Expressed for white men, 50– 54 years old at diagnosis; mean diabetes duration 10 years Adjusted incidence per 1000 patient years (%) 80 MI Microvascular endpoints 60 40 20 0 5 6 7 8 9 10 Updated mean Hb. A 1 c concentration (%) Adapted from Stratton IM et al BMJ 2000; 321: 405 -412. 11
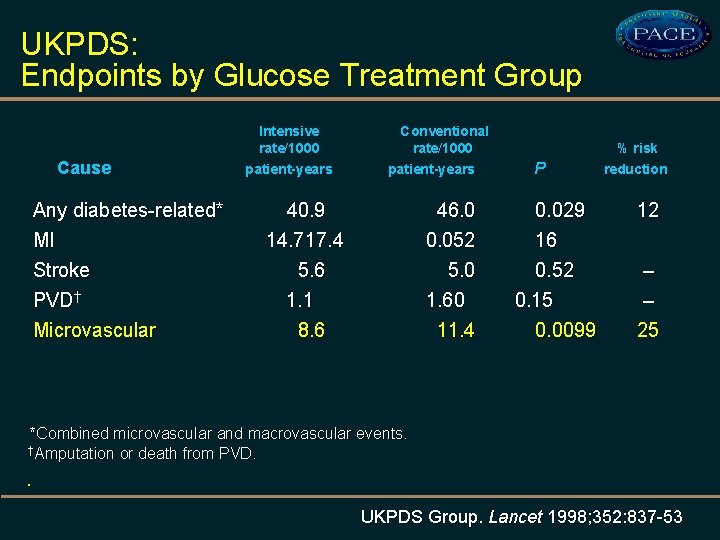
UKPDS: Endpoints by Glucose Treatment Group Cause Any diabetes-related* MI Stroke PVD† Microvascular Intensive rate/1000 patient-years Conventional rate/1000 patient-years P % risk reduction 40. 9 14. 7 17. 4 46. 0 0. 052 0. 029 16 12 5. 6 1. 1 8. 6 5. 0 1. 60 11. 4 0. 52 0. 15 0. 0099 – – 25 *Combined microvascular and macrovascular events. †Amputation or death from PVD. . UKPDS Group. Lancet 1998; 352: 837 -53
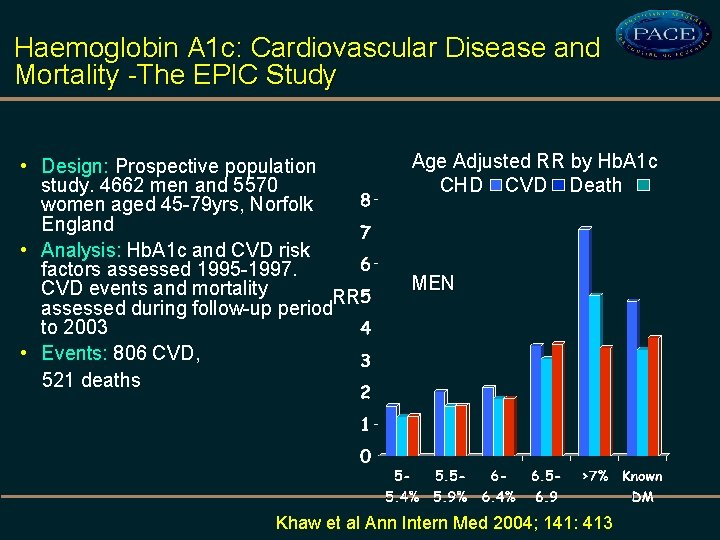
Haemoglobin A 1 c: Cardiovascular Disease and Mortality -The EPIC Study • Design: Prospective population study. 4662 men and 5570 women aged 45 -79 yrs, Norfolk England • Analysis: Hb. A 1 c and CVD risk factors assessed 1995 -1997. CVD events and mortality RR assessed during follow-up period to 2003 • Events: 806 CVD, 521 deaths Age Adjusted RR by Hb. A 1 c CHD CVD Death MEN Khaw et al Ann Intern Med 2004; 141: 413

Conclusions: RCTs of intensive versus standard glycaemic therapy have not shown a significant reduction in CVD outcomes during the randomized period of the trials. NB Long-term follow-up of DCCT and UKPDS suggests that Hb. A 1 c around 7% in years soon after diagnosis is associated with long-term risk reduction. Less stringent Hb. A 1 c goals may be appropriate for patients with history of severe hypoglycaemia, advanced CVD complications or extensive co-morbid conditions or those with long-standing diabetes in whom the general goal is difficult to maintain. Evidence-based recommendations should be followed for blood pressure management, statins smoking cessation, aspirin and healthy lifestyles Skyler et al Diabetes Care 2009, 33: 187
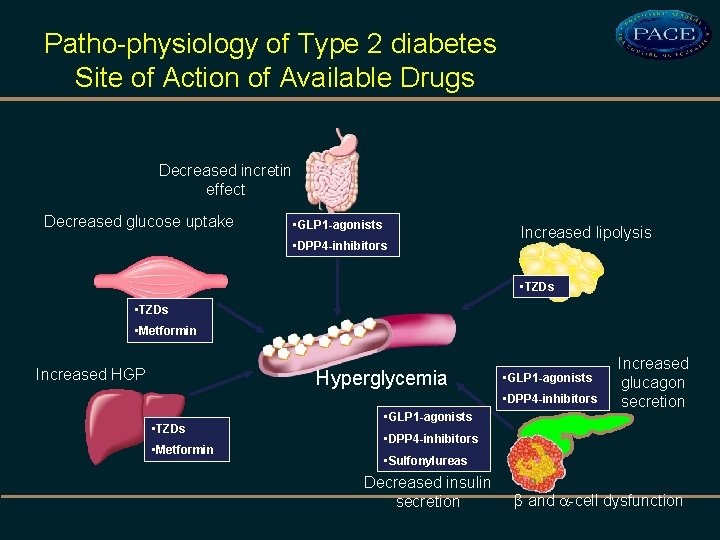
Patho-physiology of Type 2 diabetes Site of Action of Available Drugs Decreased incretin effect Decreased glucose uptake • GLP 1 -agonists • DPP 4 -inhibitors Increased lipolysis • TZDs • Metformin Increased HGP Hyperglycemia • GLP 1 -agonists • DPP 4 -inhibitors • TZDs • Metformin • GLP 1 -agonists Increased glucagon secretion • DPP 4 -inhibitors • Sulfonylureas Decreased insulin secretion β and -cell dysfunction
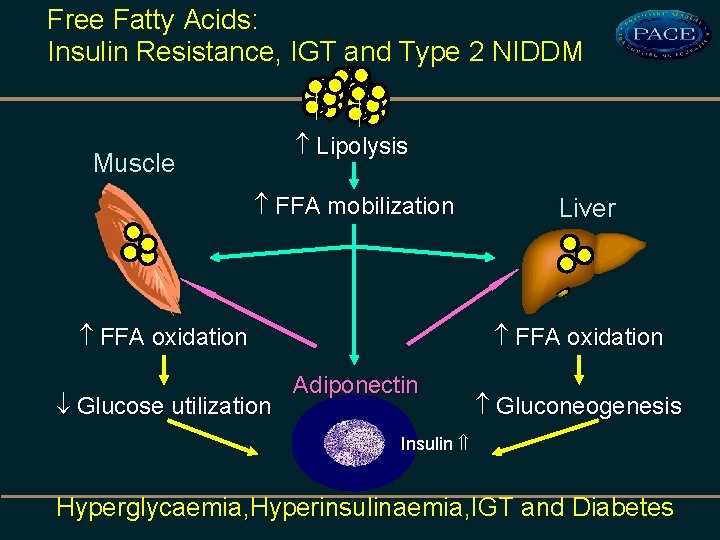
Free Fatty Acids: Insulin Resistance, IGT and Type 2 NIDDM Lipolysis Muscle FFA mobilization FFA oxidation Glucose utilization Liver Adiponectin Gluconeogenesis Insulin Hyperglycaemia, Hyperinsulinaemia, IGT and Diabetes
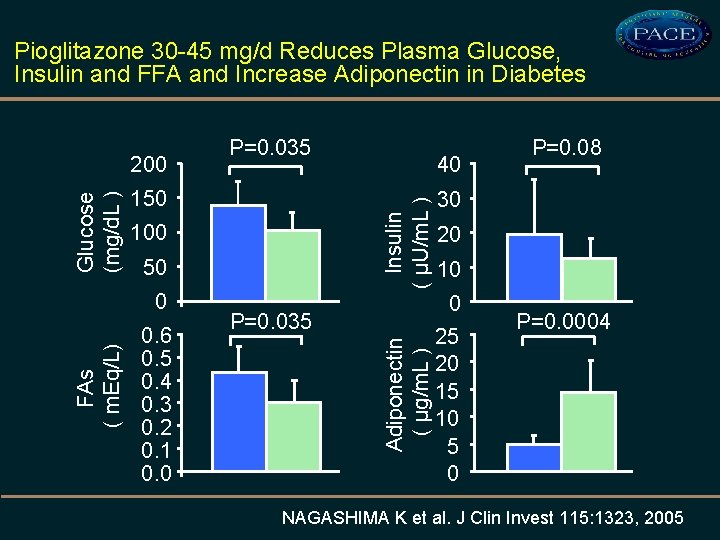
Pioglitazone 30 -45 mg/d Reduces Plasma Glucose, Insulin and FFA and Increase Adiponectin in Diabetes 100 50 0 FAs ( m. Eq/L) Insulin ( µU/m. L ) 150 0. 6 0. 5 0. 4 0. 3 0. 2 0. 1 0. 0 40 P=0. 035 Adiponectin ( µg/m. L ) Glucose (mg/d. L ) 200 P=0. 035 P=0. 08 30 20 10 0 25 20 15 10 5 0 P=0. 0004 NAGASHIMA K et al. J Clin Invest 115: 1323, 2005
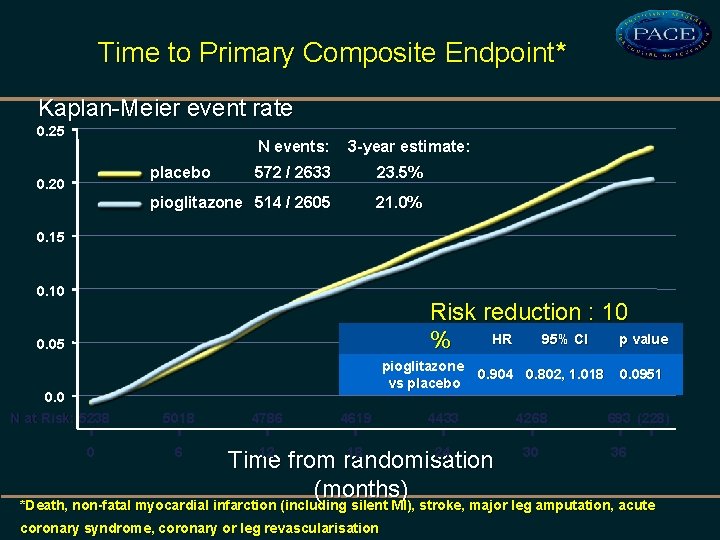
Time to Primary Composite Endpoint* Kaplan-Meier event rate 0. 25 N events: placebo 0. 20 3 -year estimate: 572 / 2633 23. 5% pioglitazone 514 / 2605 21. 0% 0. 15 0. 10 Risk reduction : 10 HR 95% CI p value % 0. 05 0. 0 N at Risk: 5238 0 pioglitazone vs placebo 5018 6 4786 4619 0. 904 0. 802, 1. 018 4433 12 18 24 Time from randomisation (months) 4268 30 0. 0951 693 (228) 36 *Death, non-fatal myocardial infarction (including silent MI), stroke, major leg amputation, acute coronary syndrome, coronary or leg revascularisation
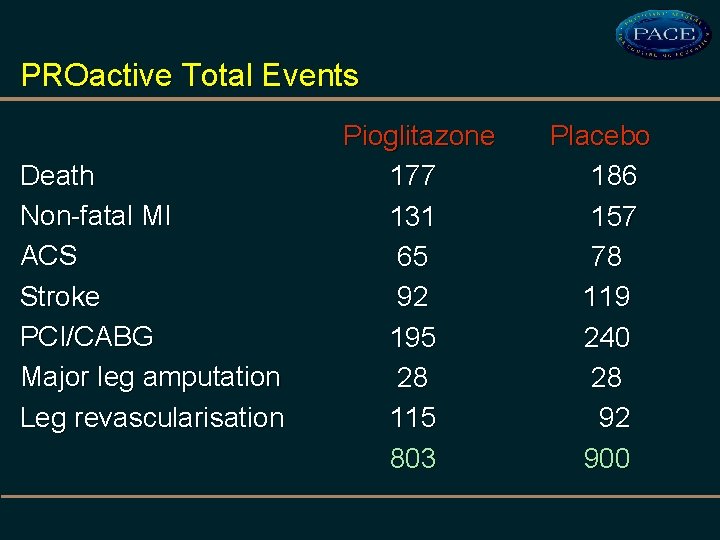
PROactive Total Events Death Non-fatal MI ACS Stroke PCI/CABG Major leg amputation Leg revascularisation Pioglitazone 177 131 65 92 195 28 115 803 Placebo 186 157 78 119 240 28 92 900
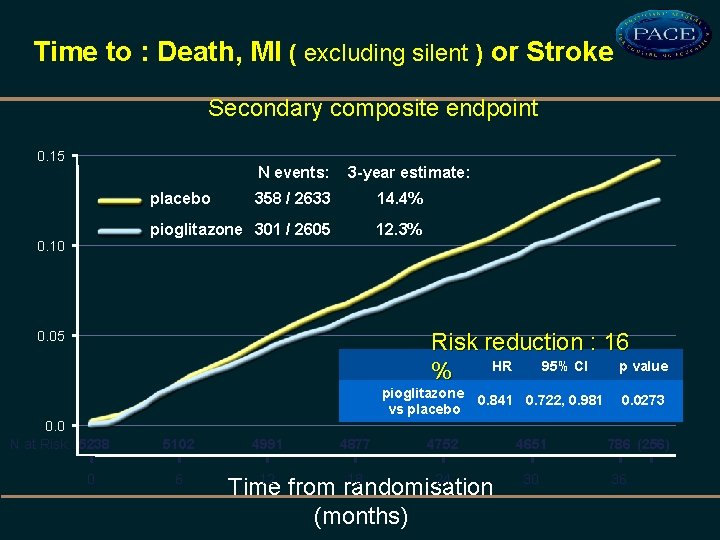
Time to : Death, MI ( excluding silent ) or Stroke Secondary composite endpoint 0. 15 N events: placebo 0. 10 3 -year estimate: 358 / 2633 14. 4% pioglitazone 301 / 2605 12. 3% 0. 05 Risk reduction : 16 HR 95% CI p value % pioglitazone vs placebo 0. 0 N at Risk: 5238 0 5102 6 4991 4877 0. 841 0. 722, 0. 981 4752 12 18 24 Time from randomisation (months) 4651 30 0. 0273 786 (256) 36

In high-risk patients with type 2 diabetes and previous MI, pioglitazone significantly reduced the occurrence of recurrent fatal and nonfatal MI and ACS
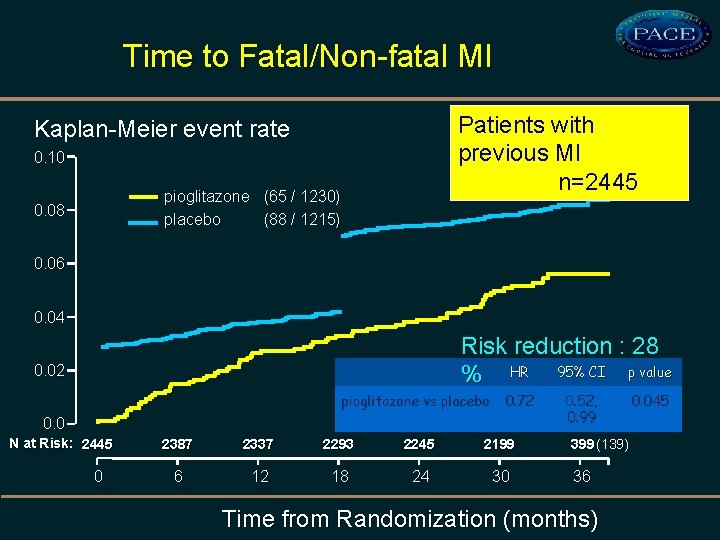
Time to Fatal/Non-fatal MI Patients with previous MI n=2445 Kaplan-Meier event rate 0. 10 pioglitazone (65 / 1230) placebo (88 / 1215) 0. 08 0. 06 0. 04 Risk reduction : 28 % HR 95% CI p value 0. 02 pioglitazone vs placebo 0. 72 0. 0 N at Risk: 2445 0 2387 2337 2293 2245 2199 6 12 18 24 30 0. 52, 0. 99 399 (139) 36 Time from Randomization (months) 0. 045

In high-risk patients with type 2 diabetes and previous stroke, pioglitazone significantly reduced the occurrence of recurrent fatal and nonfatal stroke. Stroke, 2007; 38: 865 -873
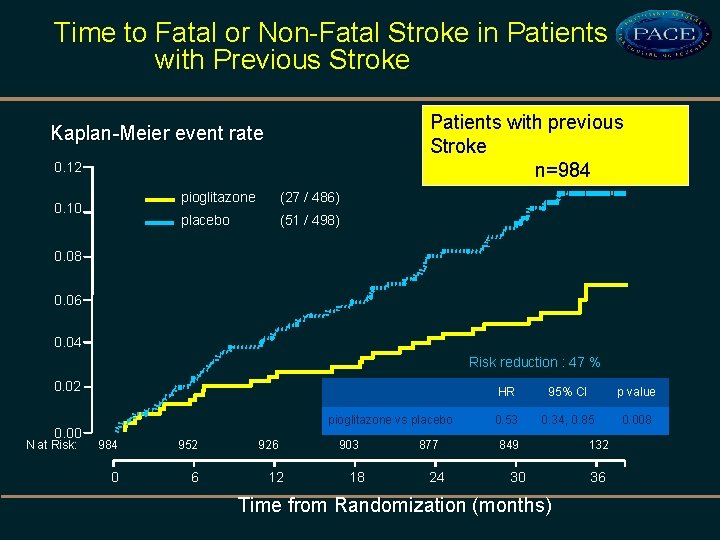
Time to Fatal or Non-Fatal Stroke in Patients with Previous Stroke Patients with previous Stroke n=984 Kaplan-Meier event rate 0. 12 0. 10 pioglitazone (27 / 486) placebo (51 / 498) 0. 08 0. 06 0. 04 Risk reduction : 47 % 0. 02 0. 00 N at Risk: pioglitazone vs placebo 984 952 0 6 926 12 903 18 877 24 HR 95% CI p value 0. 53 0. 34, 0. 85 0. 008 849 30 Time from Randomization (months) 132 36
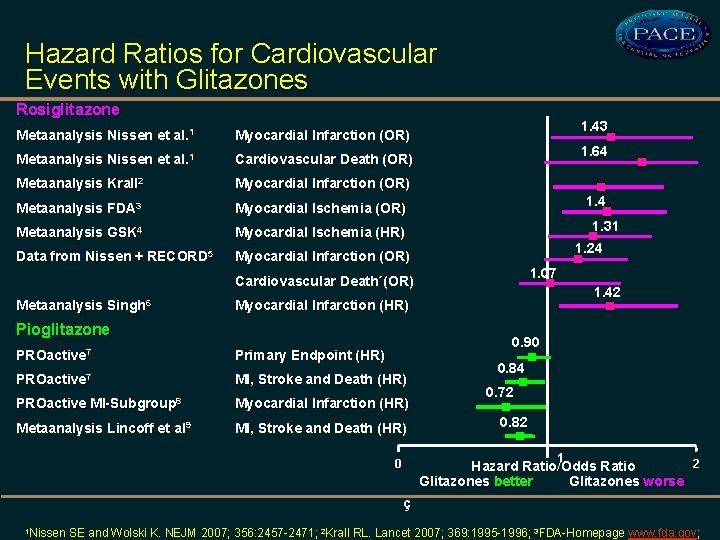
Hazard Ratios for Cardiovascular Events with Glitazones Rosiglitazone Metaanalysis Nissen et al. 1 Myocardial Infarction (OR) Metaanalysis Nissen et al. 1 Cardiovascular Death (OR) Metaanalysis Krall 2 Myocardial Infarction (OR) Metaanalysis FDA 3 Myocardial Ischemia (OR) Metaanalysis GSK 4 Myocardial Ischemia (HR) Data from Nissen + RECORD 5 Myocardial Infarction (OR) 1. 43 1. 64 1. 31 1. 24 1. 07 Cardiovascular Death´(OR) Metaanalysis Singh 6 1. 42 Myocardial Infarction (HR) Pioglitazone 0. 90 PROactive 7 Primary Endpoint (HR) PROactive 7 MI, Stroke and Death (HR) PROactive MI-Subgroup 8 Myocardial Infarction (HR) Metaanalysis Lincoff et al 9 MI, Stroke and Death (HR) 0. 84 0. 72 0. 82 1 2 Hazard Ratio/Odds Ratio Glitazones better Glitazones worse 0 ç 1 Nissen SE and Wolski K. NEJM 2007; 356: 2457 -2471; 2 Krall RL. Lancet 2007; 369: 1995 -1996; 3 FDA-Homepage www. fda. gov;
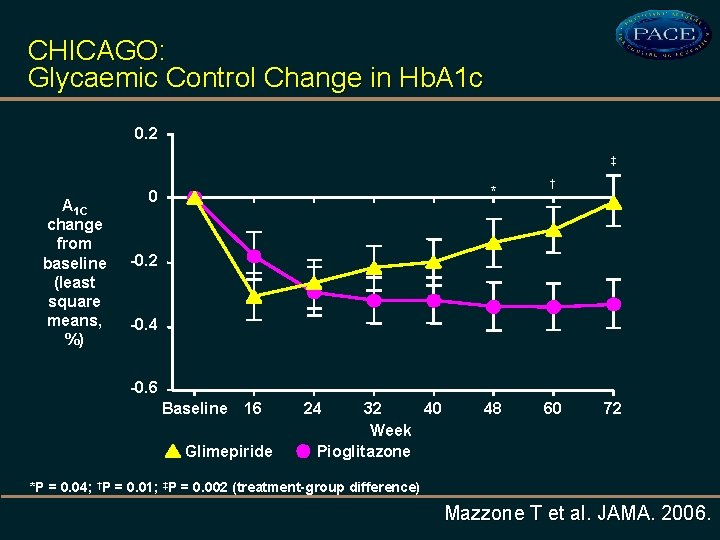
CHICAGO: Glycaemic Control Change in Hb. A 1 c 0. 2 ‡ A 1 C change from baseline (least square means, %) * 0 † -0. 2 -0. 4 -0. 6 Baseline 16 Glimepiride 24 32 40 Week Pioglitazone 48 60 72 *P = 0. 04; †P = 0. 01; ‡P = 0. 002 (treatment-group difference) Mazzone T et al. JAMA. 2006.
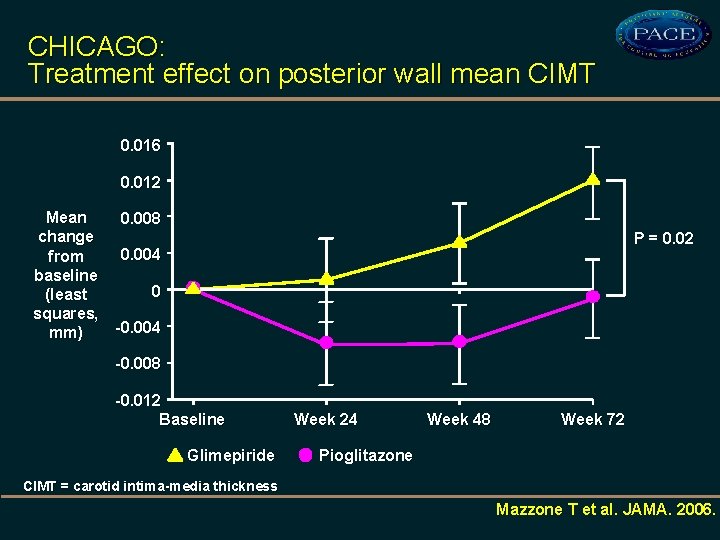
CHICAGO: Treatment effect on posterior wall mean CIMT 0. 016 0. 012 Mean 0. 008 change 0. 004 from baseline 0 (least squares, -0. 004 mm) P = 0. 02 -0. 008 -0. 012 Baseline Glimepiride Week 24 Week 48 Week 72 Pioglitazone CIMT = carotid intima-media thickness Mazzone T et al. JAMA. 2006.

Objective: Will pioglitazone stabilize carotid artery vulnerable plaque in patients with acute coronary syndromes (ACS) and type 2 diabetes. Population: 61 patients with type 2 diabetes, age 63 yrs, approx 70% male and echo lucent carotid plaques within 5 days of ACS. Random allocation to pioglitazone 15 -30 mg/day or matching placebo. Methods: Vulnerable carotid plaques were assessed by measuring plaque echolucency using carotid ultrasound with integrated back scatter (IBS) An increase in IBS reflects an increase in plaque echogenicity. Echolucent plaques with low IBS represent identify lipid and macrophage-rich lesions, unstable plaques
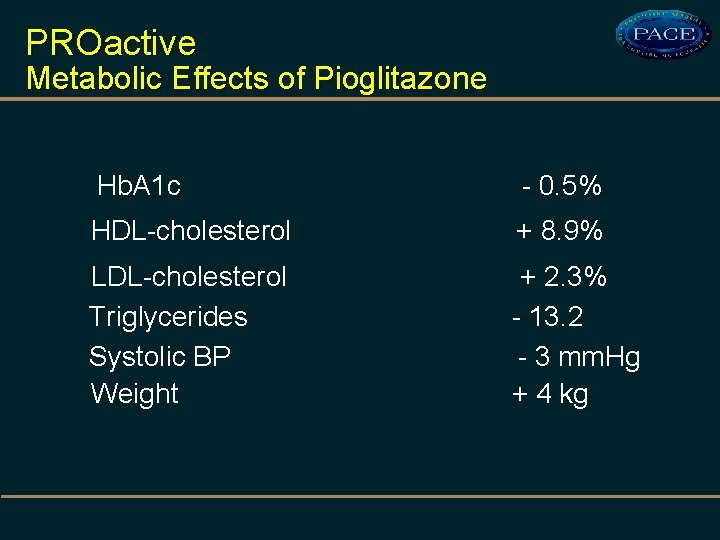
PROactive Metabolic Effects of Pioglitazone Hb. A 1 c - 0. 5% HDL-cholesterol + 8. 9% LDL-cholesterol Triglycerides Systolic BP Weight + 2. 3% - 13. 2 - 3 mm. Hg + 4 kg
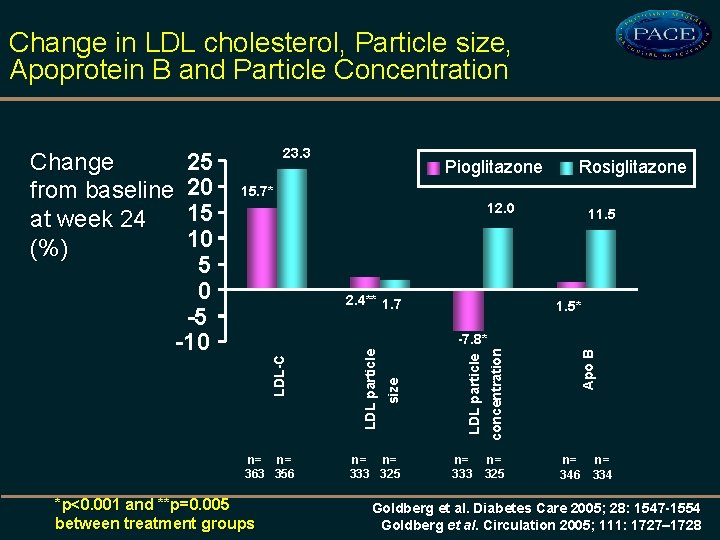
Change in LDL cholesterol, Particle size, Apoprotein B and Particle Concentration 23. 3 Pioglitazone Rosiglitazone 15. 7* 12. 0 2. 4** 1. 7 11. 5* n= n= 363 356 *p<0. 001 and **p=0. 005 between treatment groups n= n= 333 325 n= 333 n= 325 Apo B concentration LDL particle size -7. 8* LDL particle 25 20 15 10 5 0 -5 -10 LDL-C Change from baseline at week 24 (%) n= 346 n= 334 Goldberg et al. Diabetes Care 2005; 28: 1547 -1554 Goldberg et al. Circulation 2005; 111: 1727– 1728
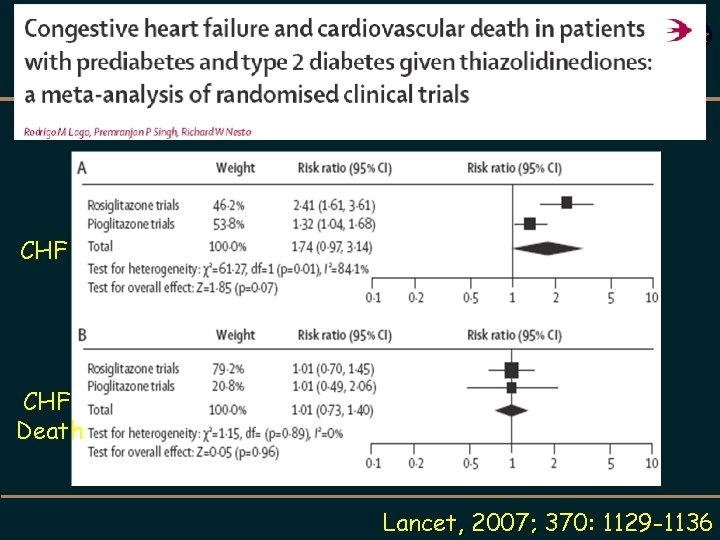
CHF Death Lancet, 2007; 370: 1129 -1136
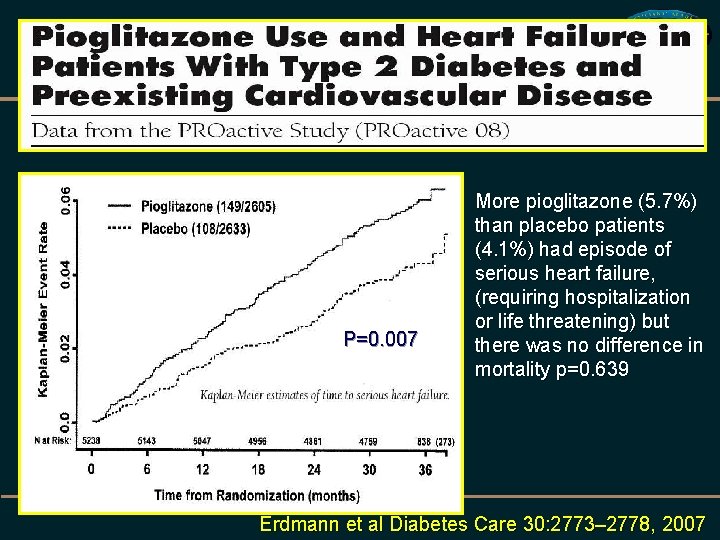
P=0. 007 More pioglitazone (5. 7%) than placebo patients (4. 1%) had episode of serious heart failure, (requiring hospitalization or life threatening) but there was no difference in mortality p=0. 639 Erdmann et al Diabetes Care 30: 2773– 2778, 2007
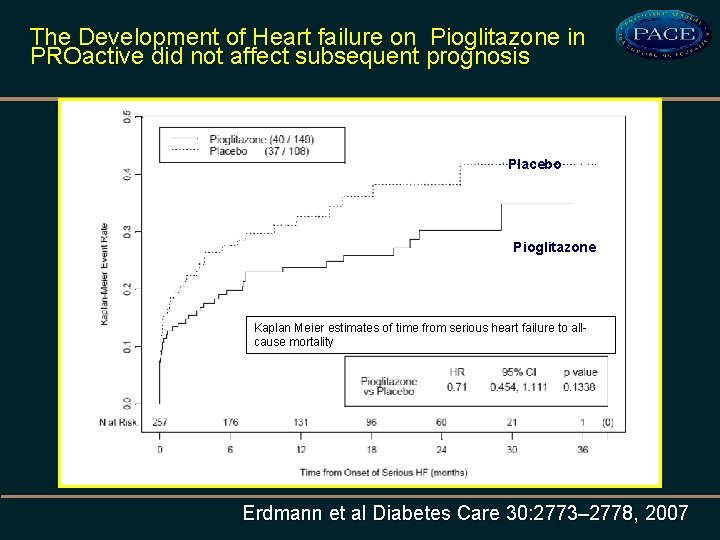
The Development of Heart failure on Pioglitazone in PROactive did not affect subsequent prognosis Placebo Pioglitazone Kaplan Meier estimates of time from serious heart failure to allcause mortality Erdmann et al Diabetes Care 30: 2773– 2778, 2007

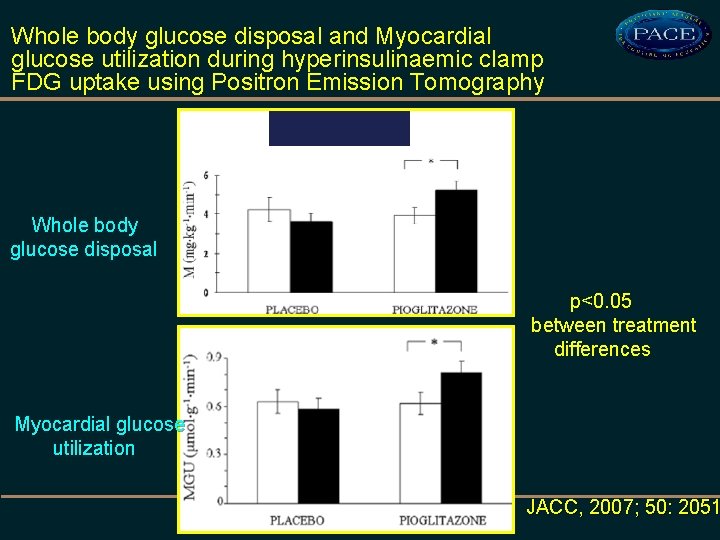
Whole body glucose disposal and Myocardial glucose utilization during hyperinsulinaemic clamp FDG uptake using Positron Emission Tomography Whole body glucose disposal p<0. 05 between treatment differences Myocardial glucose utilization JACC, 2007; 50: 2051
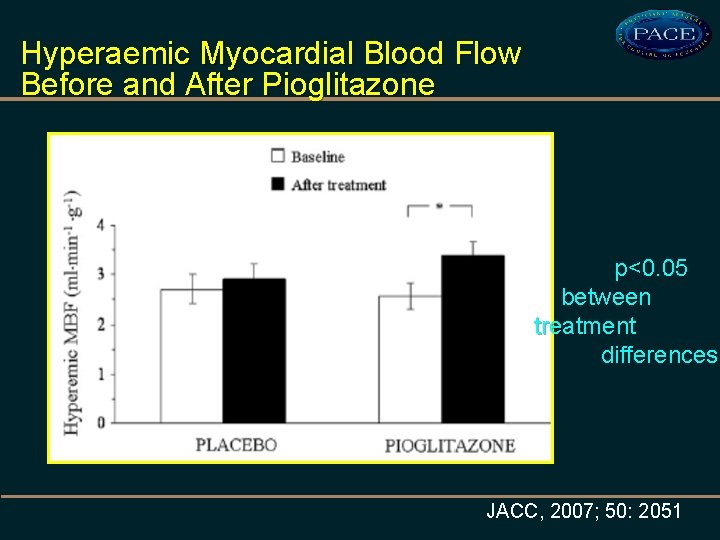
Hyperaemic Myocardial Blood Flow Before and After Pioglitazone p<0. 05 between treatment differences JACC, 2007; 50: 2051

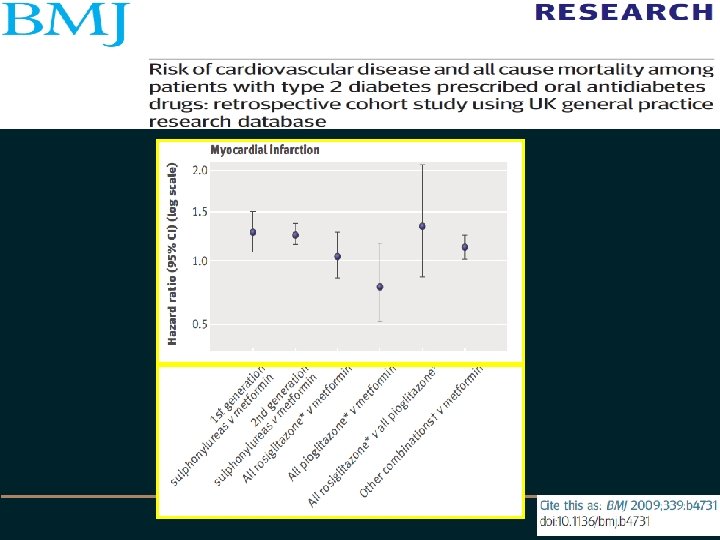
• • Setting: UK general practice research dta Objective: To investigate risk of incident MI, congestive heart failure and all cause mortality associated with prescription of oral anti diabetes drugs Design: Retrospective cohort study Setting: UK general practice research data base 1990 -2005 Participants: 91521 people with diabetes Methods: Person time intervals for drug treatment were categorised by drug class excluding non-drug intervals and intervals for insulin 3588 MIs, 6900 CCF, 18548 deaths
- Slides: 31