CARCINOMA DELLA MAMMELLA UPDATE FROM AROMATASE INHIBITORS STUDIES

CARCINOMA DELLA MAMMELLA










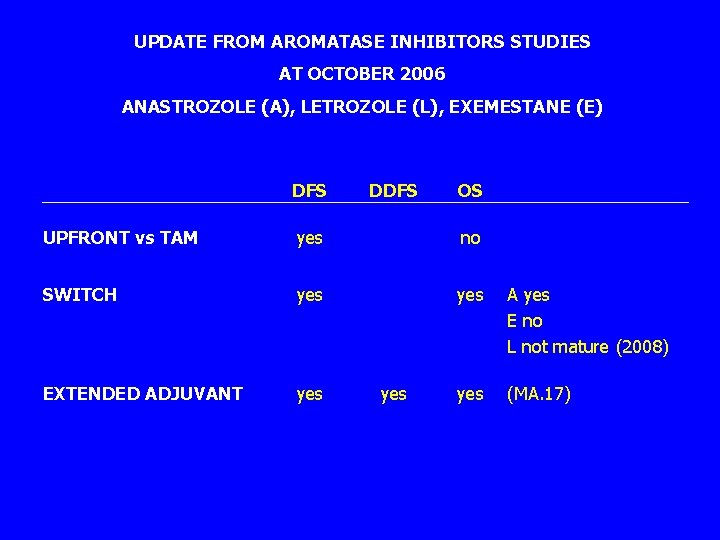
UPDATE FROM AROMATASE INHIBITORS STUDIES AT OCTOBER 2006 ANASTROZOLE (A), LETROZOLE (L), EXEMESTANE (E) DFS DDFS OS UPFRONT vs TAM yes no SWITCH yes A yes E no L not mature (2008) EXTENDED ADJUVANT yes (MA. 17) yes

What’s really new in endocrine therapy in breast cancer? Jean-Philippe SPANO, MD, Ph. D GHPS, Paris, France

The Aromatase Inhibitor Trials • • ATAC: Tamoxifen vs Anastrozole vs Combined therapy MA 17 : Letrozole vs placebo after 5 yr Tamoxifen IES : Exemestane vs Tamoxifen after 2 -3 yr Tamoxifen ITA : Anastrozole vs Tamoxifen after 2 -3 yr Tamoxifen ABCSG/ARNO : ditto BIG 1 -98 : Letrozole vs Tamoxifen TEAM: exemestane vs Tamoxifen
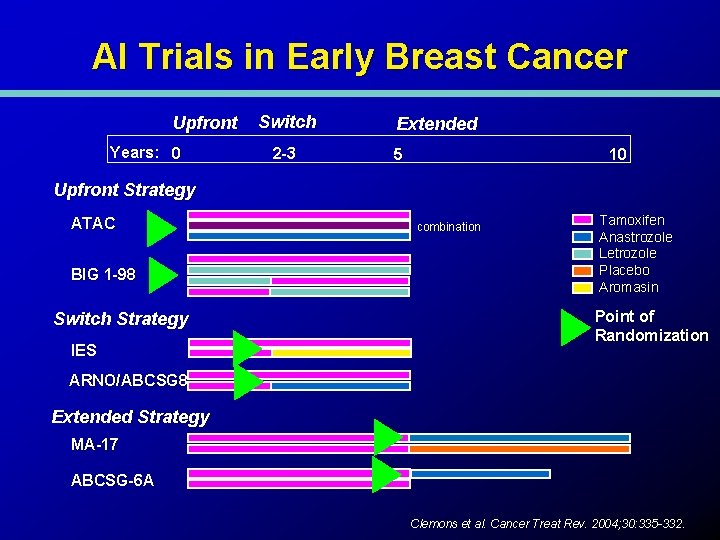
AI Trials in Early Breast Cancer Upfront Years: 0 Switch 2 -3 Extended 5 10 Upfront Strategy ATAC BIG 1 -98 Switch Strategy IES combination Tamoxifen Anastrozole Letrozole Placebo Aromasin Point of Randomization ARNO/ABCSG 8 Extended Strategy MA-17 ABCSG-6 A Clemons et al. Cancer Treat Rev. 2004; 30: 335 -332.

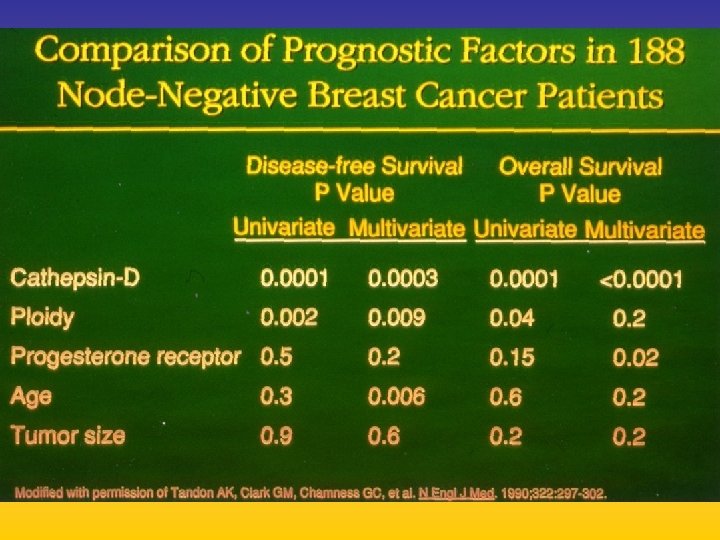
ATAC • Recruitment July 1996 – March 2000 • Median follow up 68 months (data cut 31 st March 2004) • 8 % of patients remain on trial therapy Tamoxifen (n=3, 116) Surgery ± RT ± Chemo (20 %) • 84 % HR positive • 61 % Node negative Reference Anastrozole (n=3, 125) Discontinued following initial Arimidex Tamoxifen analysis as+no efficacy or tolerability benefit compared (n=3, 125) with tamoxifen arm 5 years
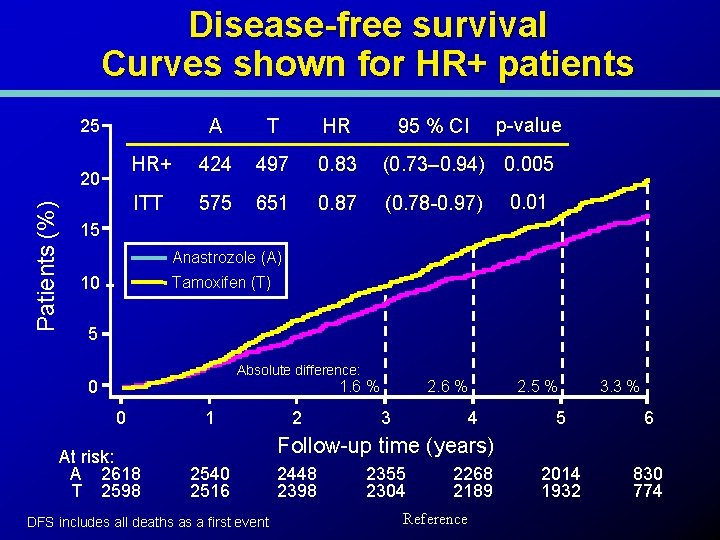
Disease-free survival Curves shown for HR+ patients Patients (%) 20 95 % CI p-value A T HR HR+ 424 497 0. 83 (0. 73– 0. 94) 0. 005 ITT 575 651 0. 87 (0. 78 -0. 97) 25 0. 01 15 Anastrozole (A) 10 Tamoxifen (T) 5 Absolute difference: 0 1. 6 % 0 At risk: A 2618 T 2598 1 2 2. 6 % 3 4 2. 5 % 3. 3 % 5 6 2014 1932 830 774 Follow-up time (years) 2540 2516 DFS includes all deaths as a first event 2448 2398 2355 2304 2268 2189 Reference
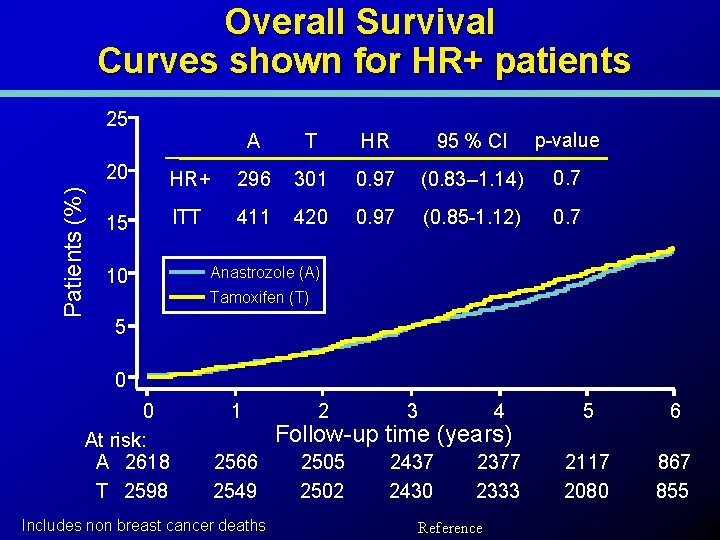
Overall Survival Curves shown for HR+ patients Patients (%) 25 A T HR 95 % CI p-value 20 HR+ 296 301 0. 97 (0. 83– 1. 14) 0. 7 15 ITT 411 420 0. 97 (0. 85 -1. 12) 0. 7 10 Anastrozole (A) Tamoxifen (T) 5 0 0 At risk: A 2618 T 2598 1 2566 2549 Includes non breast cancer deaths 2 3 4 5 6 2505 2502 2437 2430 2377 2333 2117 2080 867 855 Follow-up time (years) Reference
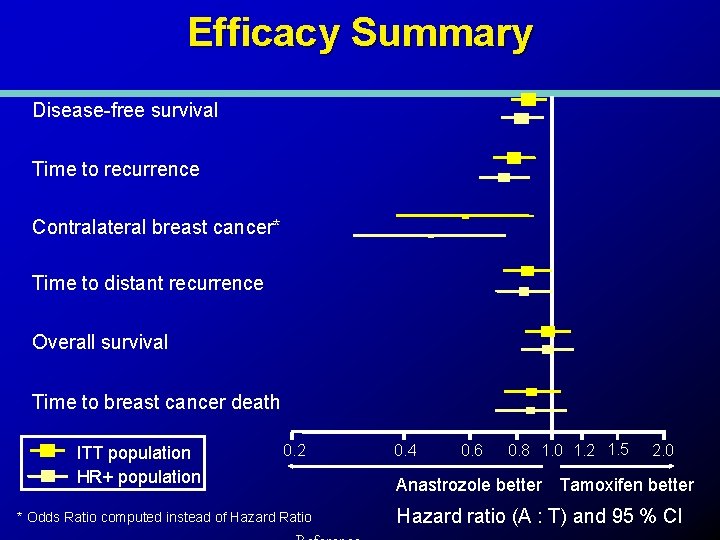
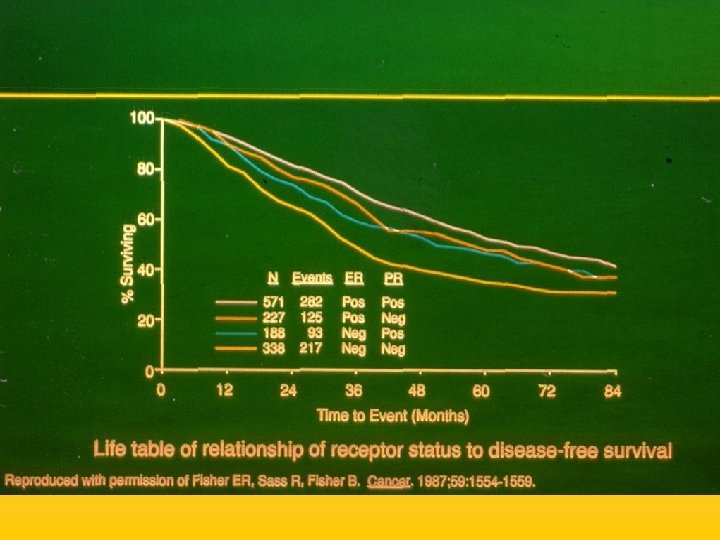
Efficacy Summary Disease-free survival Time to recurrence Contralateral breast cancer* Time to distant recurrence Overall survival Time to breast cancer death ITT population HR+ population 0. 2 * Odds Ratio computed instead of Hazard Ratio 0. 4 0. 6 0. 8 1. 0 1. 2 1. 5 2. 0 Anastrozole better Tamoxifen better Hazard ratio (A : T) and 95 % CI
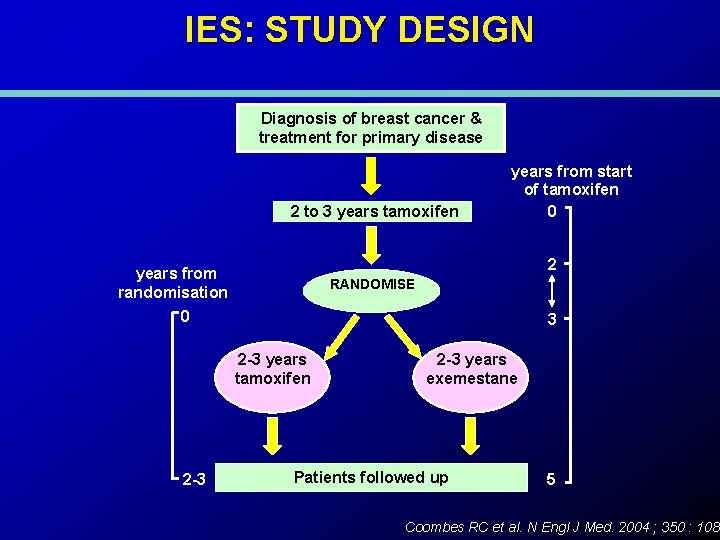
IES: STUDY DESIGN Diagnosis of breast cancer & treatment for primary disease 2 to 3 years tamoxifen 2 years from randomisation 0 RANDOMISE 3 2 -3 years tamoxifen 2 -3 years from start of tamoxifen 0 2 -3 years exemestane Patients followed up 5 Coombes RC et al. N Engl J Med. 2004 ; 350 : 1081

Why This Study ? • Patients with ER positive metastatic disease frequently respond to AI’s after tamoxifen • Tamoxifen pre-treatment can increase bone density • Long-term tamoxifen cause endometrial cancer • Prior neo-adjuvant tamoxifen studies show that patients frequently relapse after 2 -3 years
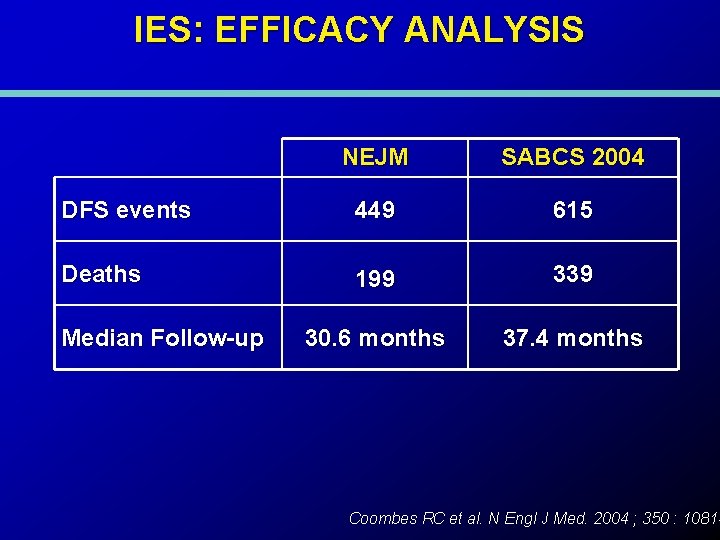
IES: EFFICACY ANALYSIS NEJM SABCS 2004 DFS events 449 615 Deaths 199 339 30. 6 months 37. 4 months Median Follow-up Coombes RC et al. N Engl J Med. 2004 ; 350 : 1081 -

IES : EVENTS CONTRIBUTING TO DFS Exemestane Tamoxifen Total Local recurrence only† 43 56 99 Distant recurrence 150 208 358 Contralateral breast primary 12 26 38 Intercurrent deaths (without recurrence) 57 63 120 Total number of patients experiencing an event 262 353 615 † Includes 1 ipsilateral breast cancer

IES : DISEASE FREE SURVIVAL Hazard Ratio 95 % CI* P value Disease free survival 0. 73 0. 62 -0. 86 0. 0001 Breast cancer free survival 0. 70 0. 58 -0. 83 0. 00005 Time to contralateral breast cancer 0. 50 0. 26 -0. 97 0. 04 * CI denotes confidence interval
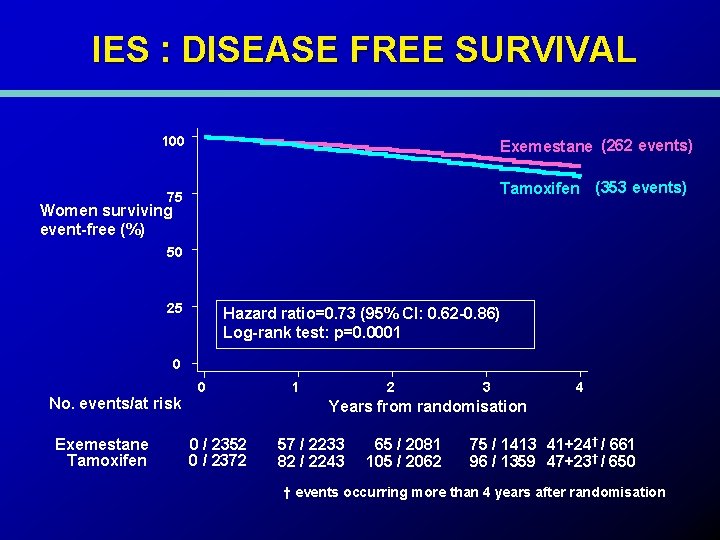
IES : DISEASE FREE SURVIVAL 100 Exemestane (262 events) Tamoxifen (353 events) 75 Women surviving event-free (%) 50 25 Hazard ratio=0. 73 (95% CI: 0. 62 -0. 86) Log-rank test: p=0. 0001 0 0 No. events/at risk Exemestane Tamoxifen 1 2 3 4 Years from randomisation 0 / 2352 0 / 2372 57 / 2233 82 / 2243 65 / 2081 105 / 2062 75 / 1413 41+24† / 661 96 / 1359 47+23† / 650 † events occurring more than 4 years after randomisation
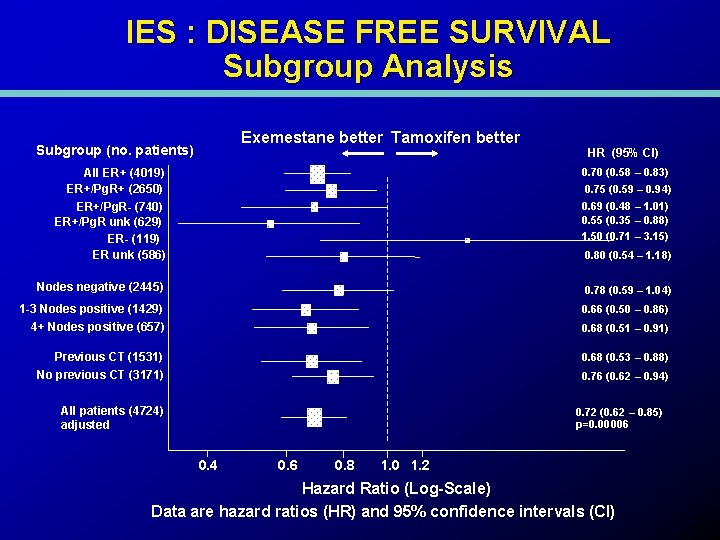
IES : DISEASE FREE SURVIVAL Subgroup Analysis Exemestane better Tamoxifen better Subgroup (no. patients) HR (95% CI) 0. 70 (0. 58 – 0. 83) All ER+ (4019) ER+/Pg. R+ (2650) ER+/Pg. R- (740) ER+/Pg. R unk (629) ER- (119) ER unk (586) 0. 75 (0. 59 – 0. 94) 0. 69 (0. 48 – 1. 01) 0. 55 (0. 35 – 0. 88) 1. 50 (0. 71 – 3. 15) 0. 80 (0. 54 – 1. 18) Nodes negative (2445) 0. 78 (0. 59 – 1. 04) 1 -3 Nodes positive (1429) 4+ Nodes positive (657) 0. 66 (0. 50 – 0. 86) Previous CT (1531) No previous CT (3171) 0. 68 (0. 53 – 0. 88) 0. 68 (0. 51 – 0. 91) 0. 76 (0. 62 – 0. 94) All patients (4724) adjusted 0. 72 (0. 62 – 0. 85) p=0. 00006 0. 4 0. 6 0. 8 1. 0 1. 2 Hazard Ratio (Log-Scale) Data are hazard ratios (HR) and 95% confidence intervals (CI)
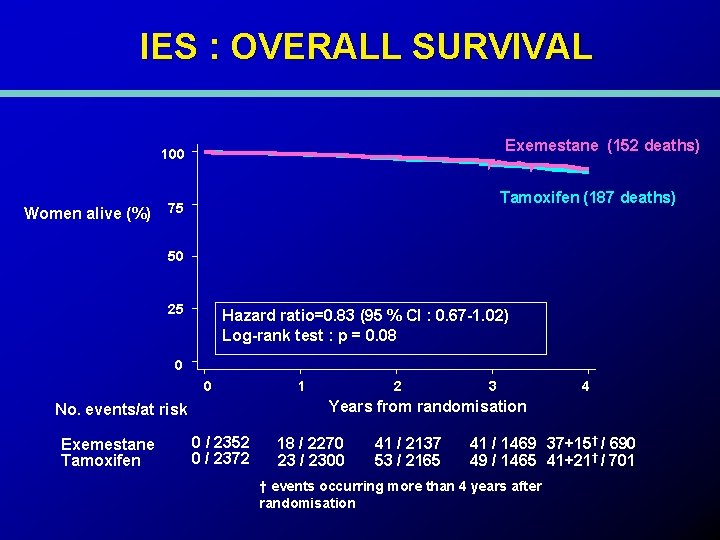
IES : OVERALL SURVIVAL Exemestane (152 deaths) 100 Women alive (%) Tamoxifen (187 deaths) 75 50 25 Hazard ratio=0. 83 (95 % CI : 0. 67 -1. 02) Log-rank test : p = 0. 08 0 0 2 3 4 Years from randomisation No. events/at risk Exemestane Tamoxifen 1 0 / 2352 0 / 2372 18 / 2270 23 / 2300 41 / 2137 53 / 2165 41 / 1469 37+15† / 690 49 / 1465 41+21† / 701 † events occurring more than 4 years after randomisation
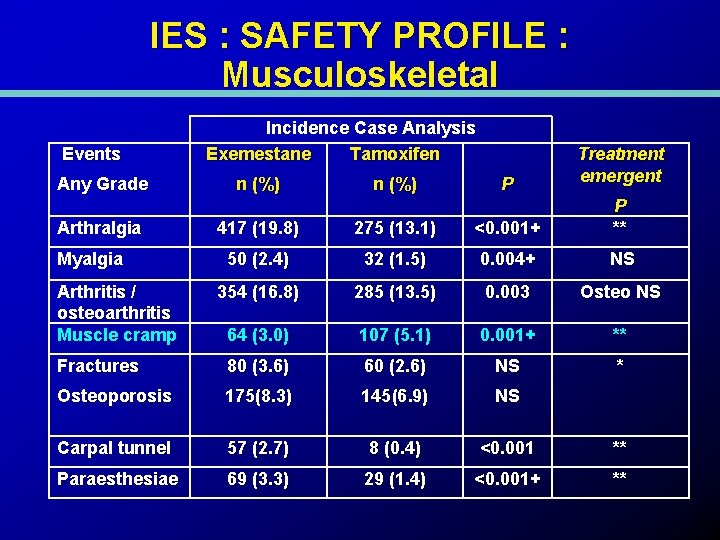
IES : SAFETY PROFILE : Musculoskeletal Events Any Grade Incidence Case Analysis Exemestane Tamoxifen n (%) P Treatment emergent 417 (19. 8) 275 (13. 1) <0. 001+ P ** 50 (2. 4) 32 (1. 5) 0. 004+ NS 354 (16. 8) 285 (13. 5) 0. 003 Osteo NS 64 (3. 0) 107 (5. 1) 0. 001+ ** Fractures 80 (3. 6) 60 (2. 6) NS * Osteoporosis 175(8. 3) 145(6. 9) NS Carpal tunnel 57 (2. 7) 8 (0. 4) <0. 001 ** Paraesthesiae 69 (3. 3) 29 (1. 4) <0. 001+ ** Arthralgia Myalgia Arthritis / osteoarthritis Muscle cramp
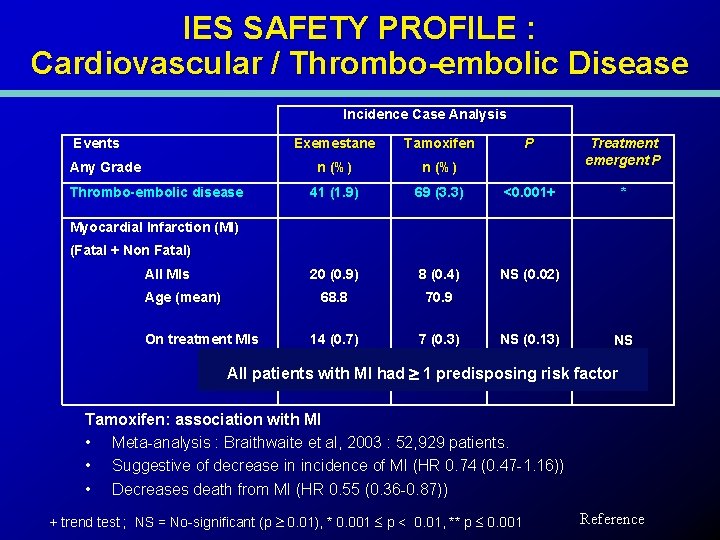
IES SAFETY PROFILE : Cardiovascular / Thrombo-embolic Disease Incidence Case Analysis Events Any Grade Thrombo-embolic disease Exemestane Tamoxifen P Treatment emergent P n (%) 41 (1. 9) 69 (3. 3) <0. 001+ * 20 (0. 9) 8 (0. 4) NS (0. 02) 68. 8 70. 9 14 (0. 7) 7 (0. 3) Myocardial Infarction (MI) (Fatal + Non Fatal) All MIs Age (mean) On treatment MIs NS (0. 13) NS All patients with MI had 1 predisposing risk factor Tamoxifen: association with MI • Meta-analysis : Braithwaite et al, 2003 : 52, 929 patients. • Suggestive of decrease in incidence of MI (HR 0. 74 (0. 47 -1. 16)) • Decreases death from MI (HR 0. 55 (0. 36 -0. 87)) + trend test ; NS = No-significant (p 0. 01), * 0. 001 p < 0. 01, ** p 0. 001 Reference

IES : EFFICACY CONCLUSIONS • Switching to exemestane reduces the risk of: – – breast cancer recurrence or death (p=0. 0001) contralateral breast cancer (p=0. 04) • Switching to exemestane appears to reduce the chances of dying (p=0. 08) but more follow-up is needed
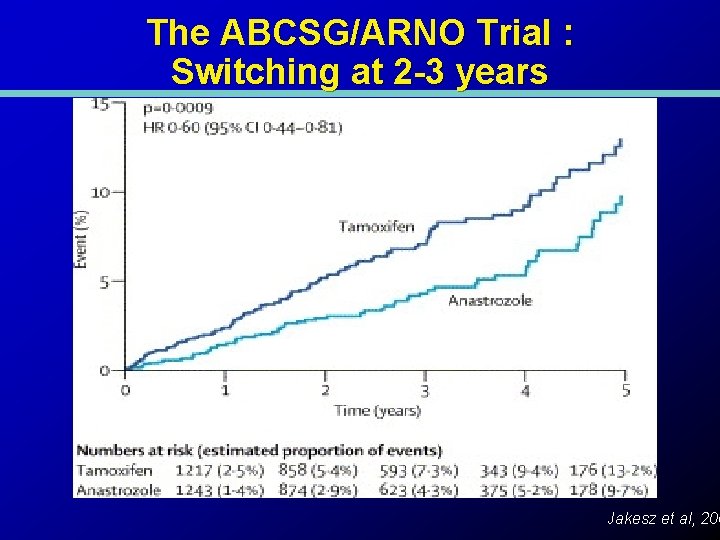
The ABCSG/ARNO Trial : Switching at 2 -3 years Jakesz et al, 200
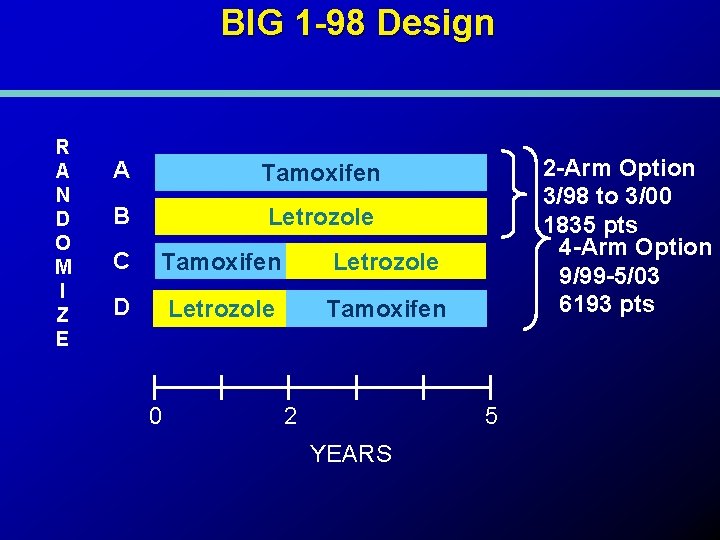
BIG 1 -98 Design R A N D O M I Z E A Tamoxifen B Letrozole C Tamoxifen Letrozole D Letrozole Tamoxifen 0 2 2 -Arm Option 3/98 to 3/00 1835 pts 4 -Arm Option 9/99 -5/03 6193 pts 5 YEARS

Primary Core Analysis 8028 Randomized 18 withdrew consent (no treatment / FU) 8010 Primary Core Analysis 4003 L versus 4007 T 133 (1. 66%) ineligible cases included in primary core analysis
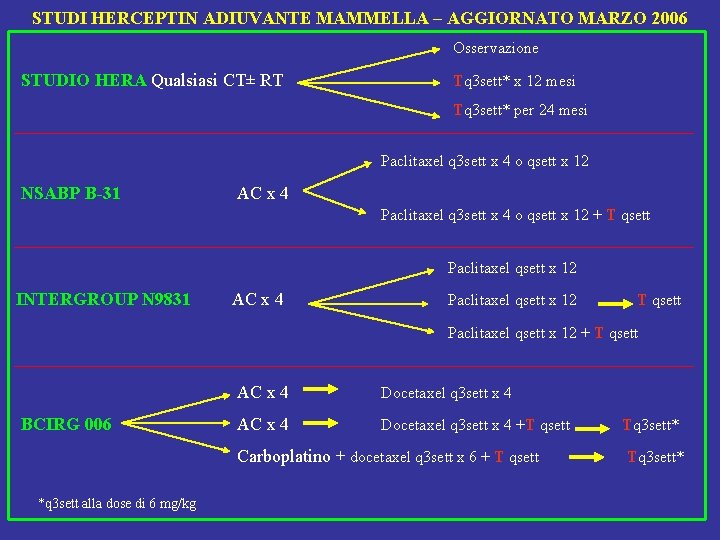
STUDI HERCEPTIN ADIUVANTE MAMMELLA – AGGIORNATO MARZO 2006 Osservazione STUDIO HERA Qualsiasi CT± RT Tq 3 sett* x 12 mesi Tq 3 sett* per 24 mesi Paclitaxel q 3 sett x 4 o qsett x 12 NSABP B-31 AC x 4 Paclitaxel q 3 sett x 4 o qsett x 12 + T qsett Paclitaxel qsett x 12 INTERGROUP N 9831 AC x 4 Paclitaxel qsett x 12 T qsett Paclitaxel qsett x 12 + T qsett BCIRG 006 AC x 4 Docetaxel q 3 sett x 4 +T qsett Carboplatino + docetaxel q 3 sett x 6 + T qsett *q 3 sett alla dose di 6 mg/kg Tq 3 sett*

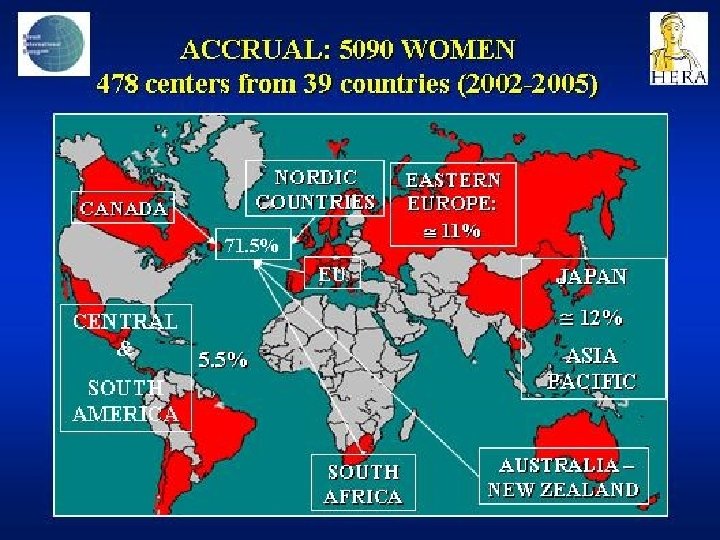
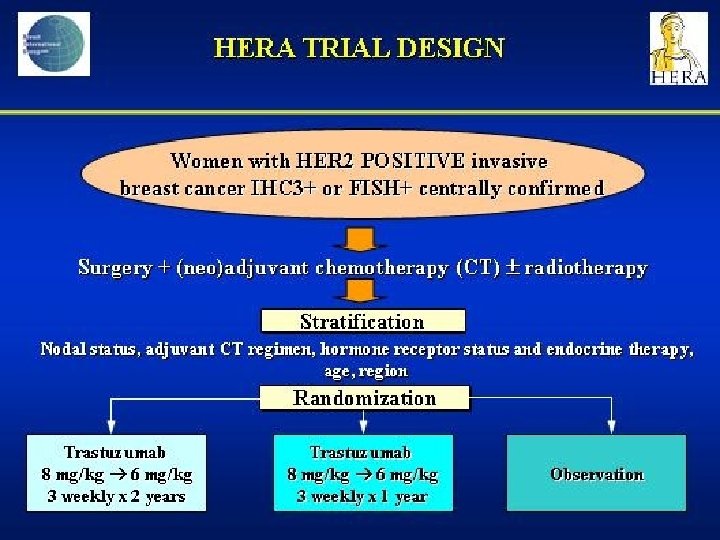

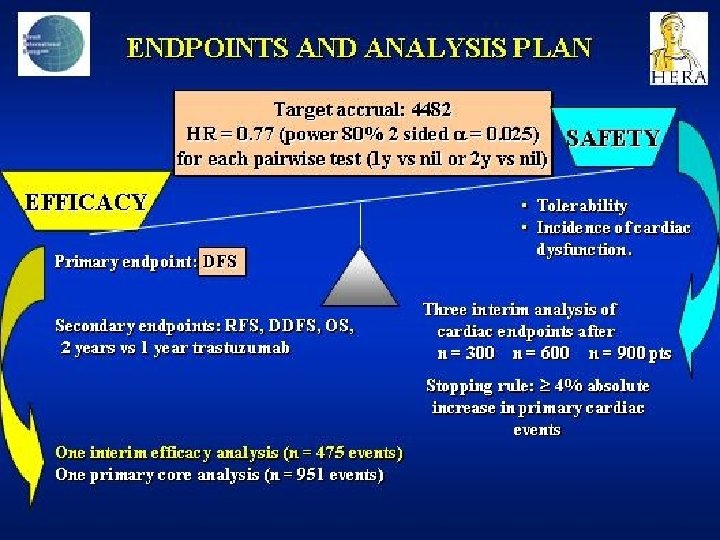
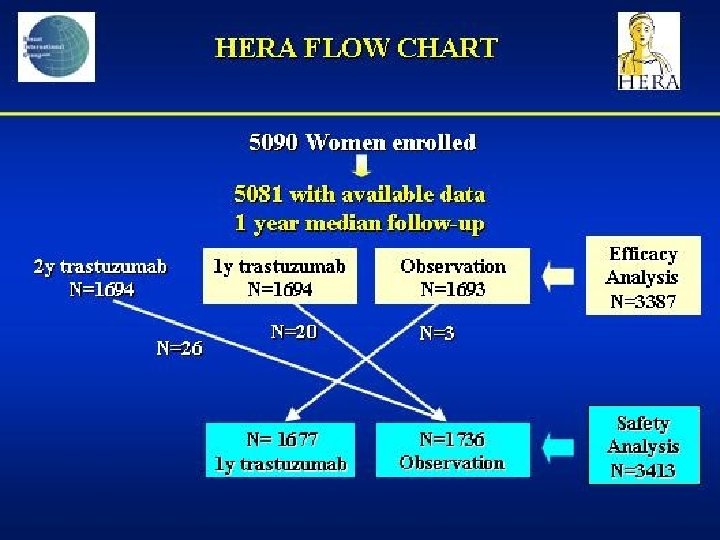
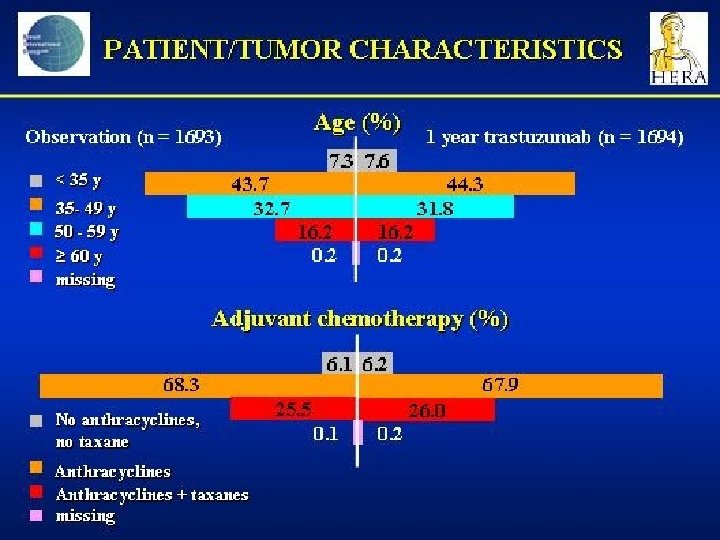

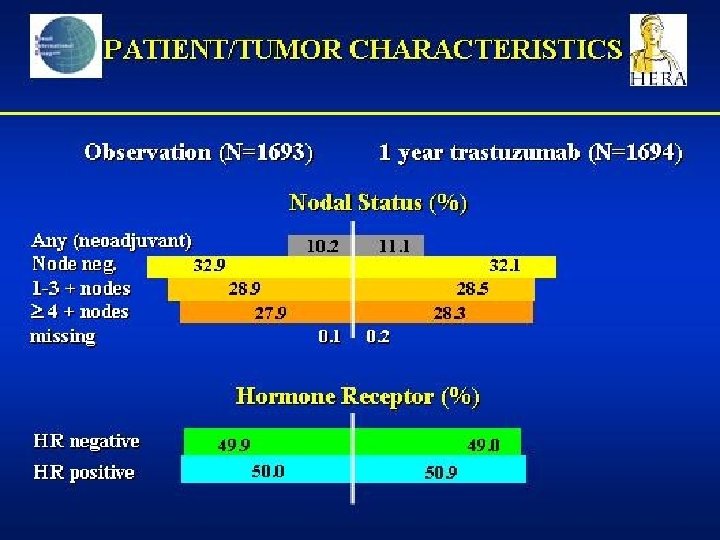

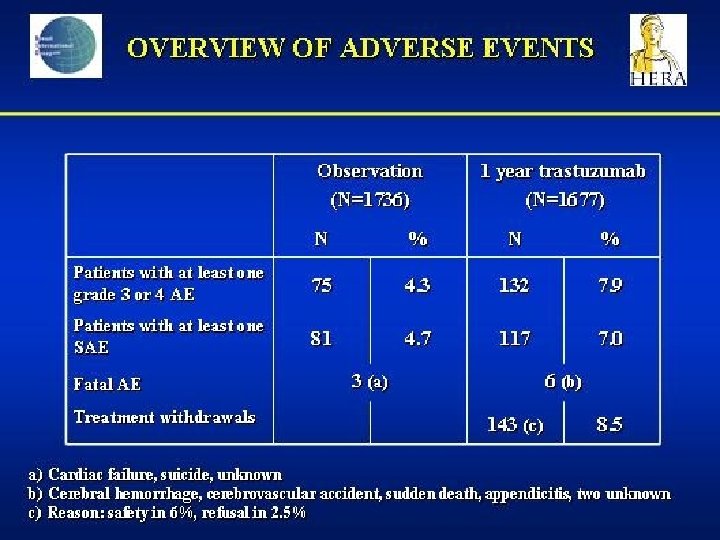
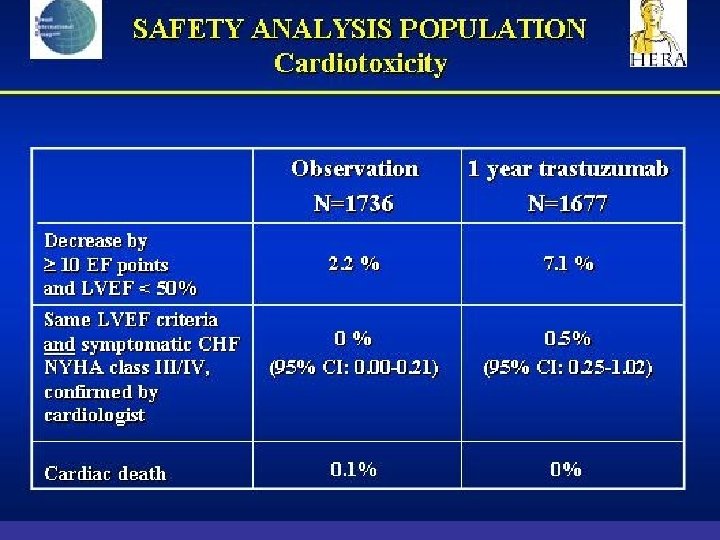
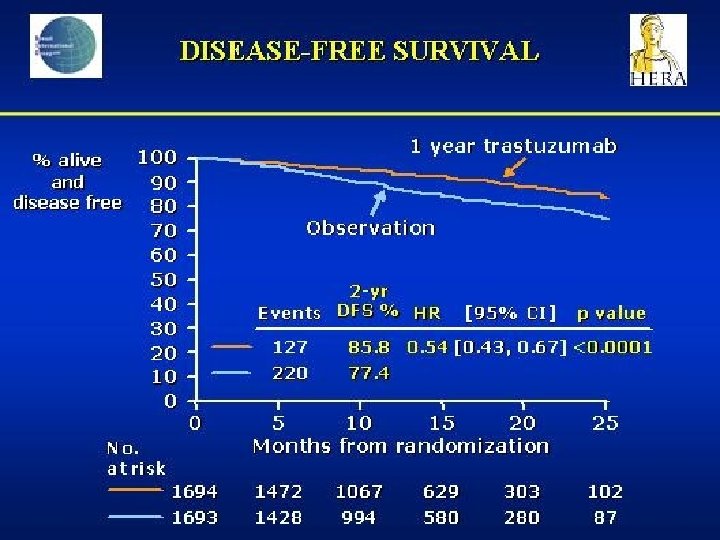
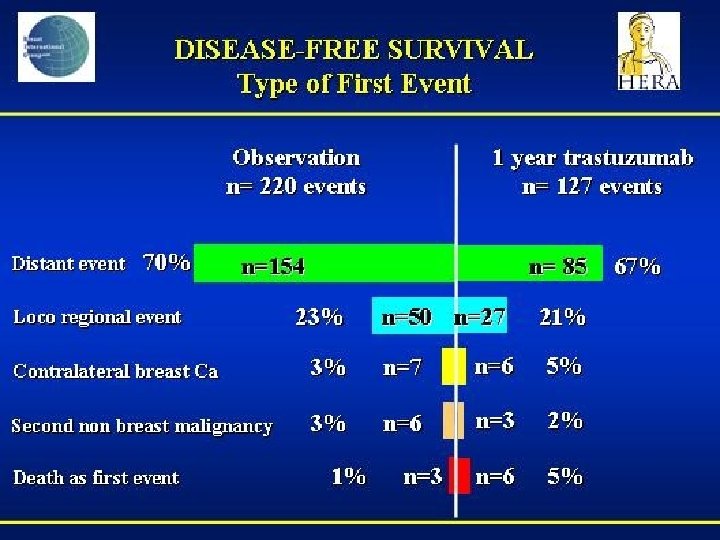
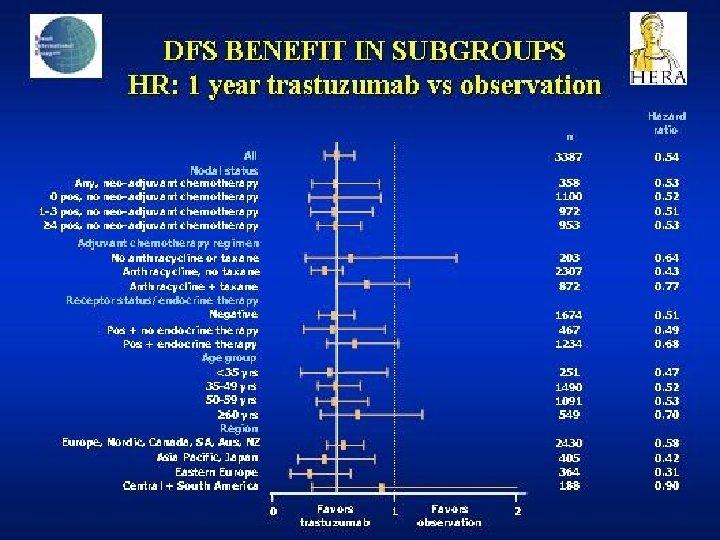
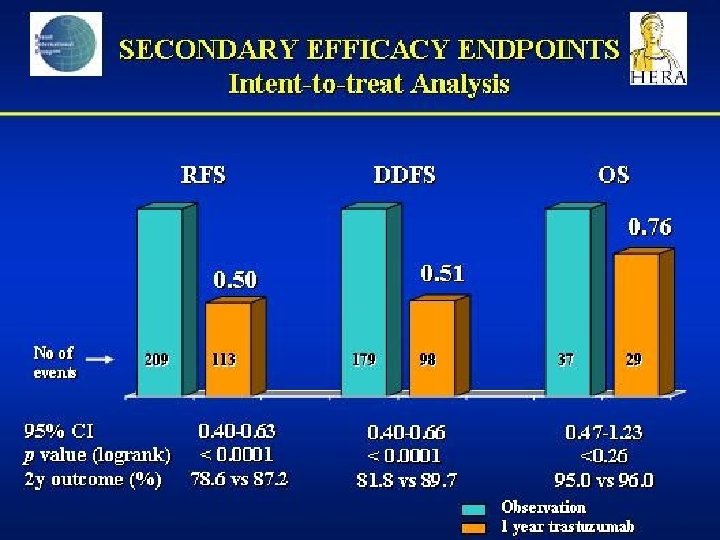




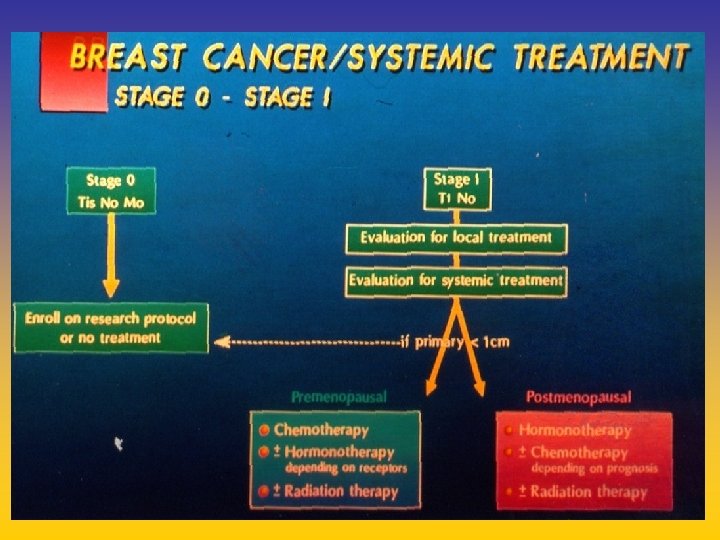
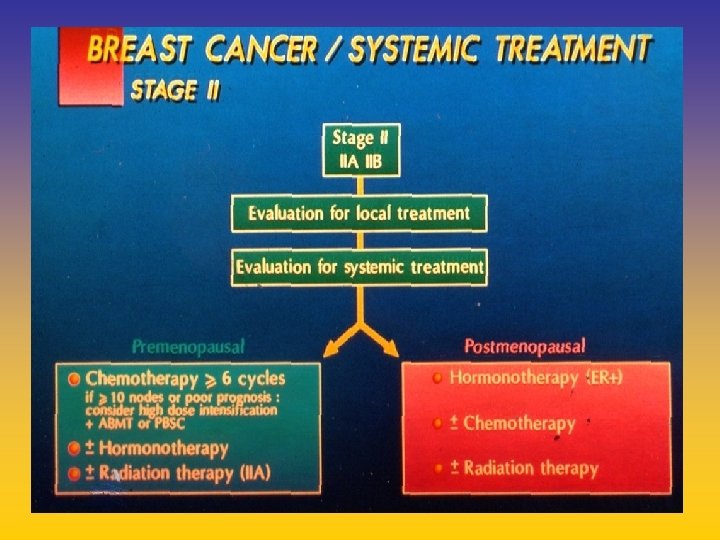
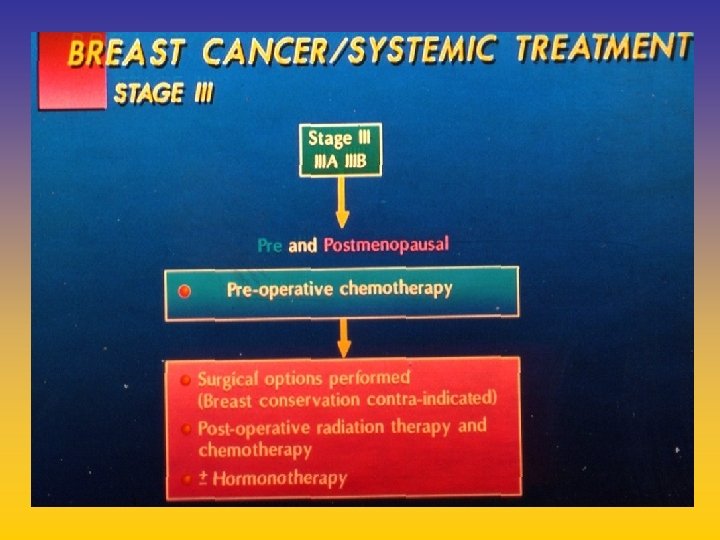








































- Slides: 142