Carboxymethylated kappacarrageenan for intestinaltargeted delivery of oral insulin

Carboxymethylated kappacarrageenan for intestinal-targeted delivery of oral insulin Ph. D candidate: Leong Kok Hoong Supervisors: Prof. Dr. Chung Lip Yong Assoc. Prof. Dr. Mohamed Ibrahim Noordin Department of Pharmacy, Faculty of Medicine, University of Malaya 1

Diabetes and insulin therapy Diabetes is projected to affect 366 million people in 2030, with the greatest increase from developing countries. 1 Underlying cause of the disease is impaired secretion of insulin and/or insensitive towards insulin. 2 Insulin is a protein hormone secreted from the pancreas into the liver. 2 Promotes the entry of glucose into cells and enhances the production of glycogen. 2 2

Routes of insulin administration To counteract low levels of insulin, long-term multiple insulin injections are administered. 3 High risk of dose error, hypoglycemia, infection and fat deposit at injection sites. 3 In 2006/2007, first commercially available inhaled-insulin, Exubera by Pfizer. But, withdrawn one year later. 4 Increase in insulin antibody, inconvenient inhaler and not suitable for smokers or asthmatic patients. 4 Withdrawal of other inhaled-insulin such as AERx by Novo Nordisk and AIR Insulin by Eli Lilly. 5 3
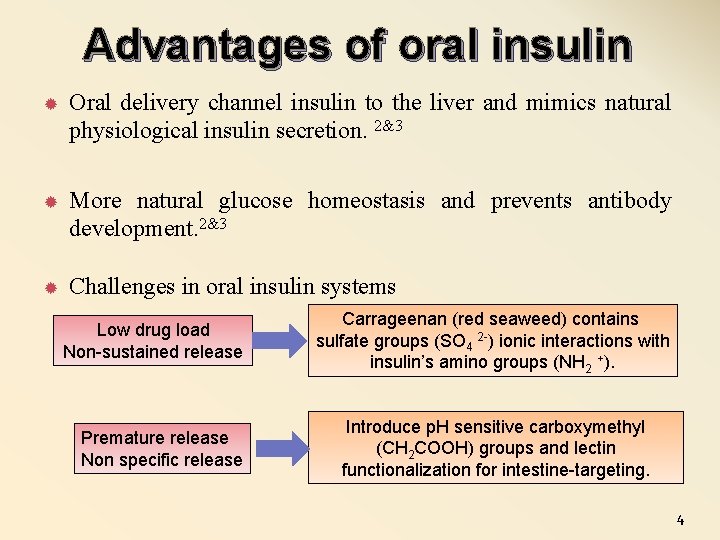
Advantages of oral insulin Oral delivery channel insulin to the liver and mimics natural physiological insulin secretion. 2&3 More natural glucose homeostasis and prevents antibody development. 2&3 Challenges in oral insulin systems Low drug load Non-sustained release Premature release Non specific release Carrageenan (red seaweed) contains sulfate groups (SO 4 2 -) ionic interactions with insulin’s amino groups (NH 2 +). Introduce p. H sensitive carboxymethyl (CH 2 COOH) groups and lectin functionalization for intestine-targeting. 4
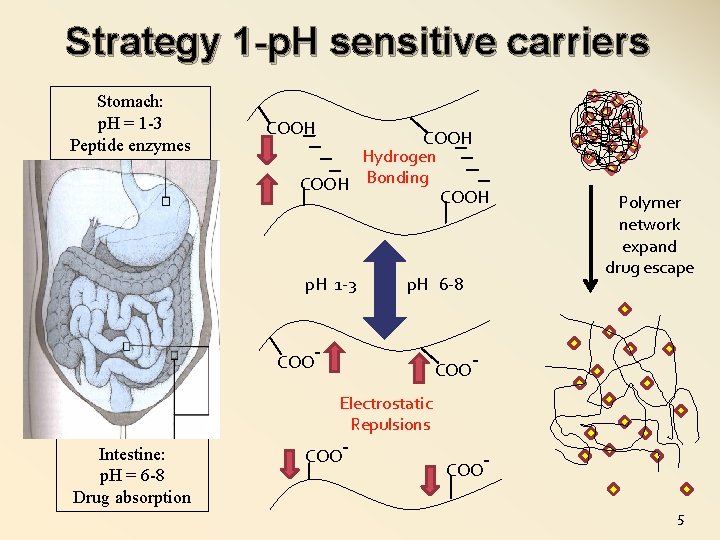
Strategy 1 -p. H sensitive carriers Stomach: p. H = 1 -3 Peptide enzymes COOH Hydrogen COOH Bonding COOH p. H 1 -3 COO p. H 6 -8 - COO Polymer network expand drug escape - Electrostatic Repulsions Intestine: p. H = 6 -8 Drug absorption COO - COO 5
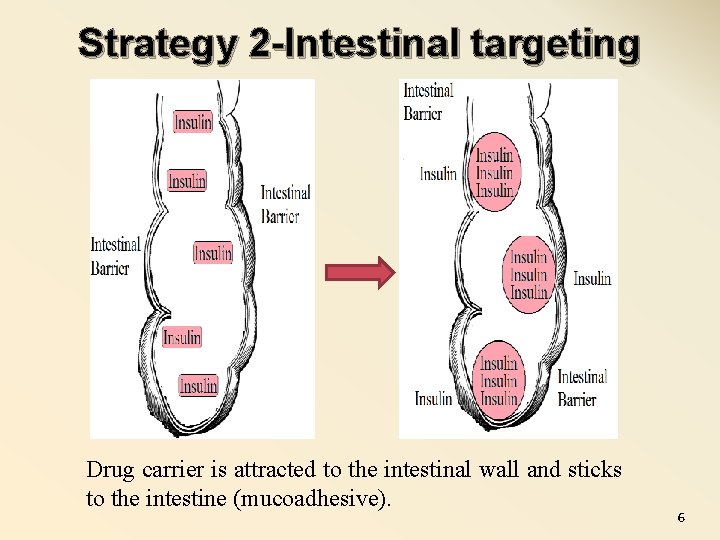
Strategy 2 -Intestinal targeting Drug carrier is attracted to the intestinal wall and sticks to the intestine (mucoadhesive). 6

Research Objectives 1. Optimization of carboxymethylation on kappa-carrageenan and encapsulation using FD-4 (FD-4 is Fluorescein isothiocyanate labelled dextran with average molecular weight of 4, 400). 2. Optimization of insulin encapsulation and lectin functionalization. 3. Cytotoxicity of the oral delivery system on human intestinal cell model. 4. In-vivo insulin bioavailability and blood glucose lowering effect in animal model. 7

Part 1 Optimization of carboxymethylation on kappa-carrageenan and encapsulation of a FD-4 marker 8
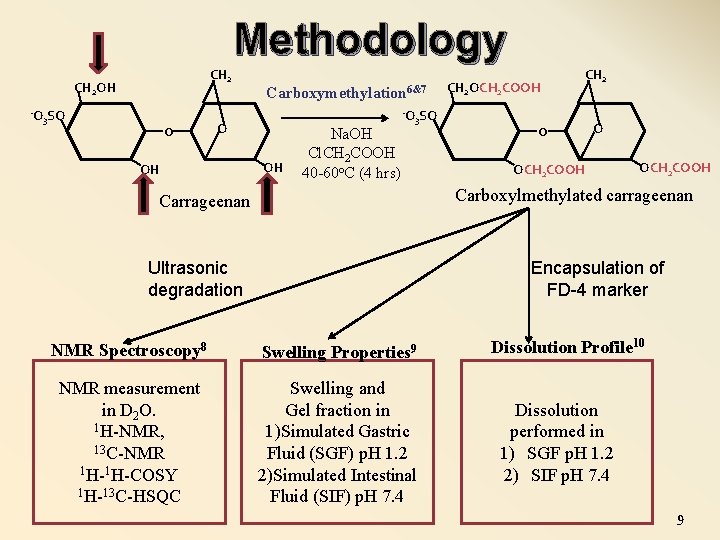
Methodology CH 2 OH -O 3 SO o Carboxymethylation 6&7 -O O OH OH Na. OH Cl. CH 2 COOH 40 -60 o. C (4 hrs) 3 SO CH 2 OCH 2 COOH o CH 2 O OCH 2 COOH Carboxylmethylated carrageenan Carrageenan Ultrasonic degradation Encapsulation of FD-4 marker NMR Spectroscopy 8 Swelling Properties 9 NMR measurement in D 2 O. 1 H-NMR, 13 C-NMR 1 H-1 H-COSY 1 H-13 C-HSQC Swelling and Gel fraction in 1)Simulated Gastric Fluid (SGF) p. H 1. 2 2)Simulated Intestinal Fluid (SIF) p. H 7. 4 Dissolution Profile 10 Dissolution performed in 1) SGF p. H 1. 2 2) SIF p. H 7. 4 9
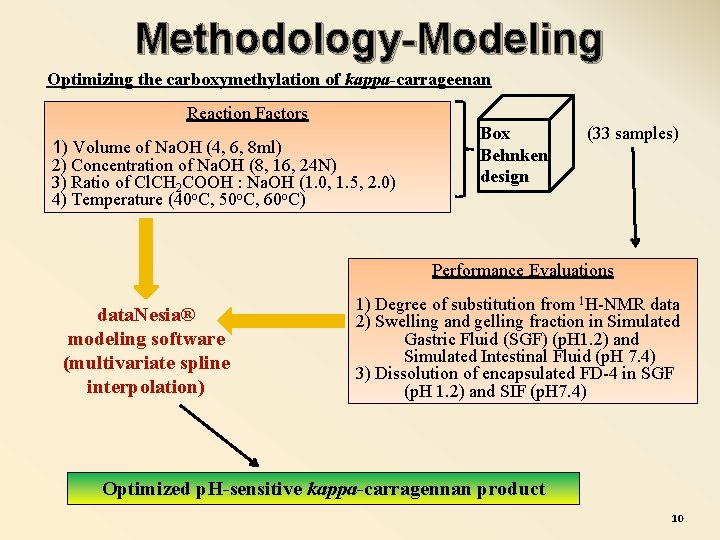
Methodology-Modeling Optimizing the carboxymethylation of kappa-carrageenan Reaction Factors 1) Volume of Na. OH (4, 6, 8 ml) 2) Concentration of Na. OH (8, 16, 24 N) 3) Ratio of Cl. CH 2 COOH : Na. OH (1. 0, 1. 5, 2. 0) 4) Temperature (40 o. C, 50 o. C, 60 o. C) Box Behnken design (33 samples) Performance Evaluations data. Nesia® modeling software (multivariate spline interpolation) 1) Degree of substitution from 1 H-NMR data 2) Swelling and gelling fraction in Simulated Gastric Fluid (SGF) (p. H 1. 2) and Simulated Intestinal Fluid (p. H 7. 4) 3) Dissolution of encapsulated FD-4 in SGF (p. H 1. 2) and SIF (p. H 7. 4) Optimized p. H-sensitive kappa-carragennan product 10
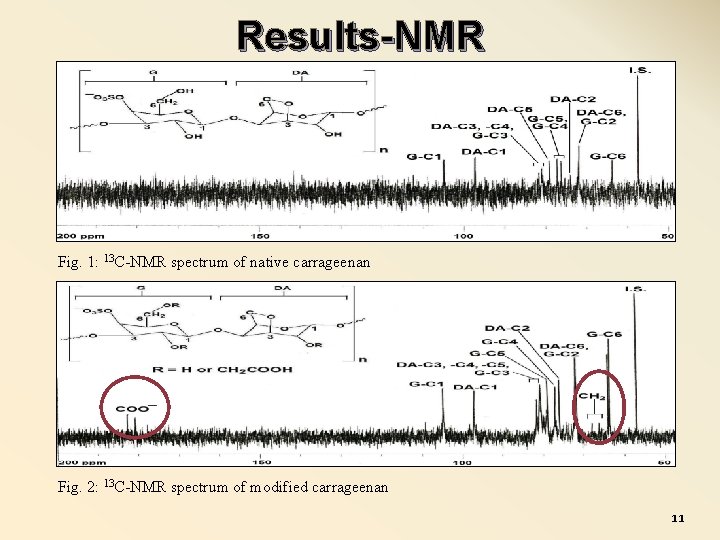
Results-NMR Fig. 1: 13 C-NMR spectrum of native carrageenan Fig. 2: 13 C-NMR spectrum of modified carrageenan 11
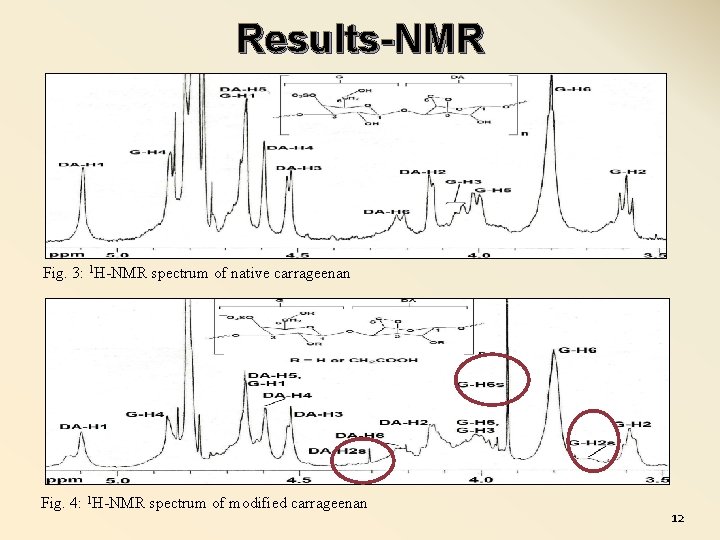
Results-NMR Fig. 3: 1 H-NMR spectrum of native carrageenan Fig. 4: 1 H-NMR spectrum of modified carrageenan 12
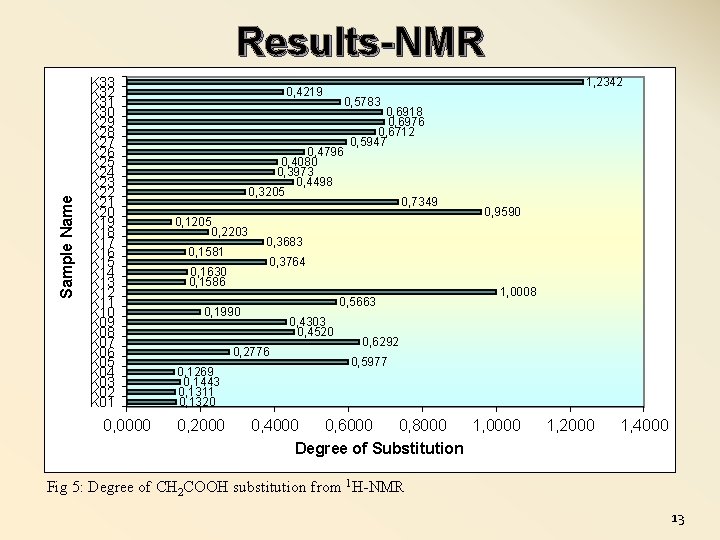
Sample Name Results-NMR K 33 K 32 K 31 K 30 K 29 K 28 K 27 K 26 K 25 K 24 K 23 K 22 K 21 K 20 K 19 K 18 K 17 K 16 K 15 K 14 K 13 K 12 K 11 K 10 K 09 K 08 K 07 K 06 K 05 K 04 K 03 K 02 K 01 0, 0000 0, 4219 1, 2342 0, 5783 0, 4796 0, 4080 0, 3973 0, 4498 0, 3205 0, 1205 0, 2203 0, 1581 0, 9590 0, 3683 0, 5663 0, 1990 0, 4303 0, 4520 0, 2776 0, 2000 0, 7349 0, 3764 0, 1630 0, 1586 0, 1269 0, 1443 0, 1311 0, 1320 0, 6918 0, 6976 0, 6712 0, 5947 1, 0008 0, 6292 0, 5977 0, 4000 0, 6000 0, 8000 1, 0000 Degree of Substitution 1, 2000 1, 4000 Fig 5: Degree of CH 2 COOH substitution from 1 H-NMR 13
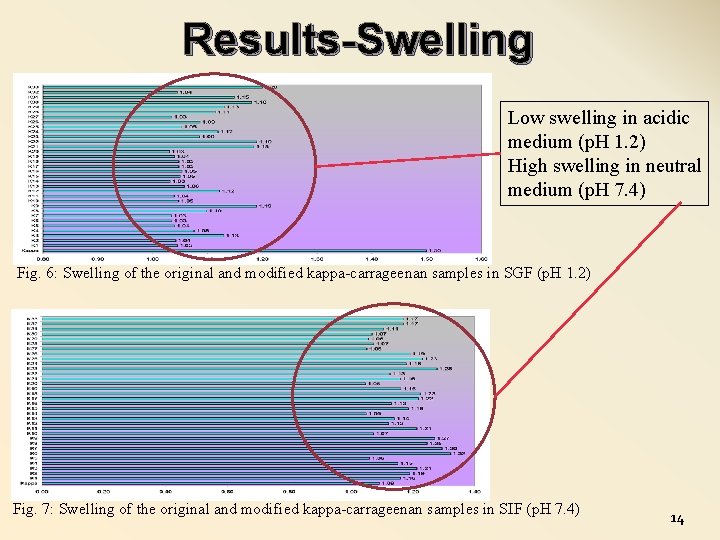
Results-Swelling Low swelling in acidic medium (p. H 1. 2) High swelling in neutral medium (p. H 7. 4) Fig. 6: Swelling of the original and modified kappa-carrageenan samples in SGF (p. H 1. 2) Fig. 7: Swelling of the original and modified kappa-carrageenan samples in SIF (p. H 7. 4) 14
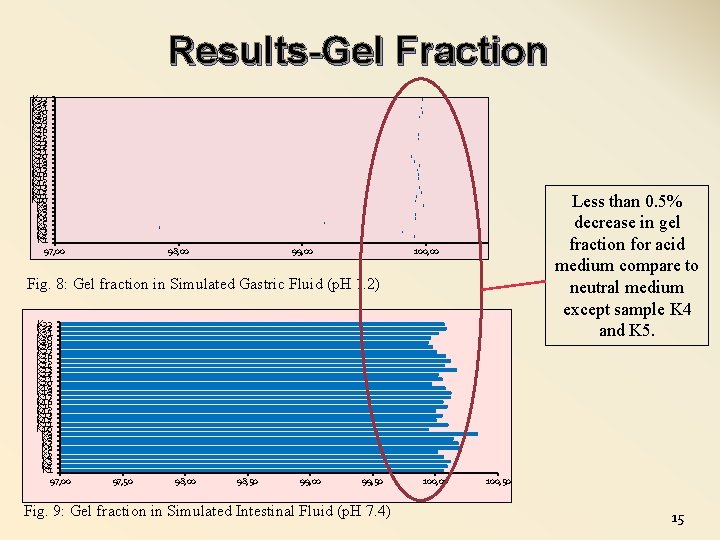
Results-Gel Fraction K 33 K 32 K 31 K 30 K 29 K 28 K 27 K 26 K 25 K 24 K 23 K 22 K 21 K 20 K 19 K 18 K 17 K 16 K 15 K 14 K 13 K 12 K 11 K 10 K 9 K 8 K 7 K 6 K 5 K 4 K 3 K 2 K 1 97, 00 98, 00 99, 00 Less than 0. 5% decrease in gel fraction for acid medium compare to neutral medium except sample K 4 and K 5. 100, 00 Fig. 8: Gel fraction in Simulated Gastric Fluid (p. H 1. 2) K 33 K 32 K 31 K 30 K 29 K 28 K 27 K 26 K 25 K 24 K 23 K 22 K 21 K 20 K 19 K 18 K 17 K 16 K 15 K 14 K 13 K 12 K 11 K 10 K 9 K 8 K 7 K 6 K 5 K 4 K 3 K 2 K 1 97, 00 97, 50 98, 00 98, 50 99, 00 99, 50 Fig. 9: Gel fraction in Simulated Intestinal Fluid (p. H 7. 4) 100, 00 100, 50 15
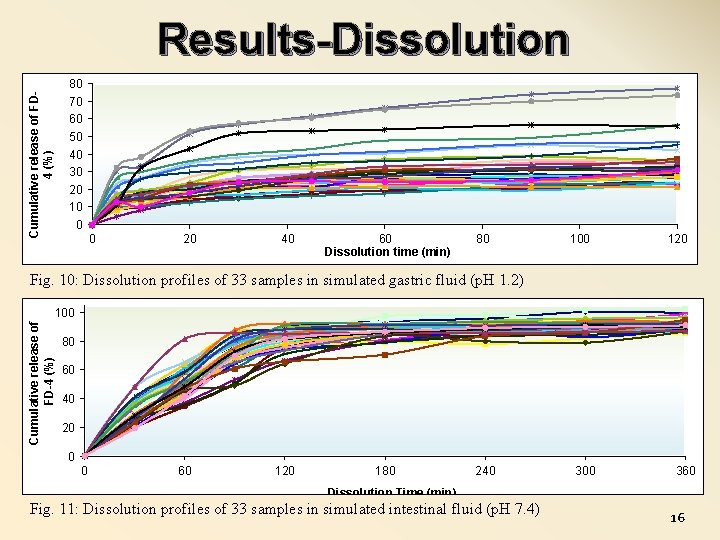
Results-Dissolution Cumulative release of FD 4 (%) 80 70 60 50 40 30 20 10 0 0 20 40 60 Dissolution time (min) 80 100 120 Fig. 10: Dissolution profiles of 33 samples in simulated gastric fluid (p. H 1. 2) Cumulative release of FD-4 (%) 100 80 60 40 20 0 0 60 120 180 240 300 360 Dissolution Time (min) Fig. 11: Dissolution profiles of 33 samples in simulated intestinal fluid (p. H 7. 4) 16
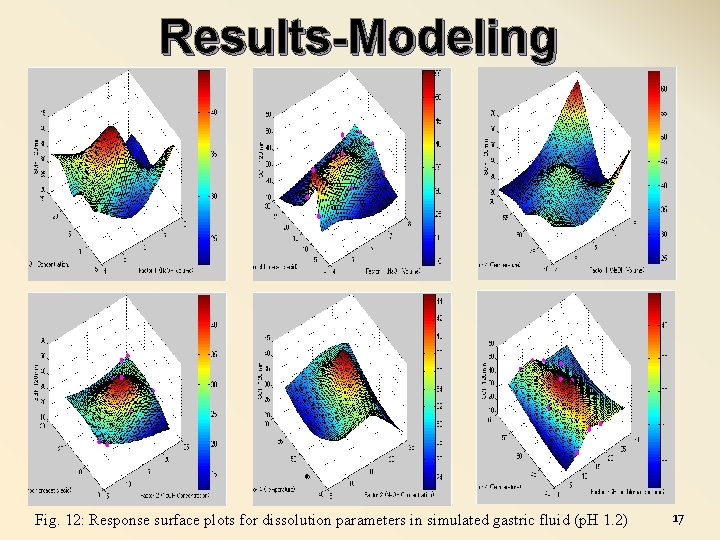
Results-Modeling Fig. 12: Response surface plots for dissolution parameters in simulated gastric fluid (p. H 1. 2) 17
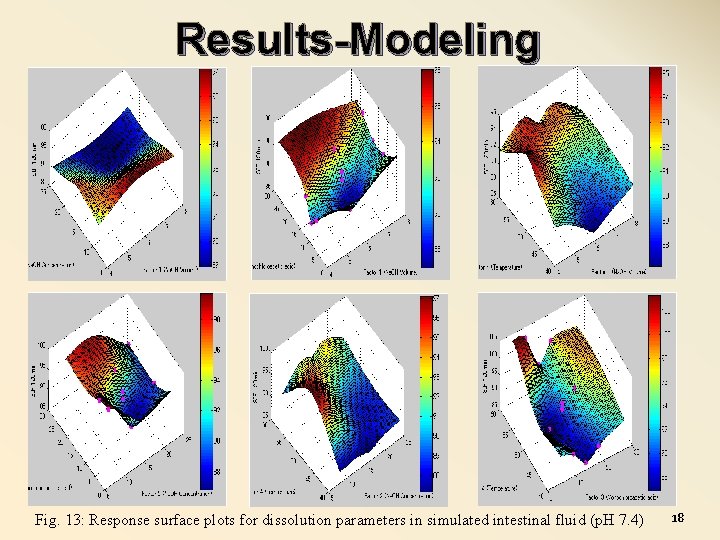
Results-Modeling Fig. 13: Response surface plots for dissolution parameters in simulated intestinal fluid (p. H 7. 4) 18
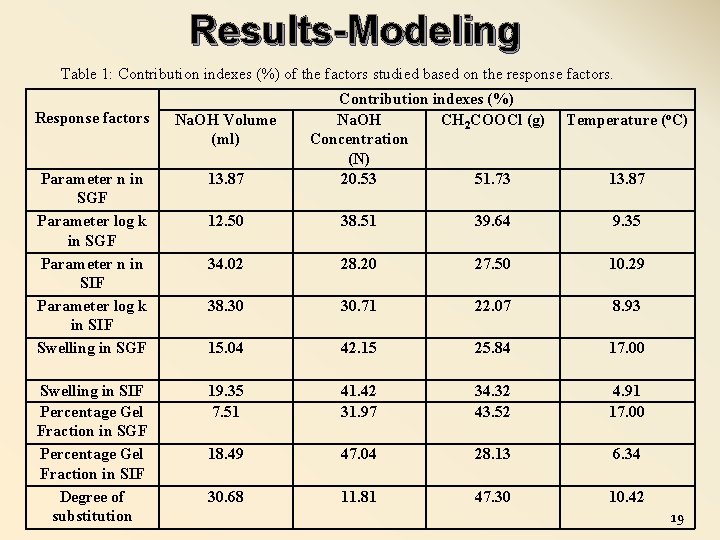
Results-Modeling Table 1: Contribution indexes (%) of the factors studied based on the response factors. Response factors Na. OH Volume (ml) Parameter n in SGF Parameter log k in SGF Parameter n in SIF Parameter log k in SIF Swelling in SGF 13. 87 Swelling in SIF Percentage Gel Fraction in SGF Percentage Gel Fraction in SIF Degree of substitution Contribution indexes (%) Na. OH CH 2 COOCl (g) Concentration (N) 20. 53 51. 73 Temperature (o. C) 13. 87 12. 50 38. 51 39. 64 9. 35 34. 02 28. 20 27. 50 10. 29 38. 30 30. 71 22. 07 8. 93 15. 04 42. 15 25. 84 17. 00 19. 35 7. 51 41. 42 31. 97 34. 32 43. 52 4. 91 17. 00 18. 49 47. 04 28. 13 6. 34 30. 68 11. 81 47. 30 10. 42 19
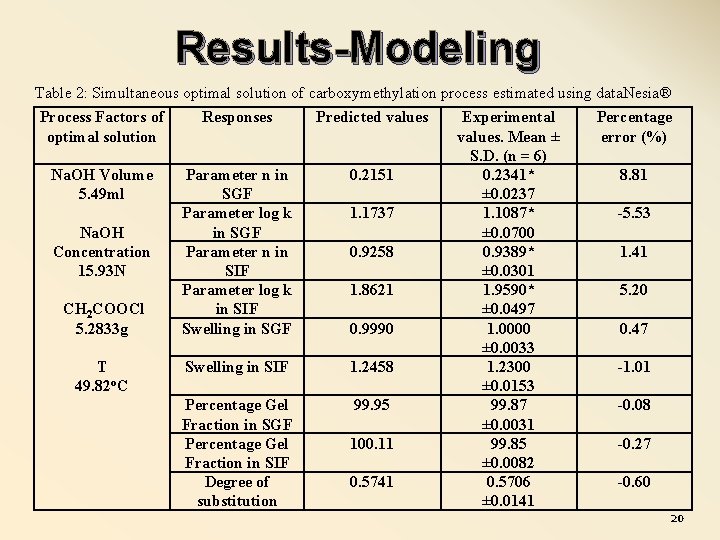
Results-Modeling Table 2: Simultaneous optimal solution of carboxymethylation process estimated using data. Nesia® Process Factors of optimal solution Responses Predicted values Na. OH Volume 5. 49 ml Parameter n in SGF Parameter log k in SGF Parameter n in SIF Parameter log k in SIF Swelling in SGF 0. 2151 Swelling in SIF 1. 2458 Percentage Gel Fraction in SGF Percentage Gel Fraction in SIF Degree of substitution 99. 95 Na. OH Concentration 15. 93 N CH 2 COOCl 5. 2833 g T 49. 82 o. C 1. 1737 0. 9258 1. 8621 0. 9990 100. 11 0. 5741 Experimental values. Mean ± S. D. (n = 6) 0. 2341* ± 0. 0237 1. 1087* ± 0. 0700 0. 9389* ± 0. 0301 1. 9590* ± 0. 0497 1. 0000 ± 0. 0033 1. 2300 ± 0. 0153 99. 87 ± 0. 0031 99. 85 ± 0. 0082 0. 5706 ± 0. 0141 Percentage error (%) 8. 81 -5. 53 1. 41 5. 20 0. 47 -1. 01 -0. 08 -0. 27 -0. 60 20
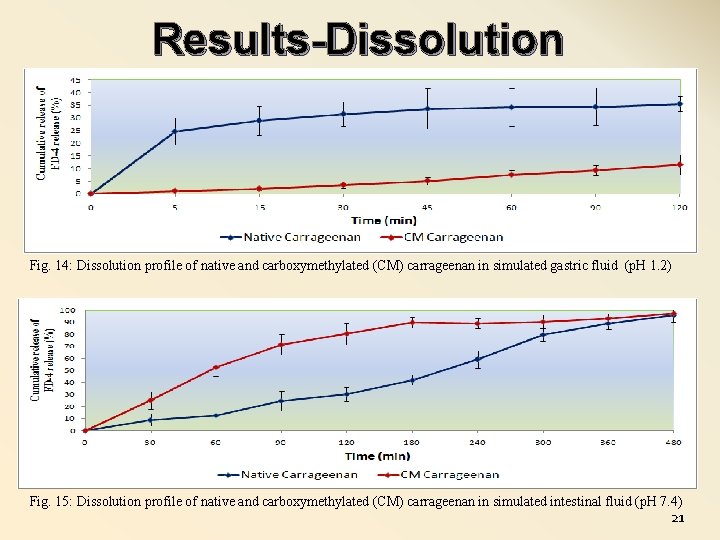
Results-Dissolution Fig. 14: Dissolution profile of native and carboxymethylated (CM) carrageenan in simulated gastric fluid (p. H 1. 2) Fig. 15: Dissolution profile of native and carboxymethylated (CM) carrageenan in simulated intestinal fluid (p. H 7. 4) 21

Part 2 Optimization of insulin encapsulation and Intestinal mucoadhesion functionalization 22
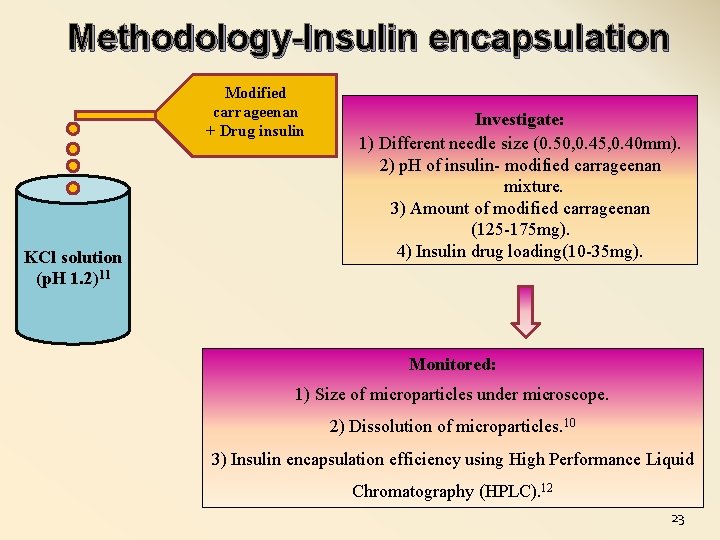
Methodology-Insulin encapsulation Modified carrageenan + Drug insulin KCl solution (p. H 1. 2)11 Investigate: 1) Different needle size (0. 50, 0. 45, 0. 40 mm). 2) p. H of insulin- modified carrageenan mixture. 3) Amount of modified carrageenan (125 -175 mg). 4) Insulin drug loading(10 -35 mg). Monitored: 1) Size of microparticles under microscope. 2) Dissolution of microparticles. 10 3) Insulin encapsulation efficiency using High Performance Liquid Chromatography (HPLC). 12 23
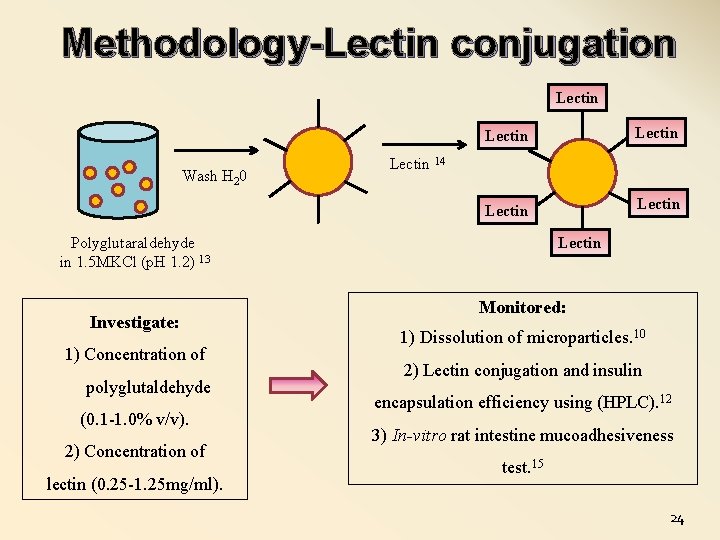
Methodology-Lectin conjugation Lectin Wash H 20 Lectin Lectin 14 Polyglutaraldehyde in 1. 5 MKCl (p. H 1. 2) 13 Investigate: 1) Concentration of polyglutaldehyde (0. 1 -1. 0% v/v). 2) Concentration of lectin (0. 25 -1. 25 mg/ml). Lectin Monitored: 1) Dissolution of microparticles. 10 2) Lectin conjugation and insulin encapsulation efficiency using (HPLC). 12 3) In-vitro rat intestine mucoadhesiveness test. 15 24
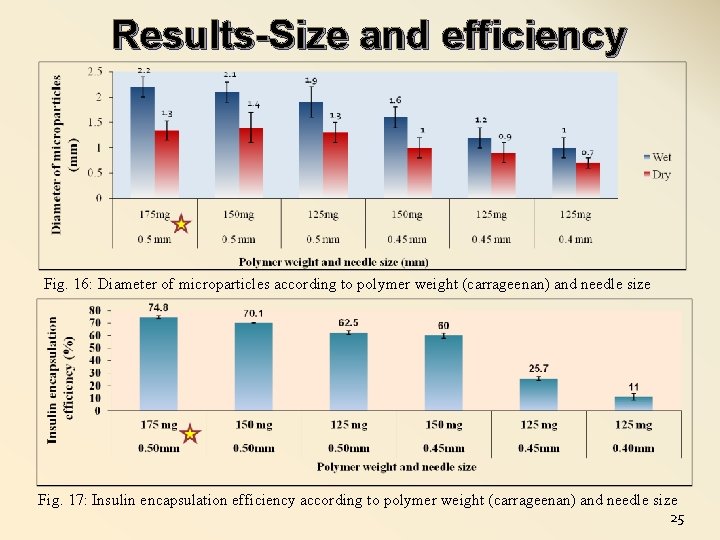
Results-Size and efficiency Fig. 16: Diameter of microparticles according to polymer weight (carrageenan) and needle size Fig. 17: Insulin encapsulation efficiency according to polymer weight (carrageenan) and needle size 25
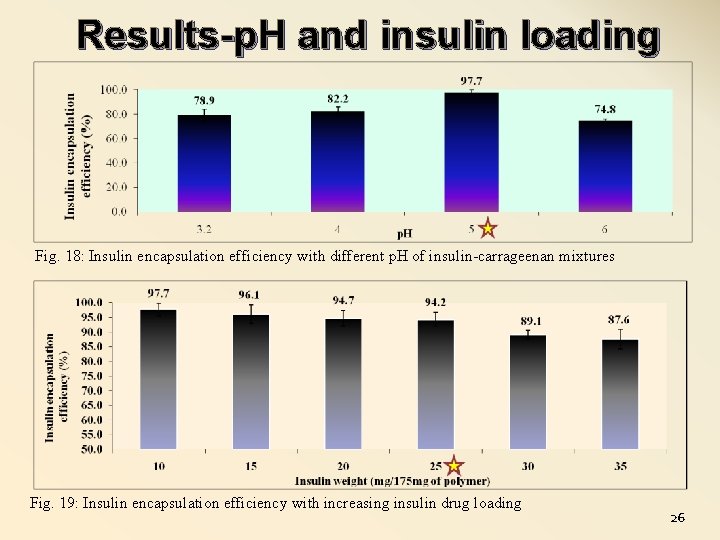
Results-p. H and insulin loading Fig. 18: Insulin encapsulation efficiency with different p. H of insulin-carrageenan mixtures Fig. 19: Insulin encapsulation efficiency with increasing insulin drug loading 26
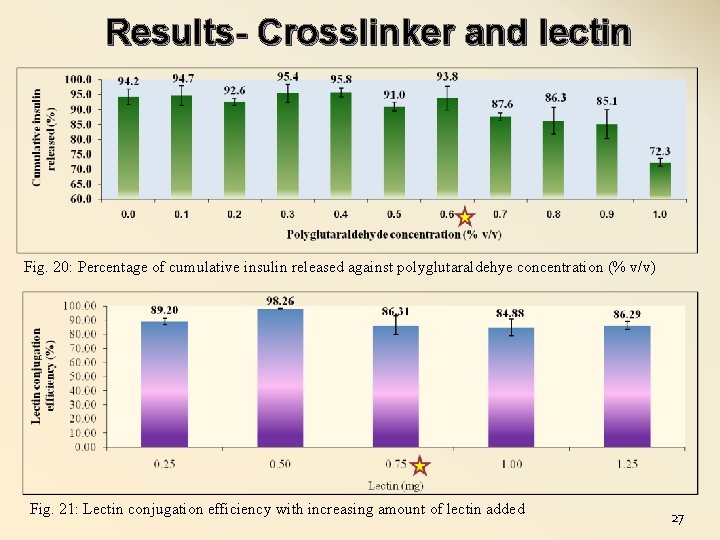
Results- Crosslinker and lectin Fig. 20: Percentage of cumulative insulin released against polyglutaraldehye concentration (% v/v) Fig. 21: Lectin conjugation efficiency with increasing amount of lectin added 27
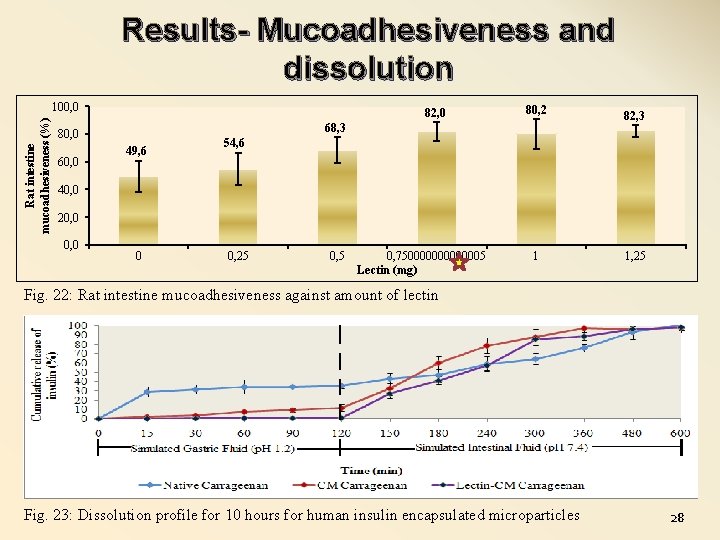
Results- Mucoadhesiveness and dissolution Rat intestine mucoadhesiveness (%) 100, 0 82, 0 68, 3 80, 0 60, 0 80, 2 49, 6 82, 3 54, 6 40, 0 20, 0 0 0, 25 0, 750000005 Lectin (mg) 1 1, 25 Fig. 22: Rat intestine mucoadhesiveness against amount of lectin Fig. 23: Dissolution profile for 10 hours for human insulin encapsulated microparticles 28

Results-SEM Images Fig. 24: Microparticle image Fig. 25: Microparticle surface image (8000 X magnification ) 1288. 65 µm 1242. 25 µm Fig. 26: Lectin surface functionalized microparticle image Fig. 27: Lectin surface functionalized image (8000 X magnification ) 29

Part 3 Cytotoxicity of the drug delivery system on human intestinal cell model 30
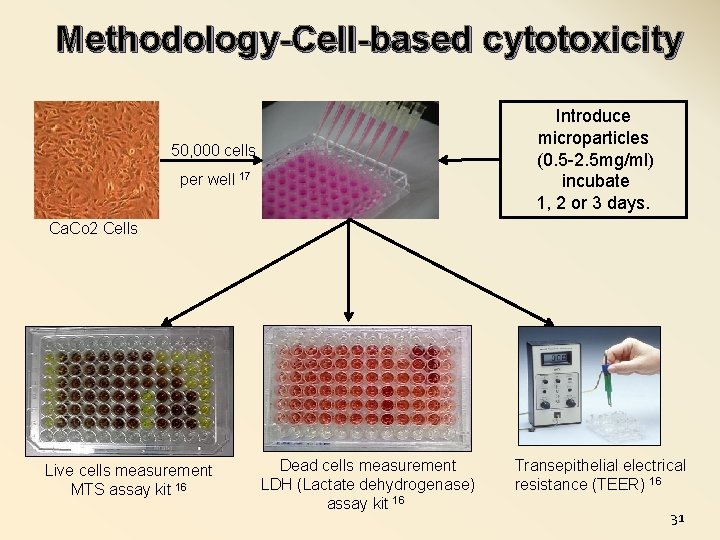
Methodology-Cell-based cytotoxicity Introduce microparticles (0. 5 -2. 5 mg/ml) incubate 1, 2 or 3 days. 50, 000 cells per well 17 Ca. Co 2 Cells Live cells measurement MTS assay kit 16 Dead cells measurement LDH (Lactate dehydrogenase) assay kit 16 Transepithelial electrical resistance (TEER) 16 31
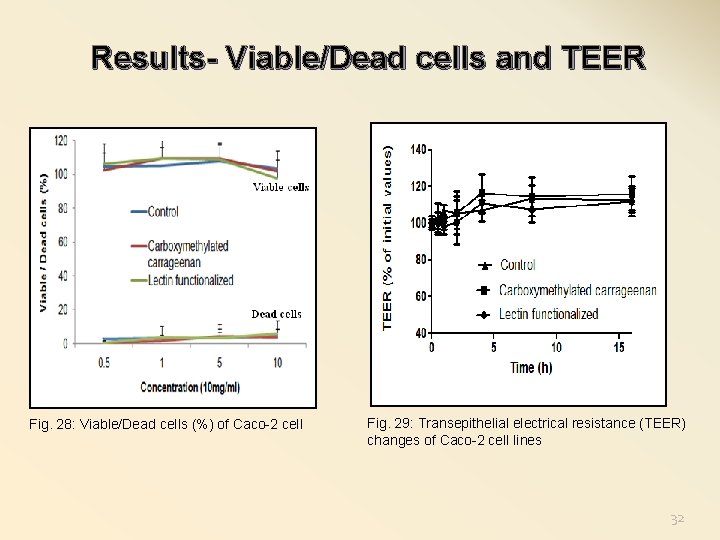
Results- Viable/Dead cells and TEER Fig. 28: Viable/Dead cells (%) of Caco-2 cell Fig. 29: Transepithelial electrical resistance (TEER) changes of Caco-2 cell lines 32

Part 4 In-vivo insulin bioavailability and blood glucose lowering effect in animal model. 33
Methodology-Rat study Intraperitoneal injection of streptozotocin 16 (45 mg/kg in citrate buffer, p. H 4. 0) Male Sprague-Dawley rats (200 -250 g) Microparticles packed in Qualicaps® Capsules 16 Type-1 diabetic rats (blood glucose ≥ 300 mg/d. L) Feeding needle Oral feeding to diabetic rats Animal Ethics Community, Faculty of Medicine, University of Malaya approved: FAR/08/12/2008/CLP(R) 34
Methodology-Rat study 20µL blood Blood glucose monitoring Accu-Check Active glucose meter (Roche, Germany) 16 100µL blood, centrifuged 13, 000 rpm and serum collected Human Insulin ELISA (Enzyme-Linked Immunosorbent Assay) Kit (Mercodia, Sweden) 16 Animal Ethics Community, Faculty of Medicine, University of Malaya approved: FAR/08/12/2008/CLP(R) 35
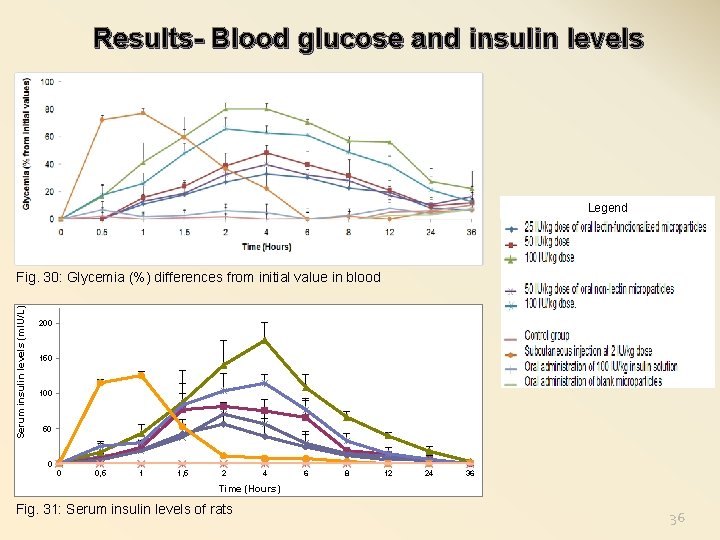
Results- Blood glucose and insulin levels Legend Serum insulin levels (m. IU/L) Fig. 30: Glycemia (%) differences from initial value in blood 200 150 100 50 0 0 0, 5 1 1, 5 2 4 6 8 12 24 36 Time (Hours) Fig. 31: Serum insulin levels of rats 36
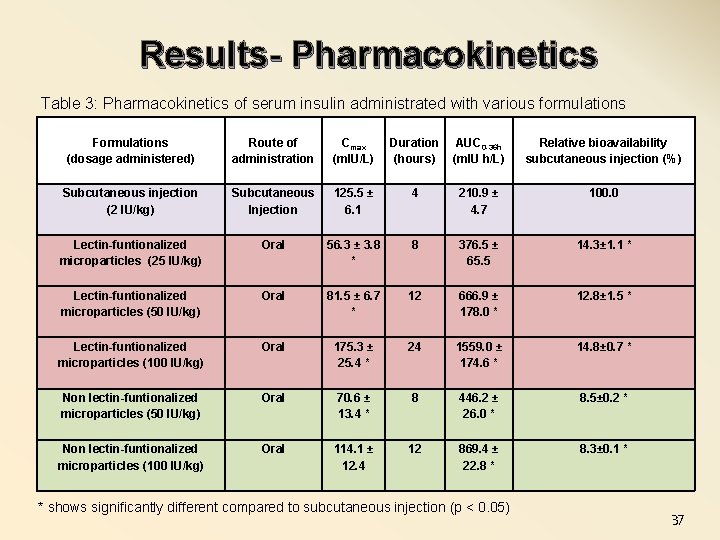
Results- Pharmacokinetics Table 3: Pharmacokinetics of serum insulin administrated with various formulations Formulations (dosage administered) Route of administration Cmax (m. IU/L) Duration (hours) AUC 0 -36 h (m. IU h/L) Relative bioavailability subcutaneous injection (%) Subcutaneous injection (2 IU/kg) Subcutaneous Injection 125. 5 ± 6. 1 4 210. 9 ± 4. 7 100. 0 Lectin-funtionalized microparticles (25 IU/kg) Oral 56. 3 ± 3. 8 * 8 376. 5 ± 65. 5 14. 3± 1. 1 * Lectin-funtionalized microparticles (50 IU/kg) Oral 81. 5 ± 6. 7 * 12 666. 9 ± 178. 0 * 12. 8± 1. 5 * Lectin-funtionalized microparticles (100 IU/kg) Oral 175. 3 ± 25. 4 * 24 1559. 0 ± 174. 6 * 14. 8± 0. 7 * Non lectin-funtionalized microparticles (50 IU/kg) Oral 70. 6 ± 13. 4 * 8 446. 2 ± 26. 0 * 8. 5± 0. 2 * Non lectin-funtionalized microparticles (100 IU/kg) Oral 114. 1 ± 12. 4 12 869. 4 ± 22. 8 * 8. 3± 0. 1 * * shows significantly different compared to subcutaneous injection (p < 0. 05) 37

Conclusion 1) Carboxymethylation of kappa-carrageenan was successfully optimized with the aid of the data. Nesia® modeling software. 2) Insulin loading improved to 13. 5% with carboxymethylated kappa-carrageenan and further surface-functionalized with lectin. 3) The microparticles were shown to be safe towards human intestinal cell cultures. 4) Oral microparticles showed prolonged 12 -24 hours glucose lowering effect and serum insulin in diabetic rat model. 38

List of presentations and published articles Oral presentation 1. Leong K. H. Modified carrageenan in encapsulating insulin as an oral drug delivery system. 5 th Yearly National Science Fellowship (NSF) research scholarship seminar. Ministry of Science and Technology (MOSTI), University Putra Malaysia, November 2008. (Best presenter award for other Science and Technology cluster). Published articles (ISI/Scopus-Cited Tier 1) 1. Leong, K. H. , Chung, L. Y. , Noordin, M. I. , Mohamad, K. , Nishikawa, M. , Onuki, Y. , Morishita, M. , Takayama, K. (2011). Carboxymethylation of kappa-carrageenan for intestinal-targeted delivery of bioactive macromolecules. Carbohydrate Polymers. 83: 1507 -1515. 2. Leong, K. H. , Chung, L. Y. , Noordin, M. I. , Onuki, Y. , Morishita, M. , Takayama, K. (2011). Lectin -functionalized carboxymethylated kappa-carrageenan microparticles for oral insulin delivery. Carbohydrate Polymers. 86: 555 -565. 39

Acknowledgement 1) 2) 3) 4) 5) Ministry of Science, Technology and Inovation, Malaysia (MOSTI); NSF scholarship. Tacara Sdn. Bhd. Tawau, Sabah University Malaya Short Term Research Grants (Vote. No: PS 129 -2007 C and FS 328/2008 C) Supervisors: Prof. Dr. Chung Lip Yong and Assoc. Prof. Dr. Mohamed Ibrahim Noordin Advisers: Prof. Dr. Kozo Takayama, Assoc. Prof. Dr. Mariko Morishita and Dr. Yoshinori Onuki, Hoshi University, Tokyo. Japan. References 1. Wild S. , Roglic G. , Green A. , Sicree R. and King H. (2004) Global prevalence of diabetes. Diabetes Care. 27(5): 1047 -1053. 2. Heller S. , Kozlovski P. and Kurtzhals P. (2007) Insulin’s 85 th anniversary –An enduring medical miracle. Diabetes Research and Clinical Practice. 78: 149 -158. 3. Khafagy E-S. ; Morishita M. , Onuki Y. and Takayama K. (2007) Current challenges in non-invasive insulin delivery systems: A comparative review. Advanced Drug Delivery Reviews. 59: 1521 -1546. 4. Amin A. , Shah T. , Patel J. and Gajjar A. (2008) Non-invasive insulin: Annual update on non-invasive insulin delivery technologies. Drug Delivery Technology. 8(3): 43 -48. 5. Bonferoni M. C. , Rossi S. , Ferrari F. , Bertoni M. , Bolhuis G. K. and Caramella C. (1998). On the employment of λ carrageenan in a matrix system. III. Optimization of a λ carrageenan-HPMC hydrophilic matrix. Journal of Controlled Release. 51: 231 -239. 6. Kooijman L. M. , Ganzeveld K. J. , Manurung R. M. and Heeres H. J. (2003) Experimental studies on the carboxymethylation of arrowroot starch in isopropanol-water media. Starch. 55: 495– 503. 7. Heinze T. H. , Pfeiffer K. and Lazik W. (2001) Starch derivatives with high degree of functionalization. III. Influence of reaction conditions and starting materials on molecular structure of carboxymethyl starch. Journal of Applied Polymer Science. 81: 2036 -44. 8. van de Velde F, Rollema H. S. (2006) High resolution NMR of carrageenans. In Webb GA. Modern Magnetic Resonance. Springer, Netherlands. 1605– 1610. 40

References 9. Onuki Y. , Nishikawa M. , Morishita M. and Takayama K. (2008) Development of photocrosslinked polyacrylic acid hydrogel as an adhesive for dermatological patched: Involvement of formulation factors in physical properties and pharmacological effects. International Journal of Pharmaceutics. 349: 47– 52. 10. Sajeesh S. and Sharma C. P. (2006) Cyclodextrin-insulin complex encapsulated polymethacrylic acid based nanoparticles for oral insulin delivery. International Journal of Pharmaceutics. 325: 147– 54. 11. Sipahigil O and Dortunc B. (2001) Preparation and in vitro evaluation of verapamil HCl and ibuprofen containing carrageenan beads. International Journal of Pharmaceutics. 228: 119 -128. 12. Deeb SE. , Preu L. and Watzig H. (2007) Evaluation of monolithic HPLC columns for various pharmaceutical separation: Method transfer from conventional phases and batch to batch repeatability. Journal of Pharmaceutical and Biomedical Analysis. 44: 85 -95. 13. Tanriseven A and Olcer Z (2008) A novel method for the immobilization of glucoanylase onto polyglutaraldehydeactivated gelatine. Biochemical Engineering Journal. 39: 430 -434. 14. Montisci MJ, Giovannuci G, Duchene D and Ponchel G. (2001) Covalent coupling of asparagus pea and tomato lectins to poly(lactide) microspheres. International Journal of Pharmaceutics. 215: 153 -161. 15. Santos CA. , Jacob JS. , Hertzog BA. , Freedman BD. , Press DL. , Harnpicharnchai P. , Mathiowitz E. (1999) Correlation of two bioadhesion assay: the everted sac technique and the CAHN microbalance. Journal of Controlled Release. 61: 113 -122. 16. Morishita M. , Goto T. , Nakamura K. , Lowman AM. , Takayama K. , Peppas NA. (2006) Novel oral insulin delivery systems based on complexation polymer hydrogels: Single and multiple administration studies in type 1 and 2 diabetic rats. Journal of Controlled Release. 110: 587 -594. 17. Vihola H. , Laukkanen A. , Valtola L. , Tenhu H. , Hirvonen J. (2005) Cytotoxicity of thermosensitive polymers poly(N-isopropylacrylamide), poly(N-vinylcaprolactam) and amphiphilically modified poly(N-vinylcaprolactam). Biomaterials. 26: 3055 -3064. 18. Luzio SD, Dunseath G. , Lockett A. , Broke-Smith TP. , New RR. and Owens DR. (2010) The glucose lowering effect of an oral insulin (Capsulin) during an isoglycaemic clamp study in persons with type 2 diabetes. Diabetes, Obesity and Metabolism. 12: 82 -87. 19. Kapitza C. , Zijlstra E. , Heinemann L. , Castelli MC. , Riley G. and Heise T. (2010) Oral insulin: A comparison with subcutaneous regular human insulin in patients with type-2 diabetes. Diabetes Care. 33(6): 1288 -1290. 41

Thank You 42
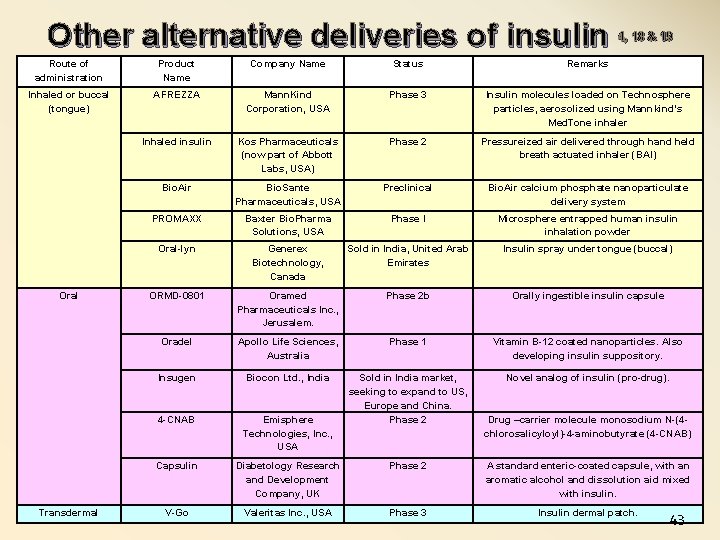
Other alternative deliveries of insulin 4, 18 & 19 Route of administration Product Name Company Name Status Remarks Inhaled or buccal (tongue) AFREZZA Mann. Kind Corporation, USA Phase 3 Insulin molecules loaded on Technosphere particles, aerosolized using Mannkind’s Med. Tone inhaler Inhaled insulin Kos Pharmaceuticals (now part of Abbott Labs, USA) Phase 2 Pressureized air delivered through hand held breath actuated inhaler (BAI) Bio. Air Bio. Sante Pharmaceuticals, USA Preclinical Bio. Air calcium phosphate nanoparticulate delivery system PROMAXX Baxter Bio. Pharma Solutions, USA Phase I Microsphere entrapped human insulin inhalation powder Oral-lyn Generex Biotechnology, Canada Sold in India, United Arab Emirates Insulin spray under tongue (buccal) ORMD-0801 Oramed Pharmaceuticals Inc. , Jerusalem. Phase 2 b Orally ingestible insulin capsule Oradel Apollo Life Sciences, Australia Phase 1 Vitamin B-12 coated nanoparticles. Also developing insulin suppository. Insugen Biocon Ltd. , India Novel analog of insulin (pro-drug). 4 -CNAB Emisphere Technologies, Inc. , USA Sold in India market, seeking to expand to US, Europe and China. Phase 2 Capsulin Diabetology Research and Development Company, UK Phase 2 A standard enteric-coated capsule, with an aromatic alcohol and dissolution aid mixed with insulin. V-Go Valeritas Inc. , USA Phase 3 Insulin dermal patch. Oral Transdermal Drug –carrier molecule monosodium N-(4 chlorosalicyloyl)-4 -aminobutyrate (4 -CNAB) 43

Types of Carrageenans Chondrus crispus - mixture of kappa and lambda. Kappaphycus alvarezii - mainly kappa. Eucheuma denticulatum - mainly iota. Gigartina skottsbergii - mainly kappa, some lambda. Sarcothalia crispata - mixture of kappa and lambda. Mc. Hugh DJ. (2003) A guide to the seaweed industry. FAO Fisheries Technical Paper. 441: 1 -105 44

Insulin ØInsulin is a peptide hormone made of two chains of amino acid sequences. ØThe A-chain has 21 amino acids and the B-chain has 30 amino acids. ØThe chains are linked together through the sulfur atoms of cysteine (Cys). ØInsulin has a molecular weight of 5808 Da. ØIt is produced in the islets of Langerhans in the pancreas. ØInsulin causes cells in the liver, muscle and fat tissue to take up glucose from the blood and storing it as gylcogen in the liver and muscle Heller S. , Kozlovski P. , Kurtzhals P. (2007) Insulin’s 85 th anniversary – An enduring medical miracle. Diabetes Research and Clinical Practice. 78: 149– 158 45
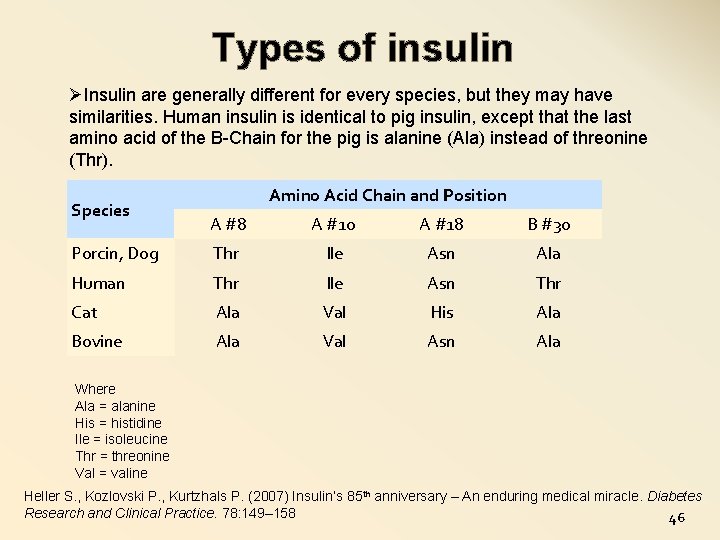
Types of insulin ØInsulin are generally different for every species, but they may have similarities. Human insulin is identical to pig insulin, except that the last amino acid of the B-Chain for the pig is alanine (Ala) instead of threonine (Thr). Species Amino Acid Chain and Position A #8 A #10 A #18 B #30 Porcin, Dog Thr Ile Asn Ala Human Thr Ile Asn Thr Cat Ala Val His Ala Bovine Ala Val Asn Ala Where Ala = alanine His = histidine Ile = isoleucine Thr = threonine Val = valine Heller S. , Kozlovski P. , Kurtzhals P. (2007) Insulin’s 85 th anniversary – An enduring medical miracle. Diabetes Research and Clinical Practice. 78: 149– 158 46
- Slides: 46