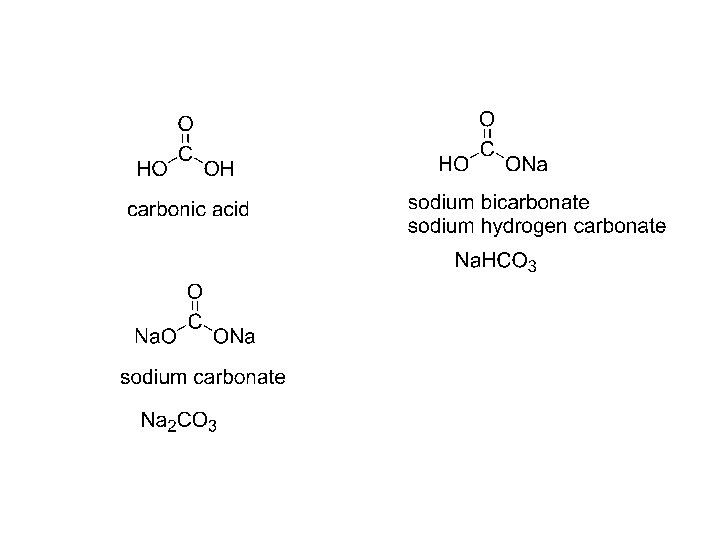
Carboxylic acids RCOOH RCO 2 H Common names
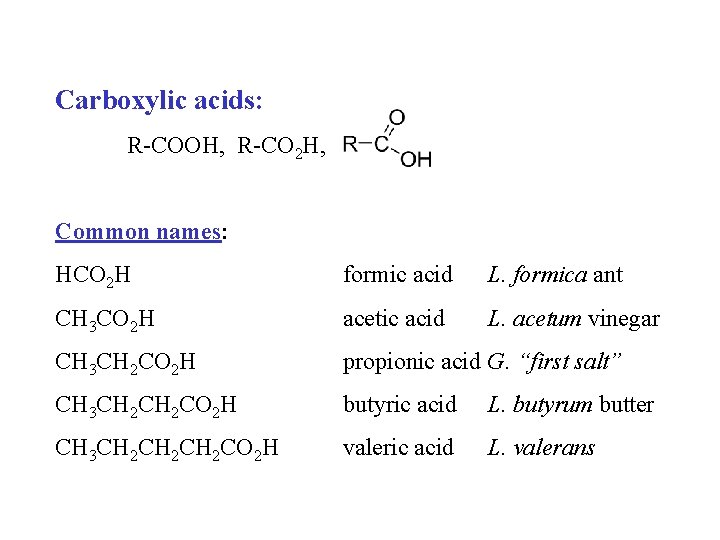
Carboxylic acids: R-COOH, R-CO 2 H, Common names: HCO 2 H formic acid L. formica ant CH 3 CO 2 H acetic acid L. acetum vinegar CH 3 CH 2 CO 2 H propionic acid G. “first salt” CH 3 CH 2 CO 2 H butyric acid L. butyrum butter CH 3 CH 2 CH 2 CO 2 H valeric acid L. valerans
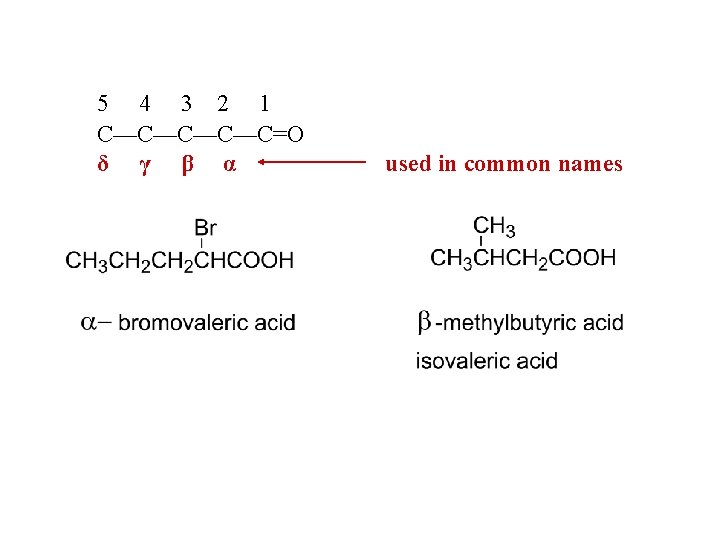
5 4 3 2 1 C—C—C=O δ γ β α used in common names
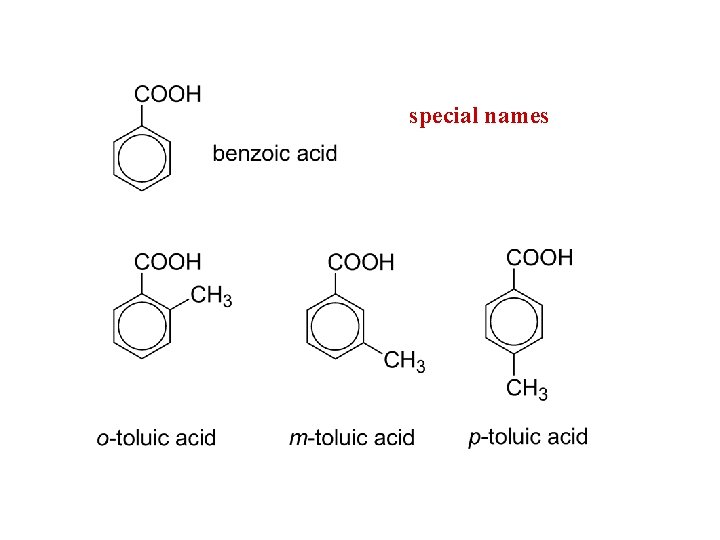
special names

IUPAC nomenclature for carboxylic acids: parent chain = longest, continuous carbon chain that contains the carboxyl group alkane, drop –e, add –oic acid HCOOH methanoic acid CH 3 CO 2 H ethanoic acid CH 3 CH 2 CO 2 H propanoic acid CH 3 CHCOOH 2 -methylpropanoic acid Br CH 3 CH 2 CHCO 2 H 2 -bromobutanoic acid

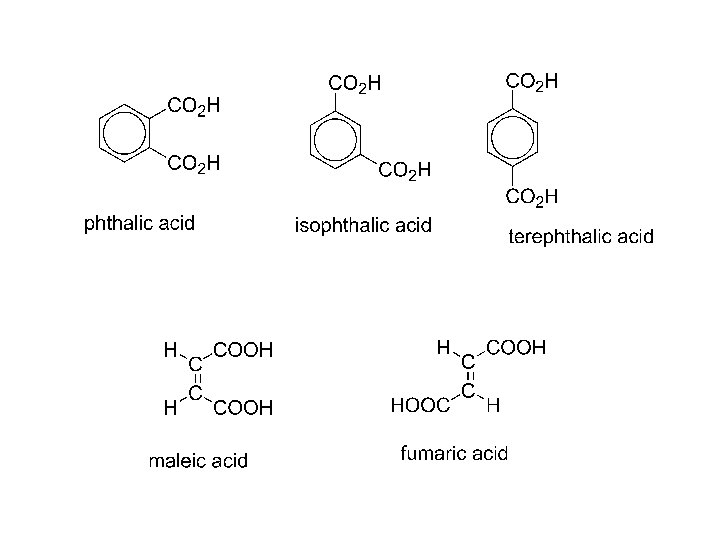
dicarboxylic acids: HOOC-COOH oxalic acid HO 2 C-CH 2 -CO 2 H malonic acid succinic acid HO 2 C-CH 2 CH 2 -CO 2 H glutaric acid HOOC-(CH 2)4 -COOH adipic acid HOOC-(CH 2)5 -COOH pimelic acid Oh, my! Such good apple pie!

salts of carboxylic acids: name of cation + name of acid: drop –ic acid, add –ate CH 3 CO 2 Na sodium acetate CH 3 CH 2 CO 2 NH 4 or sodium ethanoate ammonium butyrate ammonium butanoate (CH 3 CH 2 COO)2 Mg magnesium propionate magnesium propanoate

physical properties: polar + hydrogen bond water insoluble exceptions: four carbons or less acidic turn blue litmus red soluble in 5% Na. OH RCO 2 H + Na. OH RCO 2 -Na+ + H 2 O stronger acid stronger base weaker acid

RCO 2 H RCO 2 - covalent water insoluble ionic water soluble Carboxylic acids are insoluble in water, but soluble in 5% Na. OH. 1. Identification. 2. Separation of carboxylic acids from basic/neutral organic compounds. The carboxylic acid can be extracted with aq. Na. OH and then regenerated by the addition of strong acid.

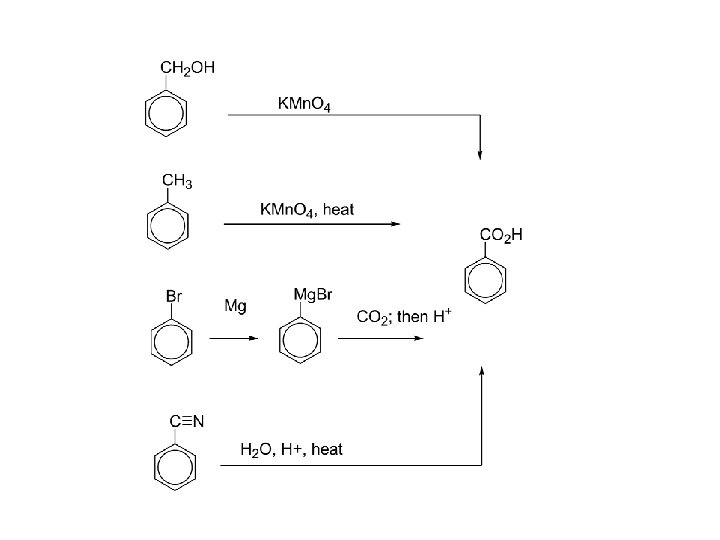
Carboxylic acids, syntheses: 1. oxidation of primary alcohols RCH 2 OH + K 2 Cr 2 O 7 RCOOH 2. oxidation of arenes Ar. R + KMn. O 4, heat Ar. COOH 3. carbonation of Grignard reagents RMg. X + CO 2 RCO 2 Mg. X + H+ RCOOH 4. hydrolysis of nitriles RCN + H 2 O, H+, heat RCOOH
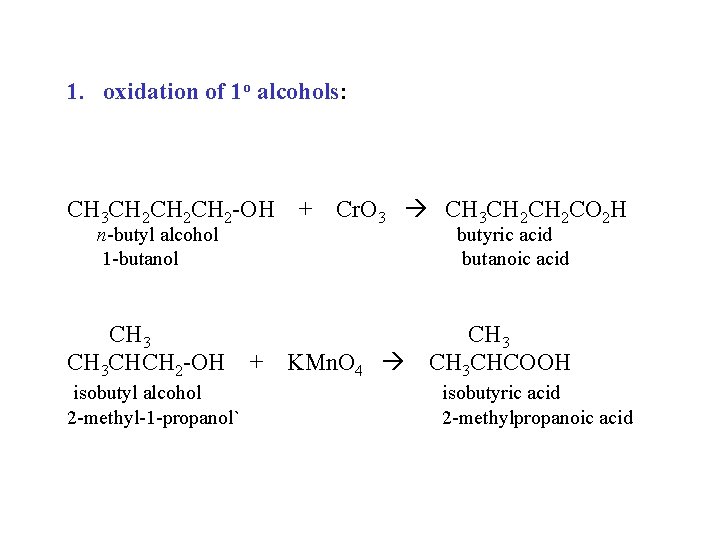
1. oxidation of 1 o alcohols: CH 3 CH 2 CH 2 -OH n-butyl alcohol 1 -butanol CH 3 CHCH 2 -OH isobutyl alcohol 2 -methyl-1 -propanol` + + Cr. O 3 CH 3 CH 2 CO 2 H butyric acid butanoic acid KMn. O 4 CH 3 CHCOOH isobutyric acid 2 -methylpropanoic acid
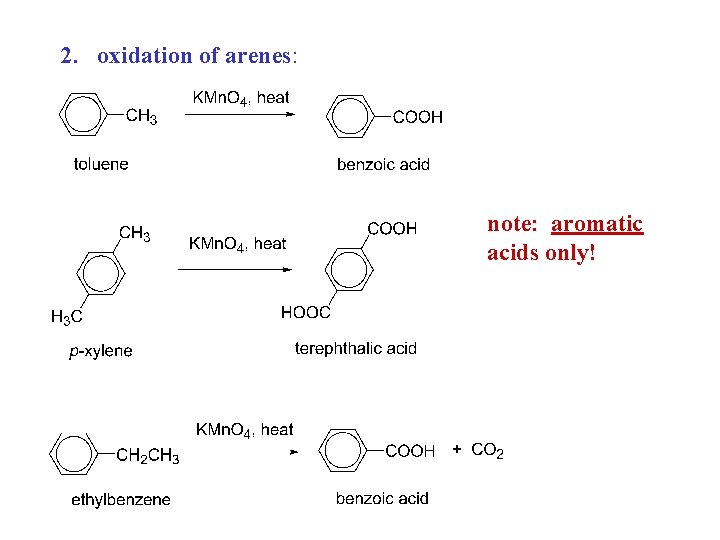
2. oxidation of arenes: note: aromatic acids only!
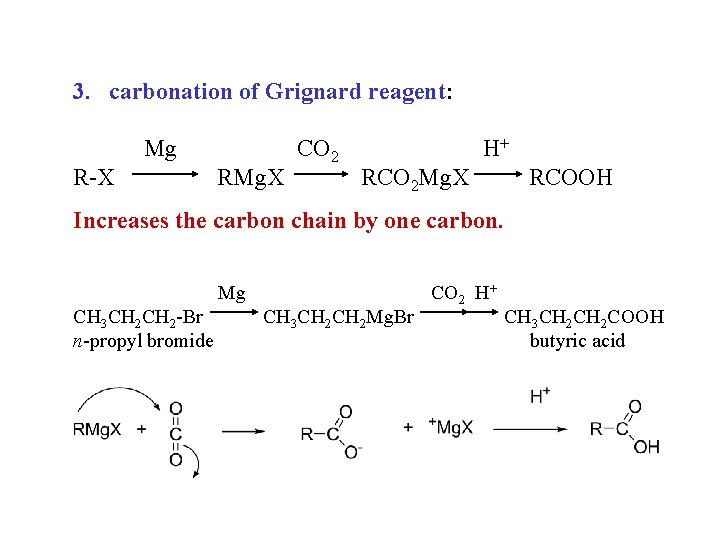
3. carbonation of Grignard reagent: Mg R-X RMg. X CO 2 H+ RCO 2 Mg. X RCOOH Increases the carbon chain by one carbon. Mg CH 3 CH 2 -Br n-propyl bromide CH 3 CH 2 Mg. Br CO 2 H+ CH 3 CH 2 COOH butyric acid
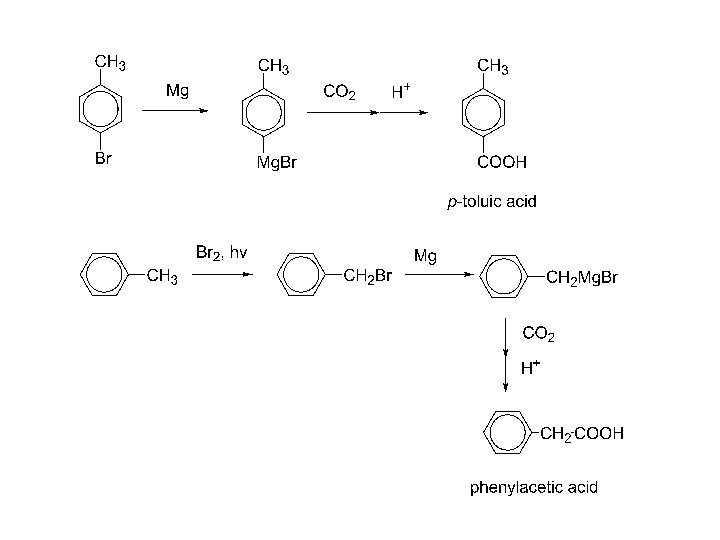

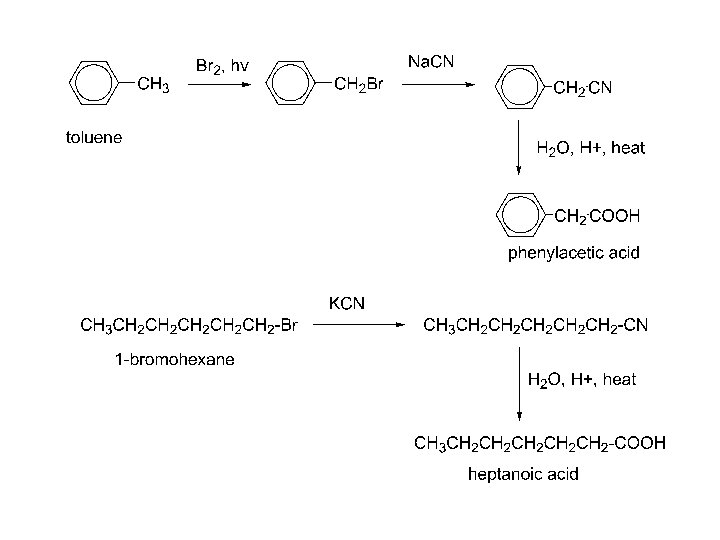
4. Hydrolysis of a nitrile: R-C N H 2 O, H+ heat R-C N H 2 O, OHheat R-CO 2 H R-CO 2 - + H+ R-CO 2 H R-X + Na. CN R-CN + H+, H 2 O, heat RCOOH 1 o alkyl halide Adds one more carbon to the chain. R-X must be 1 o or CH 3!

carboxylic acids, reactions: 1. as acids 2. conversion into functional derivatives a) acid chlorides b) esters c) amides 3. reduction 4. alpha-halogenation 5. EAS

as acids: a) with active metals RCO 2 H + Na RCO 2 -Na+ + H 2(g) b) with bases RCO 2 H + Na. OH RCO 2 -Na+ + H 2 O c) relative acid strength? CH 4 < NH 3 < HC CH < ROH < H 2 CO 3 < RCO 2 H < HF d) quantitative HA + H 2 O H 3 O+ + AKa = [H 3 O+] [A-] / [HA] ionization in water

Ka for carboxylic acids 10 -5 Why are carboxylic acids more acidic than alcohols? ROH + H 2 O H 3 O+ + RORCOOH + H 2 O H 3 O+ + RCOOΔGo = -2. 303 R T log Keq The position of the equilibrium is determined by the free energy change, ΔGo = ΔH - TΔS ΔGo ΔH Ka is inversely related to ΔH, the potential energy difference between the acid and its conjugate base. The smaller the ΔH, the larger the Ka and the stronger the acid.
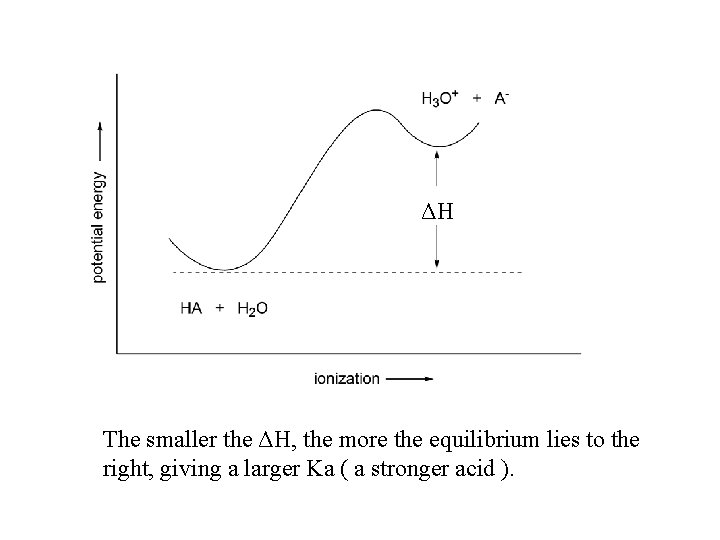
ΔH The smaller the ΔH, the more the equilibrium lies to the right, giving a larger Ka ( a stronger acid ).
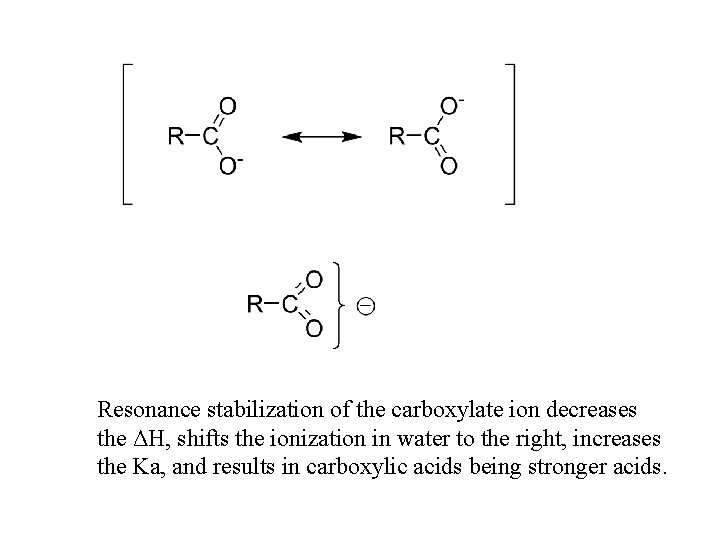
Resonance stabilization of the carboxylate ion decreases the ΔH, shifts the ionization in water to the right, increases the Ka, and results in carboxylic acids being stronger acids.

Effect of substituent groups on acid strength? CH 3 COOH 1. 75 x 10 -5 Cl. CH 2 COOH 136 x 10 -5 Cl 2 CHCOOH 5, 530 x 10 -5 Cl 3 CCOOH 23, 200 x 10 -5 -Cl is electron withdrawing and delocalizes the negative charge on the carboxylate ion, lowering the PE, decreasing the ΔH, shifting the ionization to the right and increasing acid strength.

Effect of substituent groups on acid strength of benzoic acids? Electron withdrawing groups will stabilize the anion, decrease the ΔH, shift the ionization to the right, increasing the Ka, increasing acid strength. Electron donating groups will destabilize the anion, increase the ΔH, shift the ionization in water to the left, decreasing the Ka, decreasing acid strength.

-NH 2, -NHR, -NR 2 -OH -OR -NHCOCH 3 -C 6 H 5 -R -H -X -CHO, -COR -SO 3 H -COOH, -COOR -CN -NR 3+ -NO 2 electron donating electron withdrawing

Relative acid strength? Ka p-aminobenzoic acid 1. 4 x 10 -5 p-hydroxybenzoic acid 2. 6 x 10 -5 p-methoxybenzoic acid 3. 3 x 10 -5 p-toluic acid 4. 2 x 10 -5 benzoic acid 6. 3 x 10 -5 p-chlorobenzoic acid 10. 3 x 10 -5 p-nitrobenzoic acid 36 x 10 -5
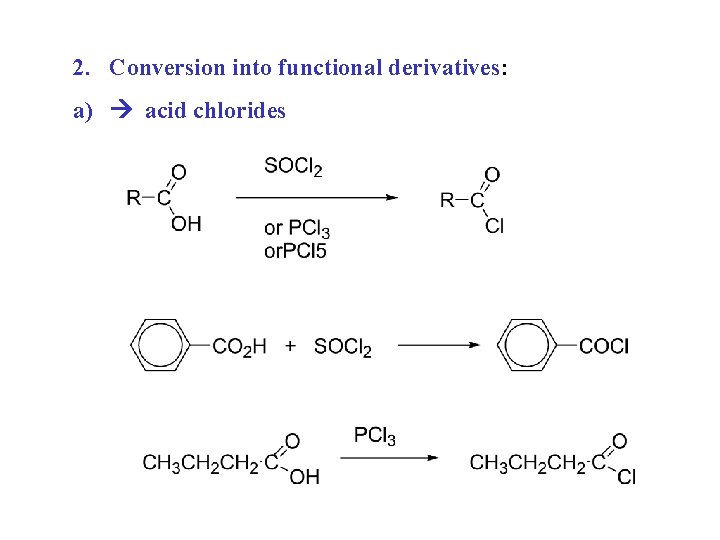
2. Conversion into functional derivatives: a) acid chlorides

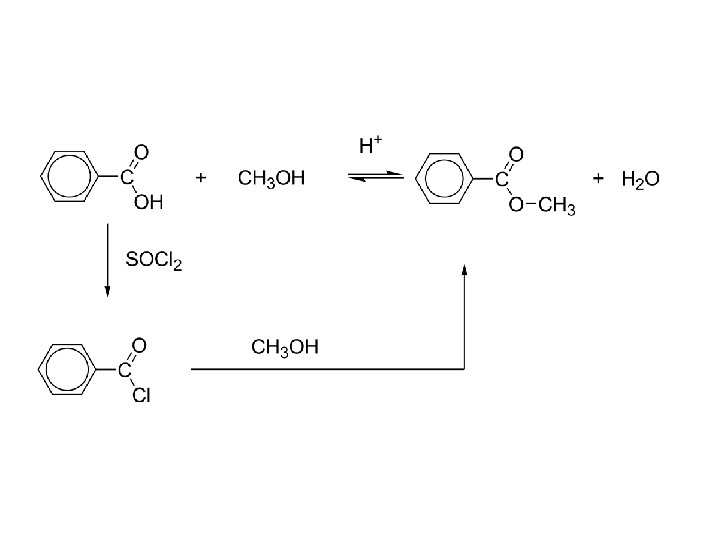
b) esters “direct” esterification: H+ RCOOH + R´OH RCO 2 R´ + H 2 O -reversible and often does not favor the ester -use an excess of the alcohol or acid to shift equilibrium -or remove the products to shift equilibrium to completion “indirect” esterification: RCOOH + PCl 3 RCOCl + R´OH RCO 2 R´ -convert the acid into the acid chloride first; not reversible
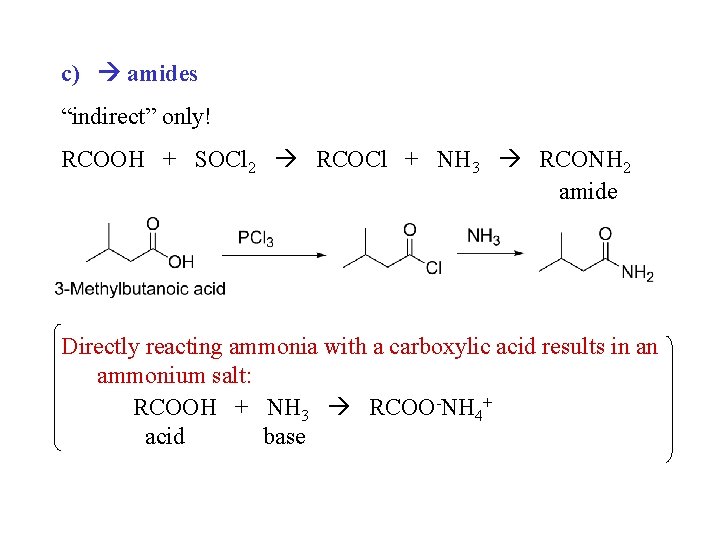
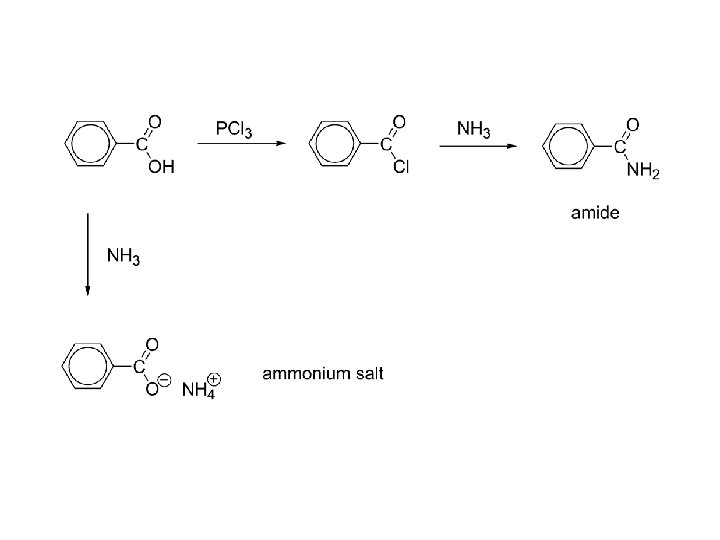
c) amides “indirect” only! RCOOH + SOCl 2 RCOCl + NH 3 RCONH 2 amide Directly reacting ammonia with a carboxylic acid results in an ammonium salt: RCOOH + NH 3 RCOO-NH 4+ acid base
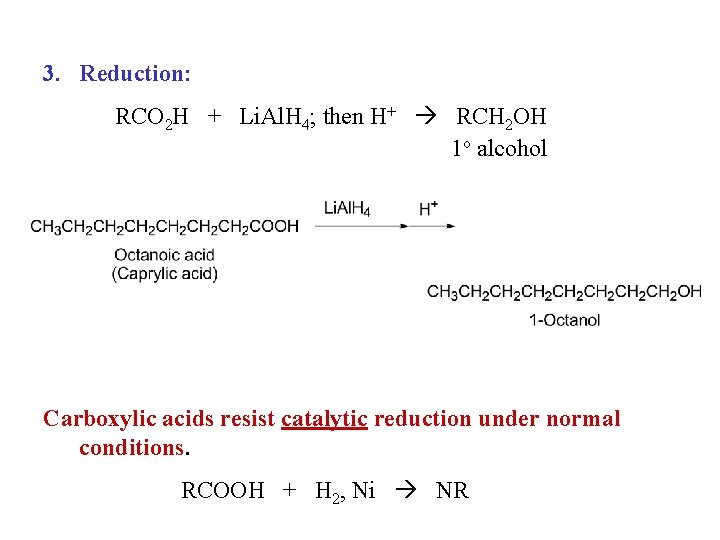
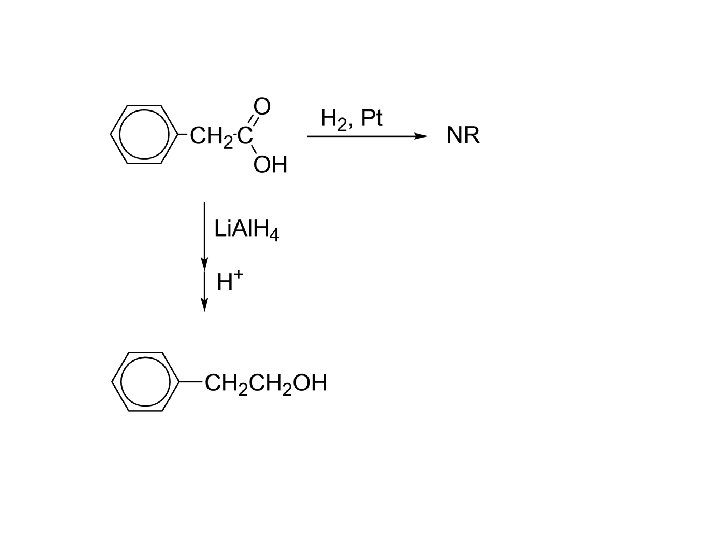
3. Reduction: RCO 2 H + Li. Al. H 4; then H+ RCH 2 OH 1 o alcohol Carboxylic acids resist catalytic reduction under normal conditions. RCOOH + H 2, Ni NR
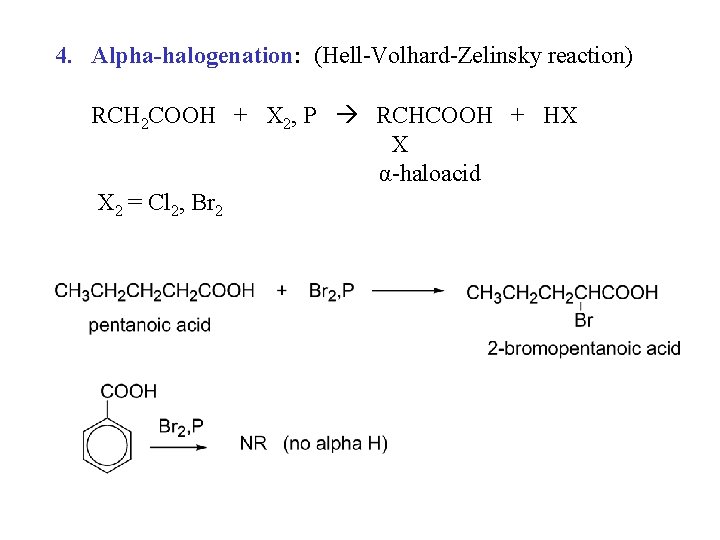
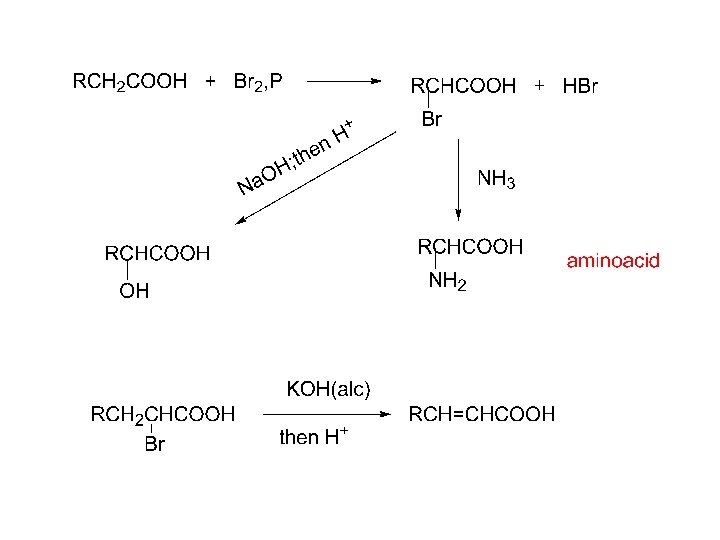
4. Alpha-halogenation: (Hell-Volhard-Zelinsky reaction) RCH 2 COOH + X 2, P RCHCOOH + HX X α-haloacid X 2 = Cl 2, Br 2
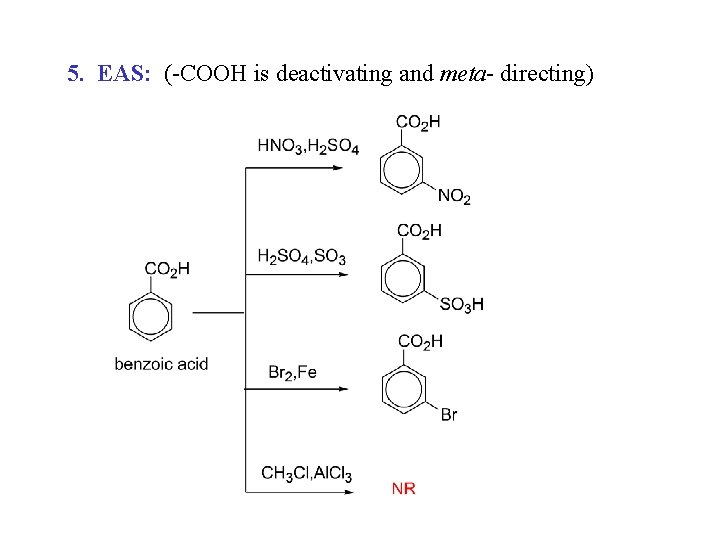
5. EAS: (-COOH is deactivating and meta- directing)

Carboxylic acids, syntheses: 1. oxidation of primary alcohols RCH 2 OH + K 2 Cr 2 O 7 RCOOH 2. oxidation of arenes Ar. R + KMn. O 4, heat Ar. COOH 3. carbonation of Grignard reagents RMg. X + CO 2 RCO 2 Mg. X + H+ RCOOH 4. hydrolysis of nitriles RCN + H 2 O, H+, heat RCOOH

carboxylic acids, reactions: 1. as acids 2. conversion into functional derivatives a) acid chlorides b) esters c) amides 3. reduction 4. alpha-halogenation 5. EAS
- Slides: 39