Carboxylic acids Nucleofilic acyl substitution reaction Dr AKM

Carboxylic acids Nucleofilic acyl substitution reaction Dr AKM Shafiqul Islam School of Bioprocess Engineering
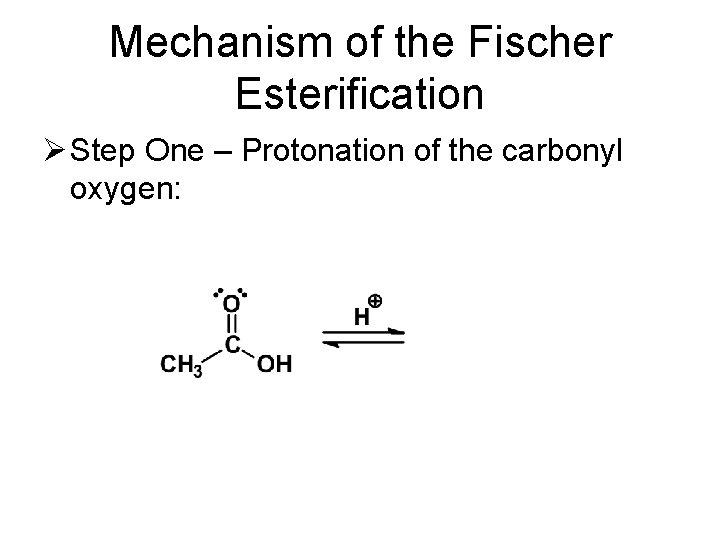
Mechanism of the Fischer Esterification Ø Step One – Protonation of the carbonyl oxygen:
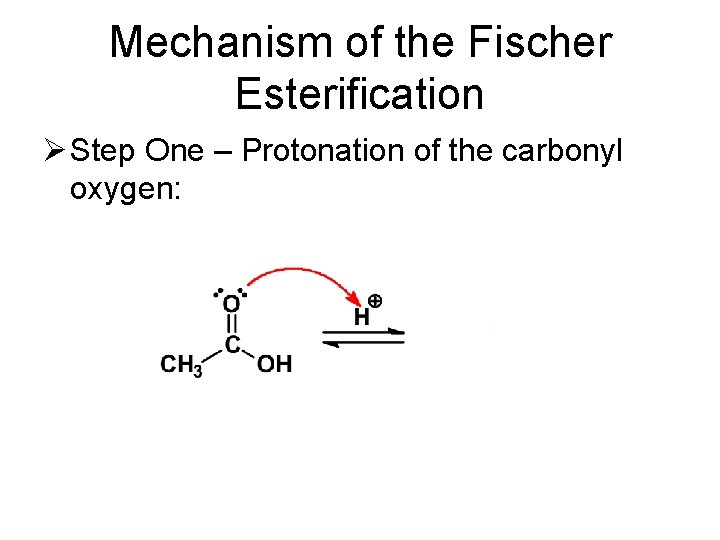
Mechanism of the Fischer Esterification Ø Step One – Protonation of the carbonyl oxygen:
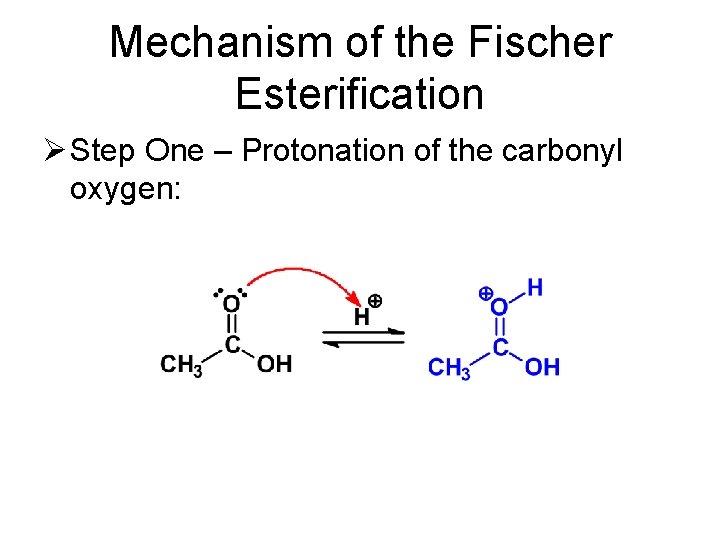
Mechanism of the Fischer Esterification Ø Step One – Protonation of the carbonyl oxygen:
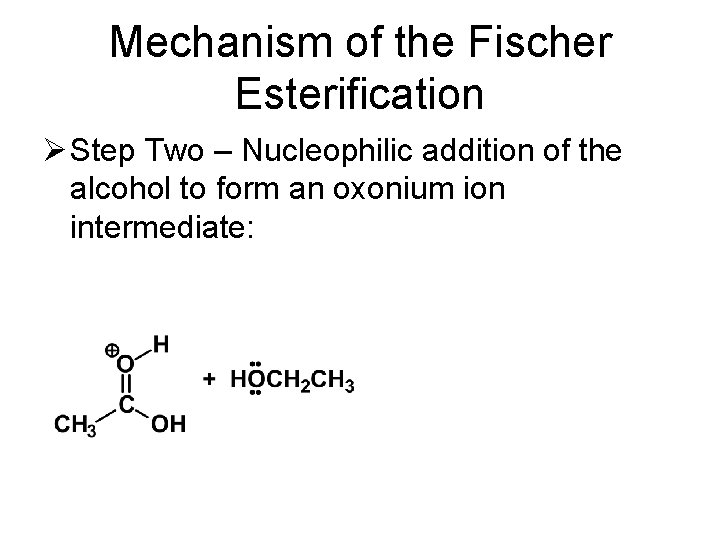
Mechanism of the Fischer Esterification Ø Step Two – Nucleophilic addition of the alcohol to form an oxonium ion intermediate:

Mechanism of the Fischer Esterification Ø Step Two – Nucleophilic addition of the alcohol to form an oxonium ion intermediate:
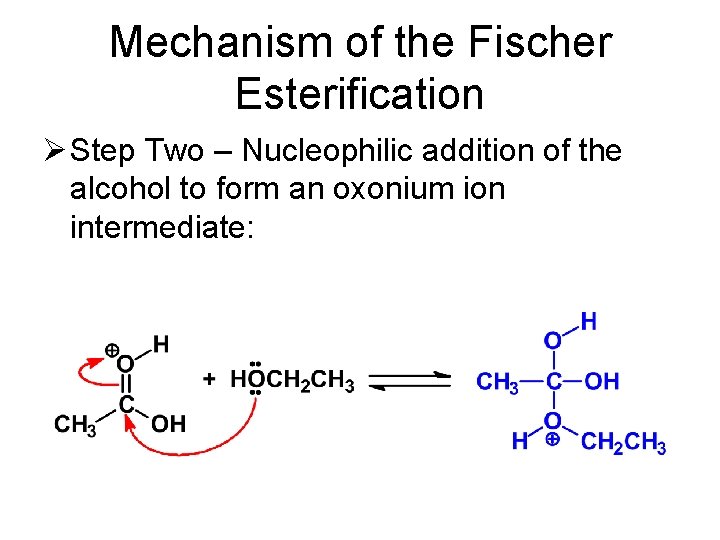
Mechanism of the Fischer Esterification Ø Step Two – Nucleophilic addition of the alcohol to form an oxonium ion intermediate:
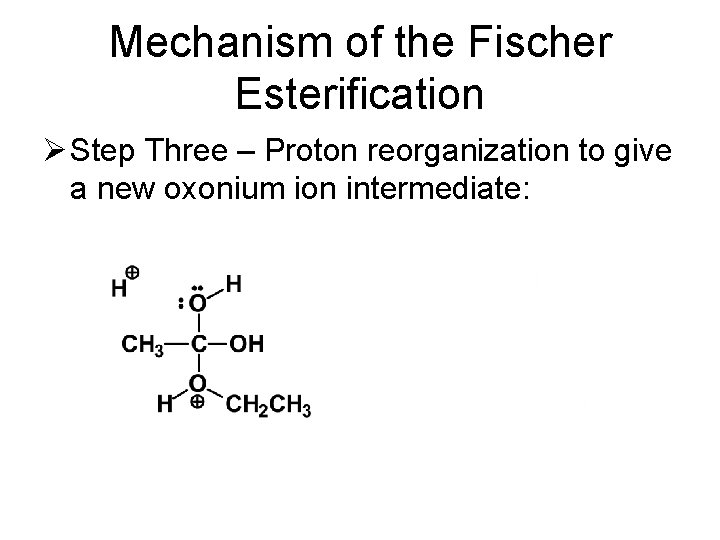
Mechanism of the Fischer Esterification Ø Step Three – Proton reorganization to give a new oxonium ion intermediate:
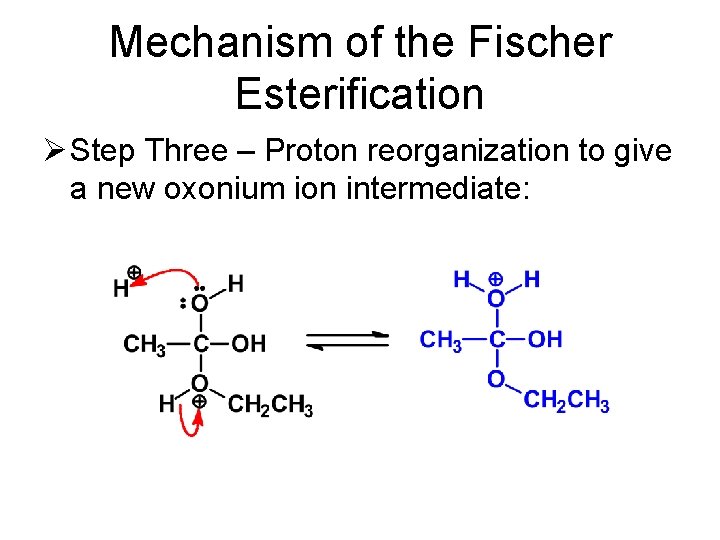
Mechanism of the Fischer Esterification Ø Step Three – Proton reorganization to give a new oxonium ion intermediate:
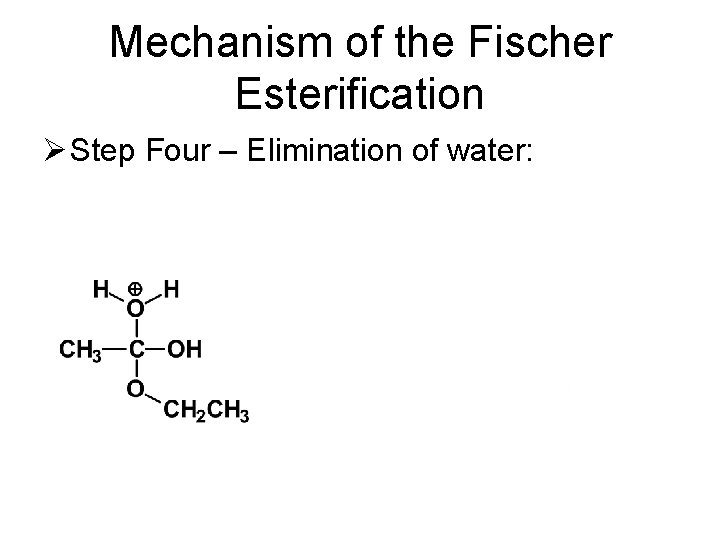
Mechanism of the Fischer Esterification Ø Step Four – Elimination of water:
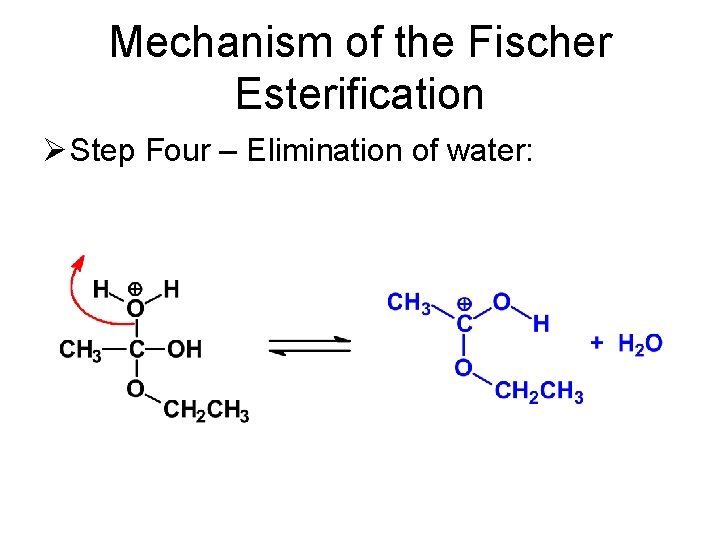
Mechanism of the Fischer Esterification Ø Step Four – Elimination of water:

Mechanism of the Fischer Esterification Ø Step Five – Deprotonation to give the ester:

Mechanism of the Fischer Esterification Ø Step Five – Deprotonation to give the ester:
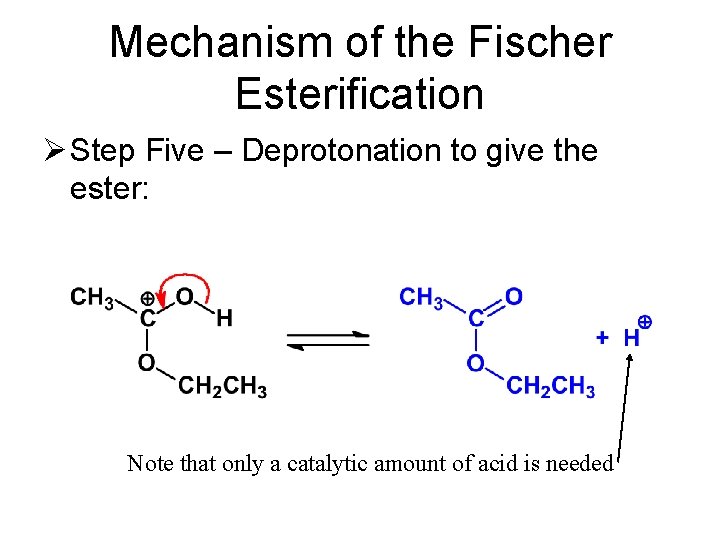
Mechanism of the Fischer Esterification Ø Step Five – Deprotonation to give the ester: Note that only a catalytic amount of acid is needed

Preparation of Esters Ø Reaction of an acid chloride with an alcohol • An alcohol reacts with an acid chloride with elimination of HCl. • Sometimes an amine base is added to precipitate the HCl that is formed.
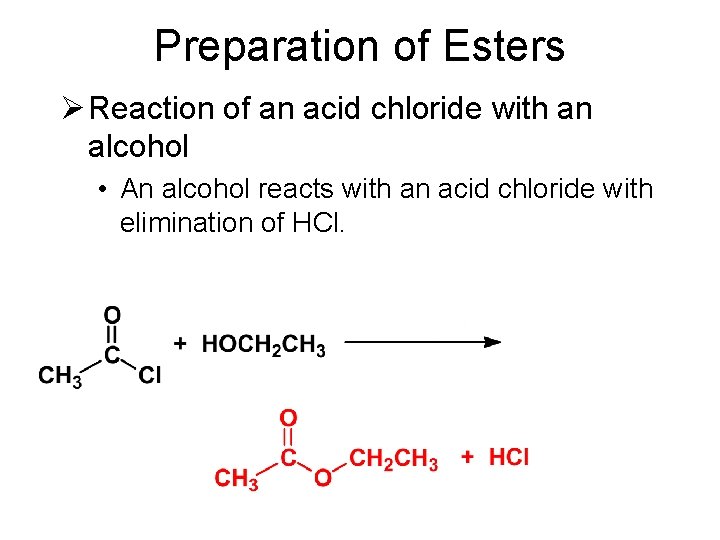
Preparation of Esters Ø Reaction of an acid chloride with an alcohol • An alcohol reacts with an acid chloride with elimination of HCl. • Sometimes an amine base is added to precipitate the HCl that is formed.
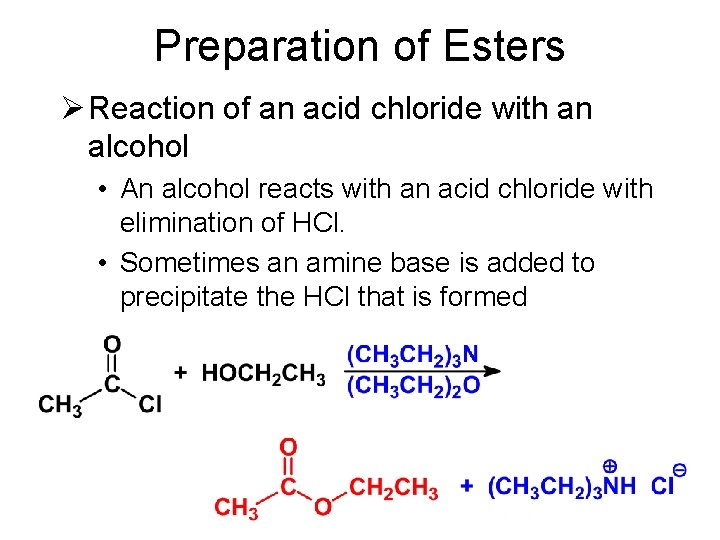
Preparation of Esters Ø Reaction of an acid chloride with an alcohol • An alcohol reacts with an acid chloride with elimination of HCl. • Sometimes an amine base is added to precipitate the HCl that is formed.
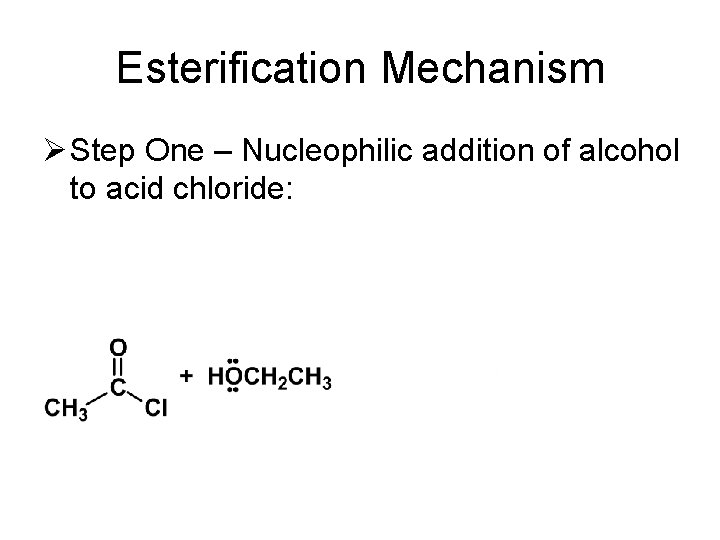
Esterification Mechanism Ø Step One – Nucleophilic addition of alcohol to acid chloride:
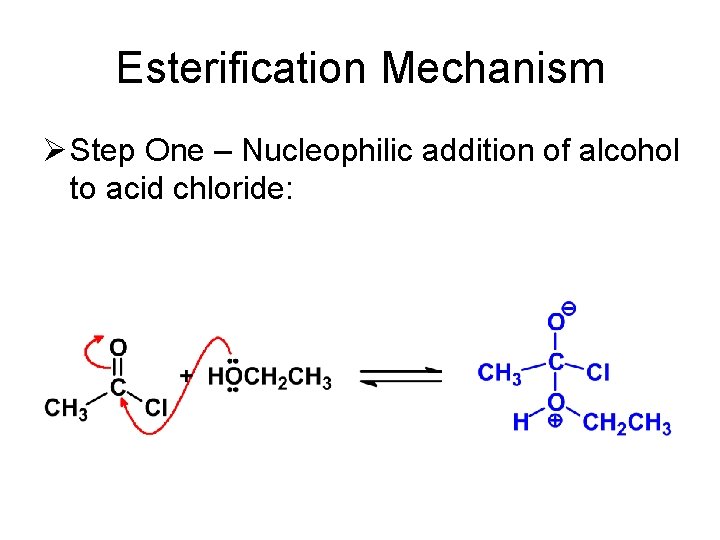
Esterification Mechanism Ø Step One – Nucleophilic addition of alcohol to acid chloride:

Esterification Mechanism Ø Step Two – Deprotonation of the tetrahedral intermediate:
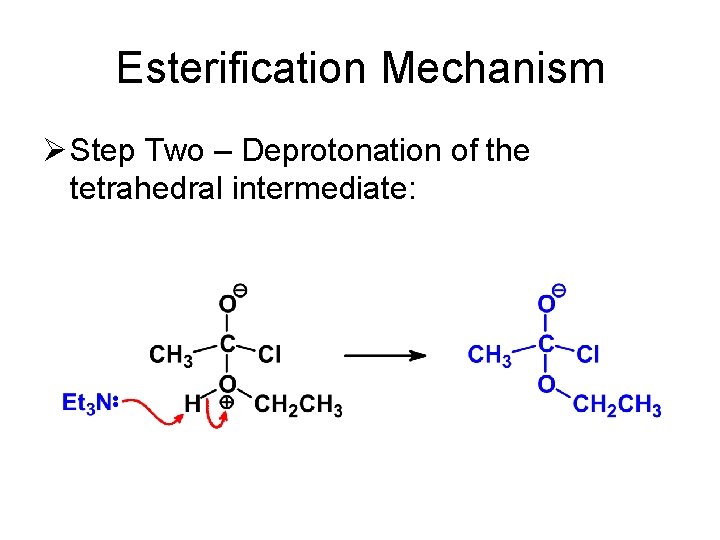
Esterification Mechanism Ø Step Two – Deprotonation of the tetrahedral intermediate:
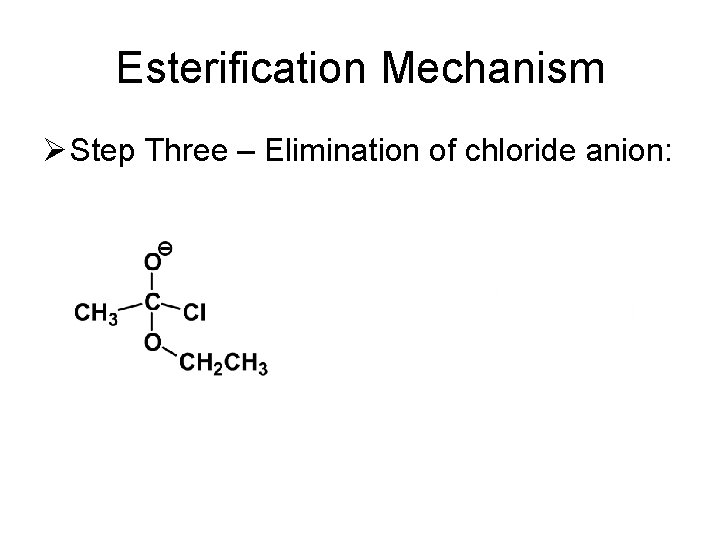
Esterification Mechanism Ø Step Three – Elimination of chloride anion:
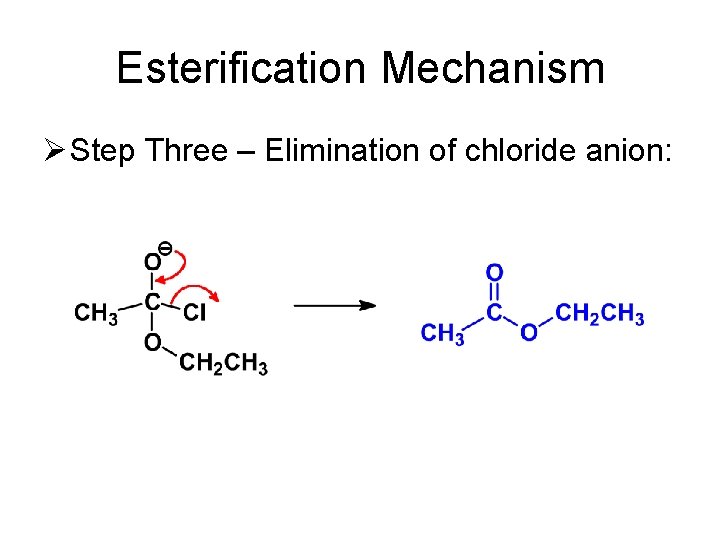
Esterification Mechanism Ø Step Three – Elimination of chloride anion:

Reactions of Esters Ø Hydrolysis • Esters are normally unreactive with water. • However, in the presence of either aqueous acid (aq. HCl or H 2 SO 4) or aqueous base (Na. OH or KOH), they can be hydrolyzed.
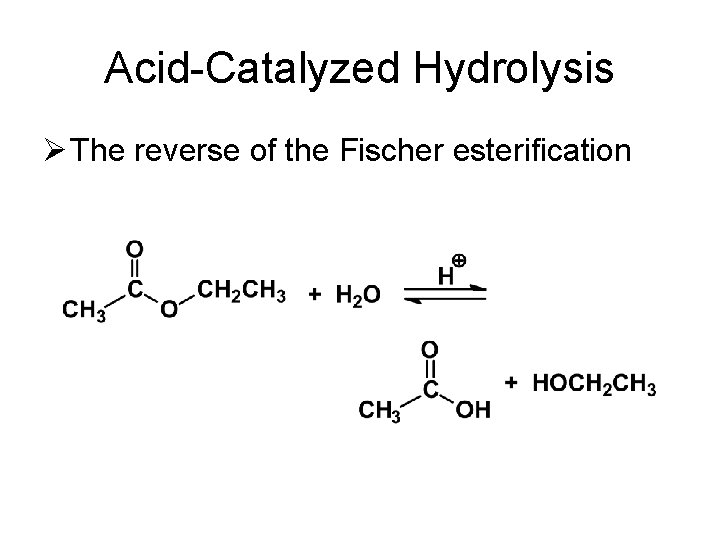
Acid-Catalyzed Hydrolysis Ø The reverse of the Fischer esterification

Base-Promoted Hydrolysis (Saponification) Ø This reaction is used to make soap
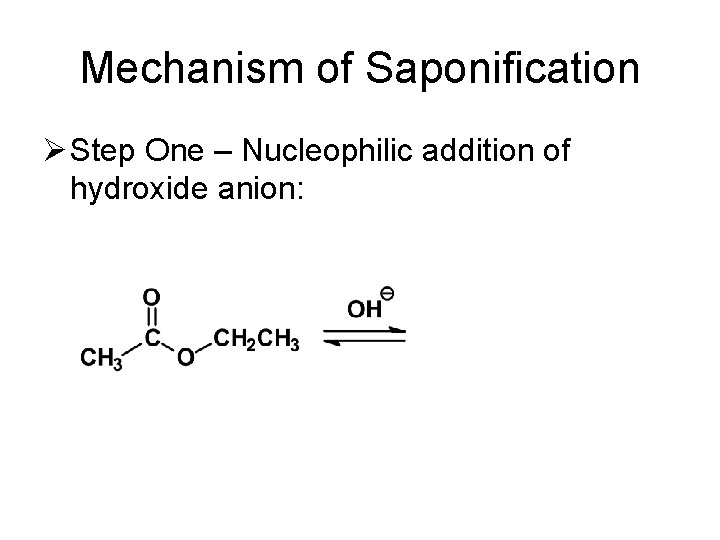
Mechanism of Saponification Ø Step One – Nucleophilic addition of hydroxide anion:
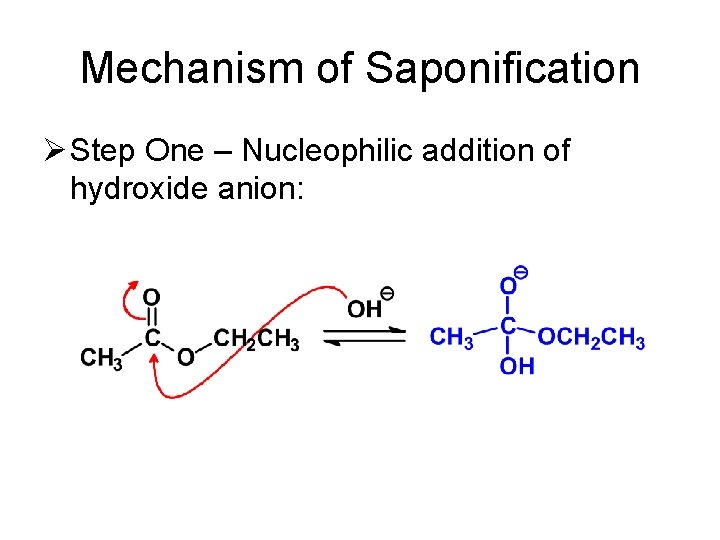
Mechanism of Saponification Ø Step One – Nucleophilic addition of hydroxide anion:
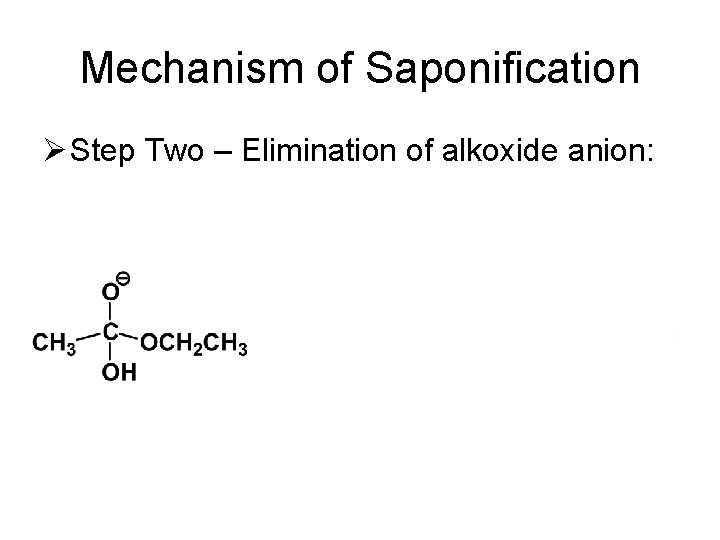
Mechanism of Saponification Ø Step Two – Elimination of alkoxide anion:
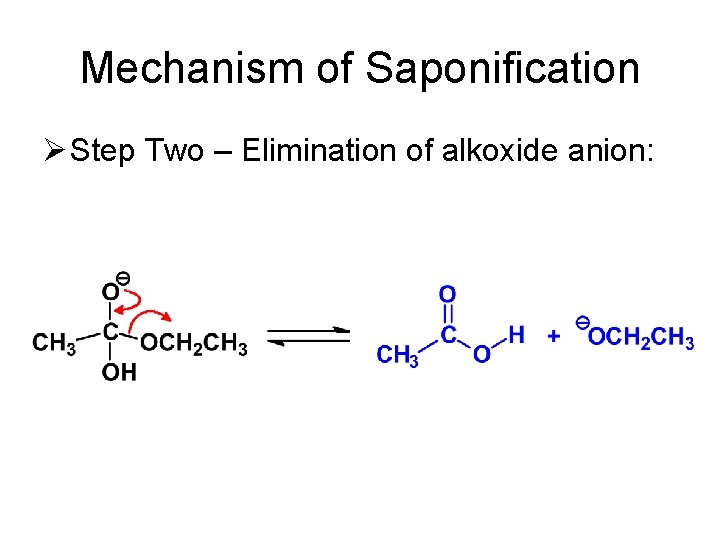
Mechanism of Saponification Ø Step Two – Elimination of alkoxide anion:
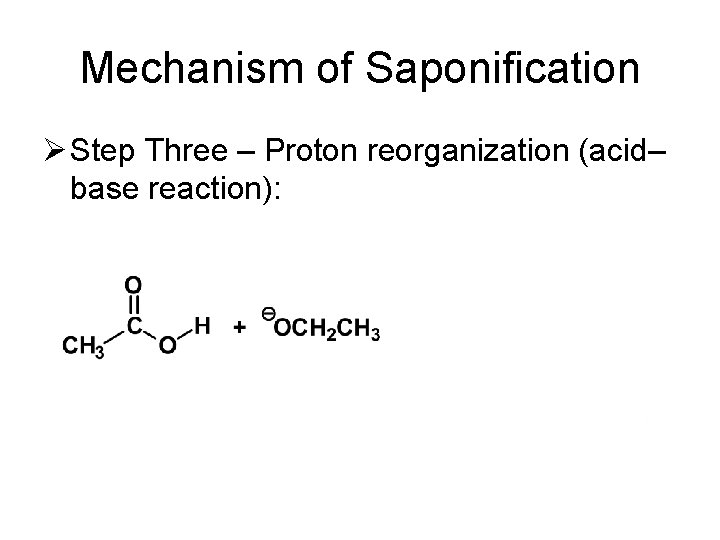
Mechanism of Saponification Ø Step Three – Proton reorganization (acid– base reaction):
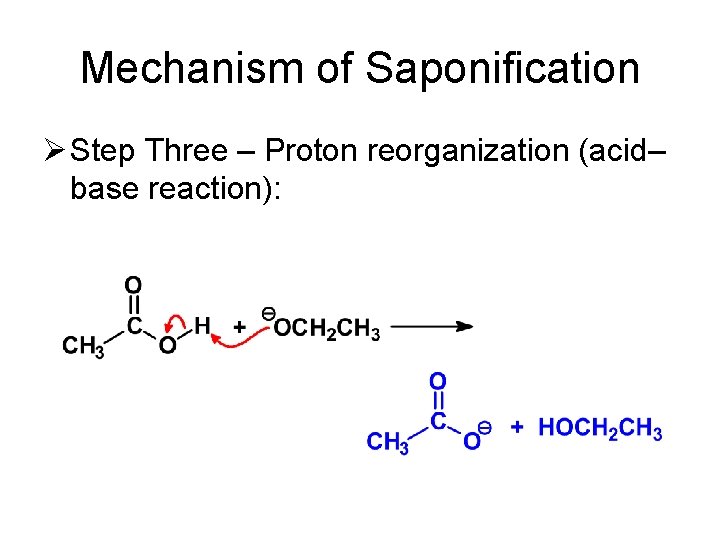
Mechanism of Saponification Ø Step Three – Proton reorganization (acid– base reaction):

Reactions of Anhydrides Ø Esterification (Alcoholysis) • Anhydrides react with alcohols to give one molecule of ester and one molecule of carboxylic acid—a useful method for the preparation of esters.
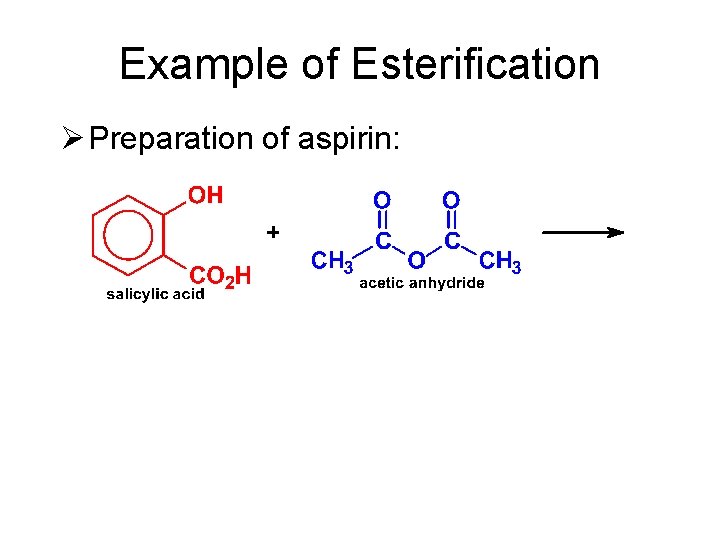
Example of Esterification Ø Preparation of aspirin:
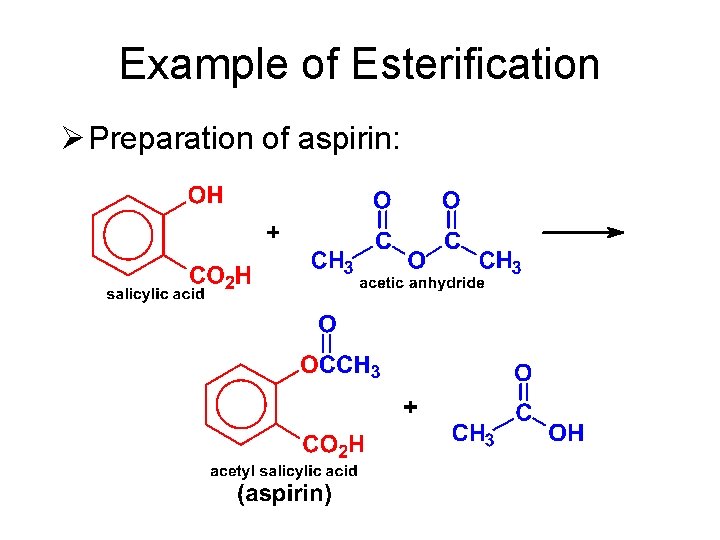
Example of Esterification Ø Preparation of aspirin:

Reactions of Anhydrides Ø Reaction with Ammonia and Amines – Formation of Amides • Anhydrides react with ammonia, as well as 1 o or 2 o amines, to form amides. • Note that two moles of amine are required (one forms the amide, the other acts as a base).
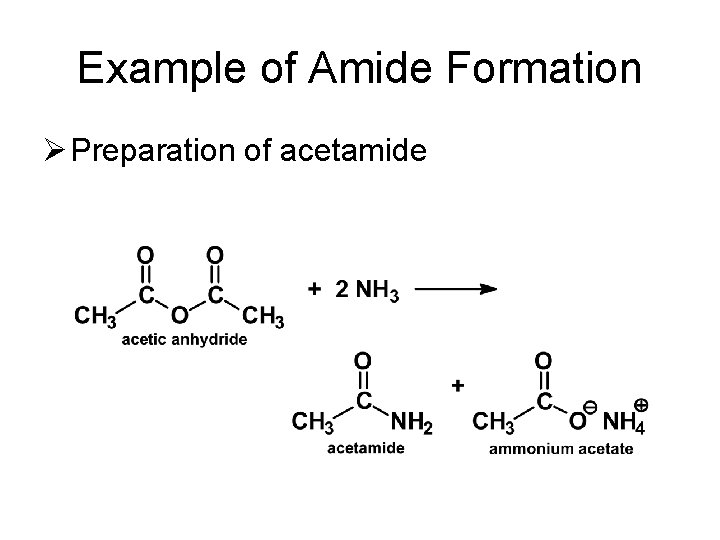
Example of Amide Formation Ø Preparation of acetamide
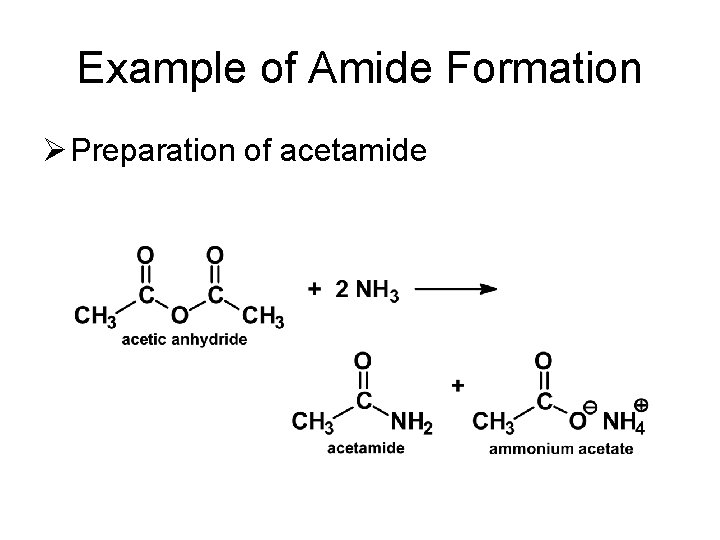
Example of Amide Formation Ø Preparation of acetamide

Example of Amide Formation Ø Preparation of acetaminophen
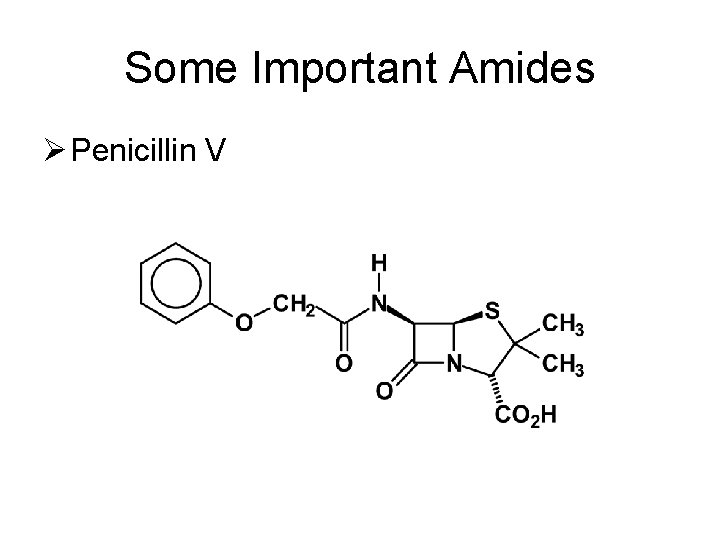
Some Important Amides Ø Penicillin V
- Slides: 40