Carboxylic Acids Reactions Acidbase reactions LDF ion Hbonding
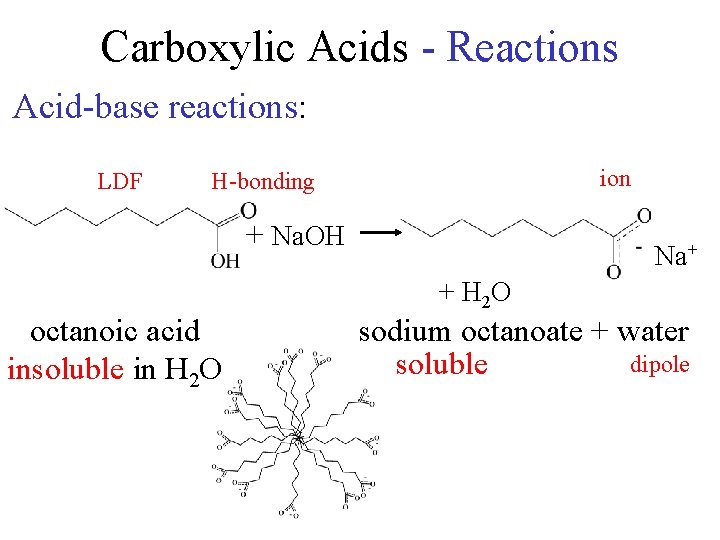
Carboxylic Acids - Reactions Acid-base reactions: LDF ion H-bonding + Na. OH Na+ + H 2 O octanoic acid insoluble in H 2 O sodium octanoate + water dipole soluble
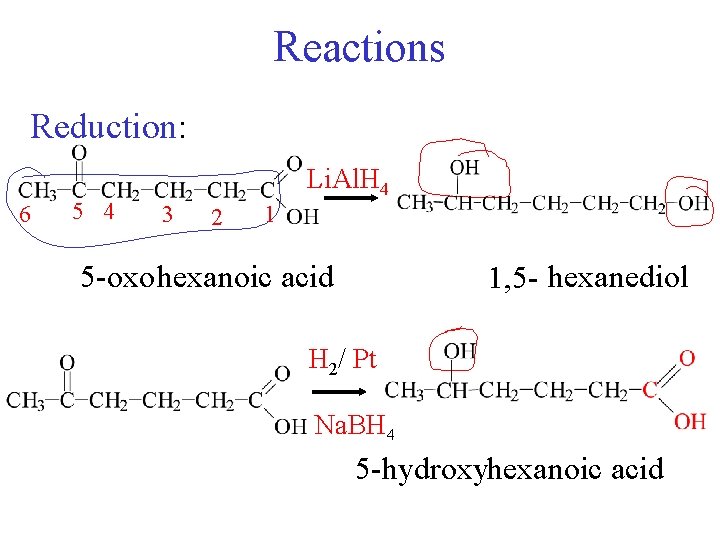
Reactions Reduction: 6 5 4 Li. Al. H 4 3 2 1 5 -oxo hexanoic acid 1, 5 - hexanediol H 2/ Pt Na. BH 4 5 -hydroxyhexanoic acid
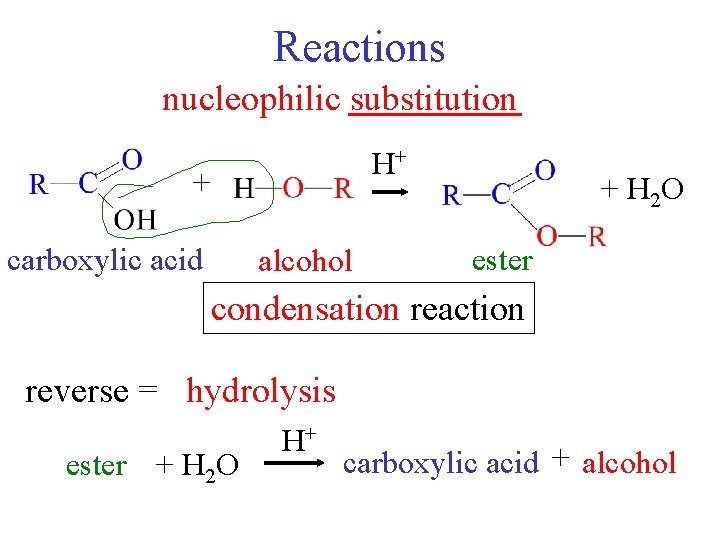
Reactions nucleophilic substitution H+ + carboxylic acid alcohol + H 2 O ester condensation reaction reverse = hydrolysis ester + H 2 O H+ carboxylic acid + alcohol
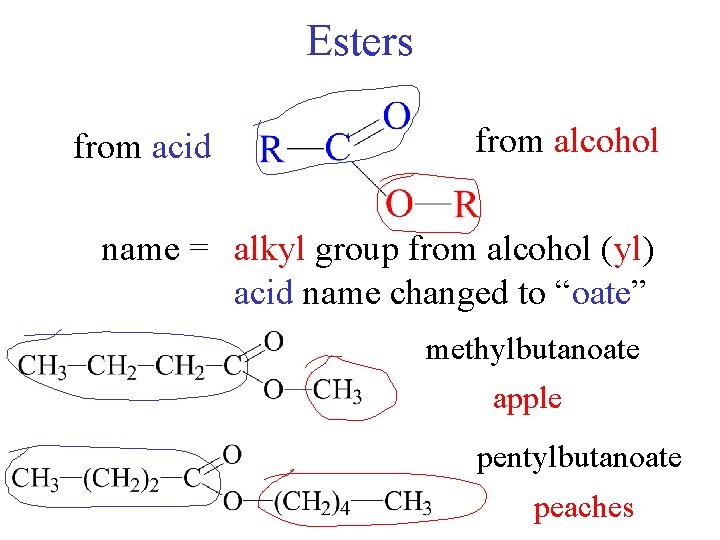
Esters from acid from alcohol name = alkyl group from alcohol (yl) acid name changed to “oate” methylbutanoate apple pentyl butanoate peaches
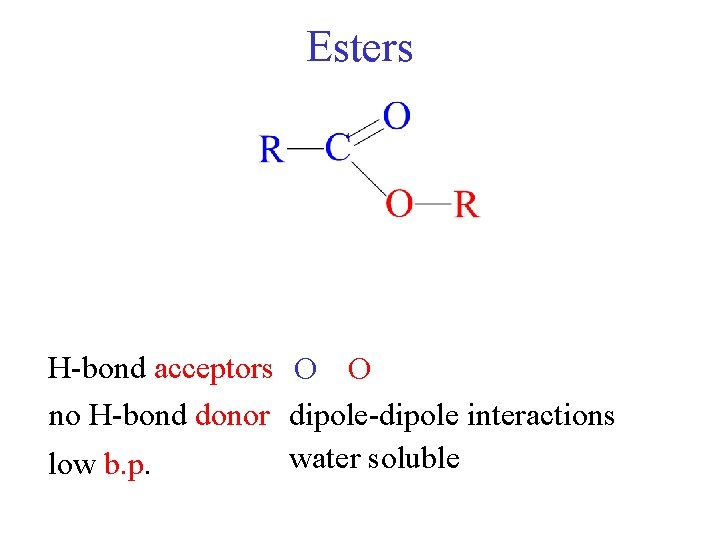
Esters H-bond acceptors O O no H-bond donor dipole-dipole interactions water soluble low b. p.
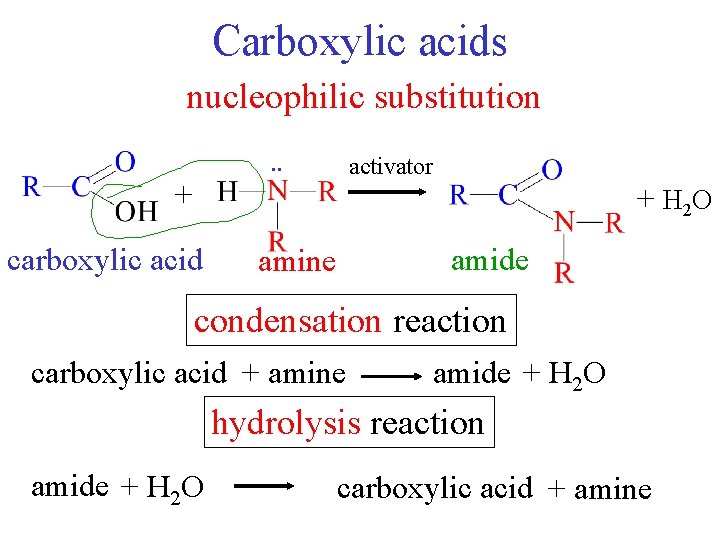
Carboxylic acids nucleophilic substitution. . activator + + H 2 O carboxylic acid amide amine condensation reaction carboxylic acid + amine amide + H 2 O hydrolysis reaction amide + H 2 O carboxylic acid + amine
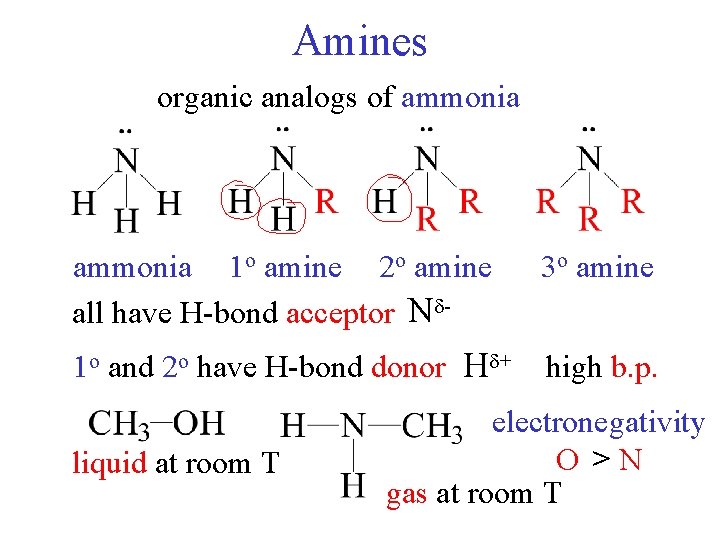
Amines organic analogs of ammonia 1 o amine 2 o amine all have H-bond acceptor N - 3 o amine 1 o and 2 o have H-bond donor H + high b. p. liquid at room T electronegativity O >N gas at room T
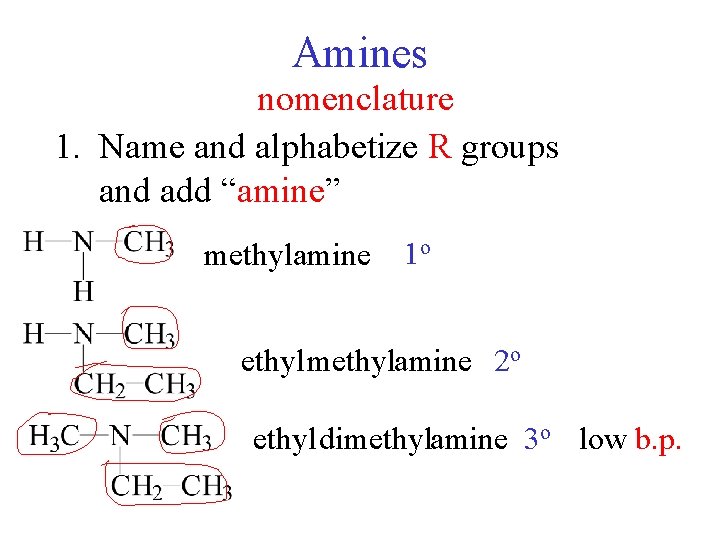
Amines nomenclature 1. Name and alphabetize R groups and add “amine” methylamine 1 o ethyl methylamine 2 o ethyl dimethylamine 3 o low b. p.
![Amines weak bases + H 2 O +][OH-] [R-NH Kb = 3 [R-NH 2] Amines weak bases + H 2 O +][OH-] [R-NH Kb = 3 [R-NH 2]](http://slidetodoc.com/presentation_image_h/9db1fdf3aad4ebaa6f3987ef895df8f6/image-9.jpg)
Amines weak bases + H 2 O +][OH-] [R-NH Kb = 3 [R-NH 2] + OH- smaller Kb weaker base
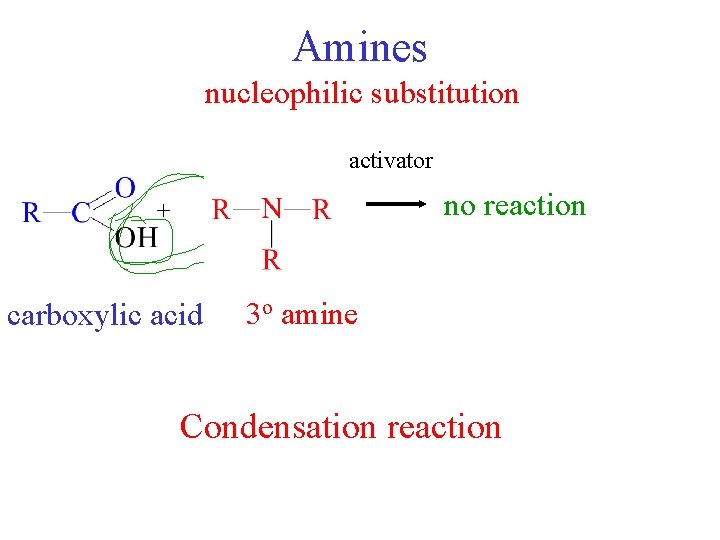
Amines nucleophilic substitution activator + carboxylic acid amine 123 oo amine +H O 2 O + H 2 no reaction amide Condensation reaction
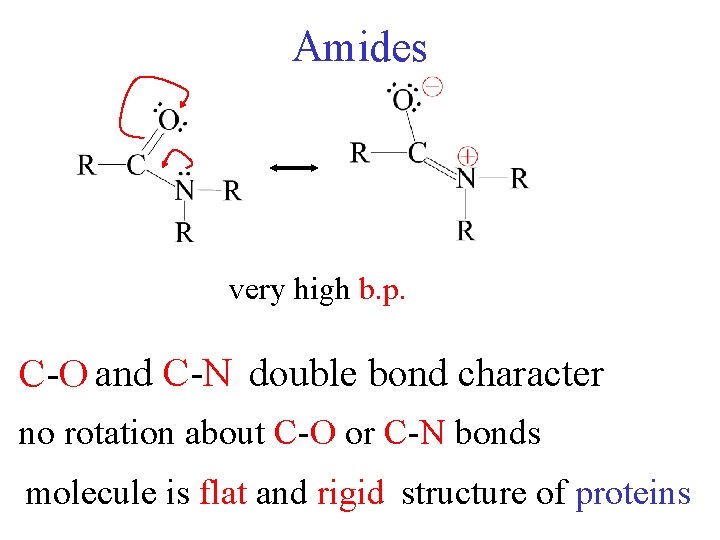
Amides very high b. p. C-O and C-N double bond character no rotation about C-O or C-N bonds molecule is flat and rigid structure of proteins
- Slides: 11