Cardiac Bioelectricity and Arrhythmia Center CBAC Jordi Heijman
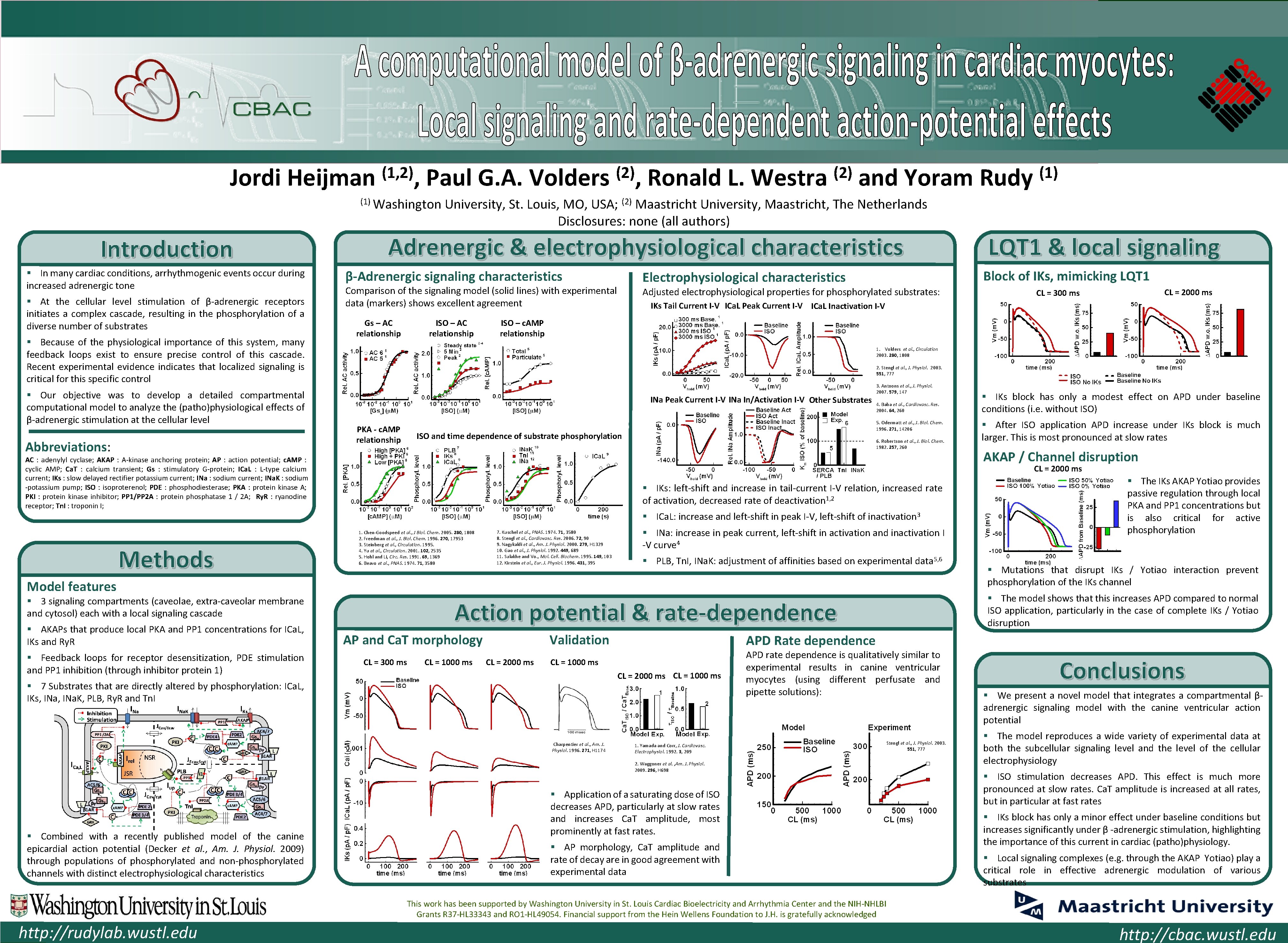
Cardiac Bioelectricity and Arrhythmia Center (CBAC) Jordi Heijman (1, 2), Paul G. A. Volders (1) Washington Introduction § In many cardiac conditions, arrhythmogenic events occur during increased adrenergic tone § At the cellular level stimulation of β-adrenergic receptors initiates a complex cascade, resulting in the phosphorylation of a diverse number of substrates § Because of the physiological importance of this system, many feedback loops exist to ensure precise control of this cascade. Recent experimental evidence indicates that localized signaling is critical for this specific control § Our objective was to develop a detailed compartmental computational model to analyze the (patho)physiological effects of β-adrenergic stimulation at the cellular level Abbreviations: Ronald L. Westra (2) and Yoram Rudy University, St. Louis, MO, USA; Maastricht University, Maastricht, The Netherlands Disclosures: none (all authors) Adrenergic & electrophysiological characteristics β-Adrenergic signaling characteristics ISO – AC relationship Adjusted electrophysiological properties for phosphorylated substrates: ISO – c. AMP relationship 1. Volders et al. , Circulation 2003. 280, 1808 2. Stengl et al. , J. Physiol. 2003. 551, 777 INa Peak Current I-V INa In/Activation I-V Other Substrates PKA - c. AMP relationship 3. Antoons et al. , J. Physiol. 2007. 579, 147 4. Baba et al. , Cardiovasc. Res. 2004. 64, 260 5. Odermatt et al. , J. Biol. Chem. 1996. 271, 14206 ISO and time dependence of substrate phosphorylation 6. Robertson et al. , J. Biol. Chem. 1982. 257, 260 § IKs: left-shift and increase in tail-current I-V relation, increased rate of activation, decreased rate of deactivation 1, 2 § ICa. L: increase and left-shift in peak I-V, left-shift of inactivation 3 1. Chen-Goodspeed et al. , J Biol. Chem. 2005. 280, 1808 2. Freedman et al. , J. Biol. Chem. 1996. 270, 17953 3. Steinberg et al. , Circulation. 1995. 4. Yu et al. , Circulation. 2001. 102, 2535 5. Hohl and Li, Circ. Res. 1991. 69, 1369 6. Beavo et al. , PNAS. 1974. 71, 3580 7. Kuschel et al. , PNAS. 1974. 71, 3580 8. Stengl et al. , Cardiovasc. Res. 2006. 72, 90 9. Nagykaldi et al. , Am. J. Physiol. 2000. 279, H 1329 10. Gao et al. , J. Physiol. 1992. 449, 689 11. Sulakhe and Vo. , Mol. Cell. Biochem. 1995. 149, 103 12. Kirstein et al. , Eur. J. Physiol. 1996. 431, 395 § INa: increase in peak current, left-shift in activation and inactivation I -V curve 4 § PLB, Tn. I, INa. K: adjustment of affinities based on experimental data 5, 6 § After ISO application APD increase under IKs block is much larger. This is most pronounced at slow rates AKAP / Channel disruption AP and Ca. T morphology CL = 300 ms CL = 1000 ms Validation CL = 2000 ms § Mutations that disrupt IKs / Yotiao interaction prevent phosphorylation of the IKs channel APD Rate dependence CL = 1000 ms CL = 2000 ms CL = 1000 ms Charpentier et al. , Am. J. Physiol. 1996. 271, H 1174 § The IKs AKAP Yotiao provides passive regulation through local PKA and PP 1 concentrations but is also critical for active phosphorylation § The model shows that this increases APD compared to normal ISO application, particularly in the case of complete IKs / Yotiao disruption Action potential & rate-dependence § 7 Substrates that are directly altered by phosphorylation: ICa. L, IKs, INa. K, PLB, Ry. R and Tn. I 1. Yamada and Corr, J. Cardiovasc. Electrophysiol. 1992. 3, 209 APD rate dependence is qualitatively similar to experimental results in canine ventricular myocytes (using different perfusate and pipette solutions): Stengl et al. , J. Physiol. 2003. 551, 777 2. Waggoner et al. , Am. J. Physiol. 2009. 296, H 698 § Combined with a recently published model of the canine epicardial action potential (Decker et al. , Am. J. Physiol. 2009) through populations of phosphorylated and non-phosphorylated channels with distinct electrophysiological characteristics § IKs block has only a modest effect on APD under baseline conditions (i. e. without ISO) CL = 2000 ms § 3 signaling compartments (caveolae, extra-caveolar membrane and cytosol) each with a local signaling cascade § Feedback loops for receptor desensitization, PDE stimulation and PP 1 inhibition (through inhibitor protein 1) CL = 2000 ms CL = 300 ms IKs Tail Current I-V ICa. L Peak Current I-V ICa. L Inactivation I-V Model features § AKAPs that produce local PKA and PP 1 concentrations for ICa. L, IKs and Ry. R LQT 1 & local signaling Block of IKs, mimicking LQT 1 Electrophysiological characteristics Comparison of the signaling model (solid lines) with experimental data (markers) shows excellent agreement Gs – AC relationship (1) (2) AC : adenylyl cyclase; AKAP : A-kinase anchoring protein; AP : action potential; c. AMP : cyclic AMP; Ca. T : calcium transient; Gs : stimulatory G-protein; ICa. L : L-type calcium current; IKs : slow delayed rectifier potassium current; INa : sodium current; INa. K : sodium -potassium pump; ISO : isoproterenol; PDE : phosphodiesterase; PKA : protein kinase A; PKI : protein kinase inhibitor; PP 1/PP 2 A : protein phosphatase 1 / 2 A; Ry. R : ryanodine receptor; Tn. I : troponin I; Methods (2), § Application of a saturating dose of ISO decreases APD, particularly at slow rates and increases Ca. T amplitude, most prominently at fast rates. § AP morphology, Ca. T amplitude and rate of decay are in good agreement with experimental data Conclusions § We present a novel model that integrates a compartmental βadrenergic signaling model with the canine ventricular action potential § The model reproduces a wide variety of experimental data at both the subcellular signaling level and the level of the cellular electrophysiology § ISO stimulation decreases APD. This effect is much more pronounced at slow rates. Ca. T amplitude is increased at all rates, but in particular at fast rates § IKs block has only a minor effect under baseline conditions but increases significantly under β -adrenergic stimulation, highlighting the importance of this current in cardiac (patho)physiology. § Local signaling complexes (e. g. through the AKAP Yotiao) play a critical role in effective adrenergic modulation of various substrates This work has been supported by Washington University in St. Louis Cardiac Bioelectricity and Arrhythmia Center and the NIH-NHLBI Grants R 37 -HL 33343 and RO 1 -HL 49054. Financial support from the Hein Wellens Foundation to J. H. is gratefully acknowledged http: //rudylab. wustl. edu http: //cbac. wustl. edu
- Slides: 1