Carboxylic Acids Natural Products Nomenclature Acidity Preparation Reactions

Carboxylic Acids Natural Products Nomenclature Acidity Preparation Reactions
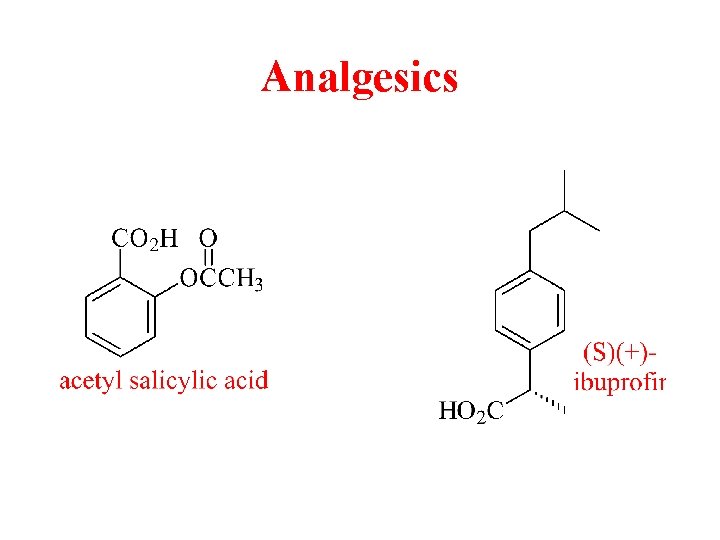
Analgesics
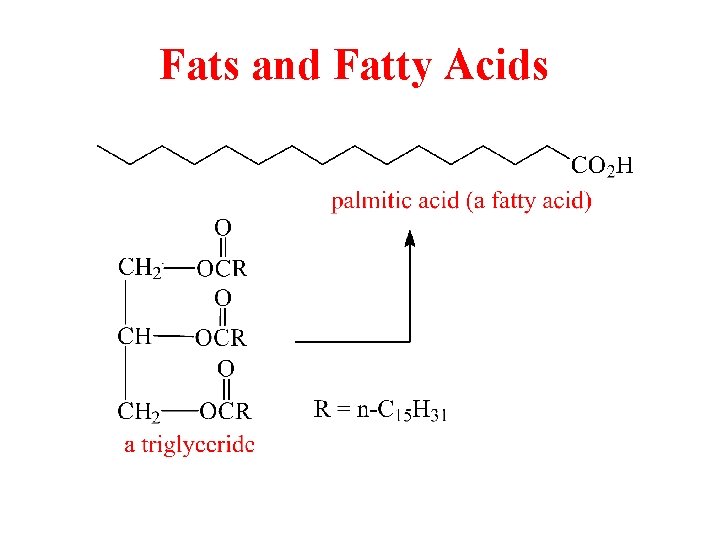
Fats and Fatty Acids
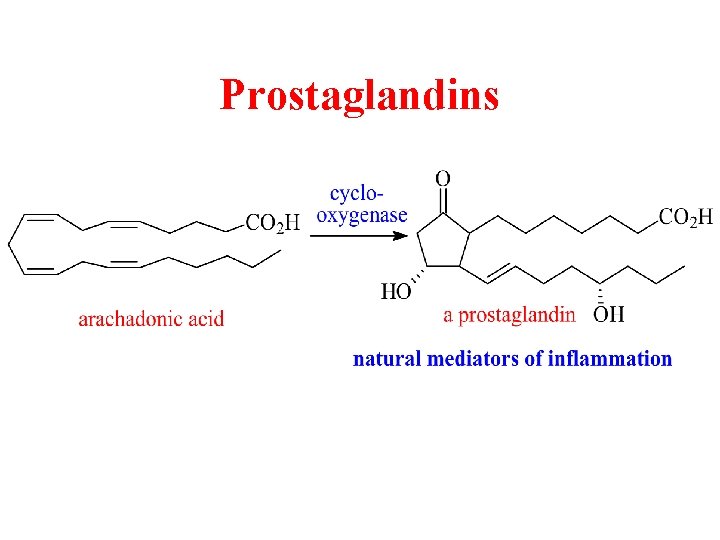
Prostaglandins
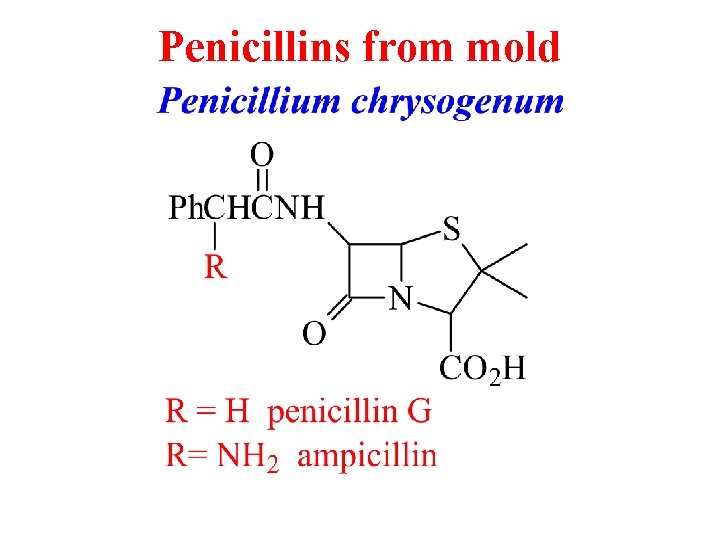
Penicillins from mold
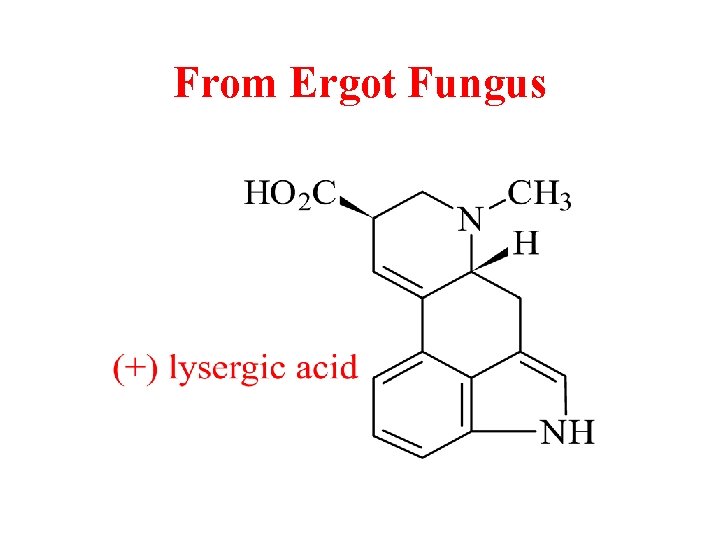
From Ergot Fungus
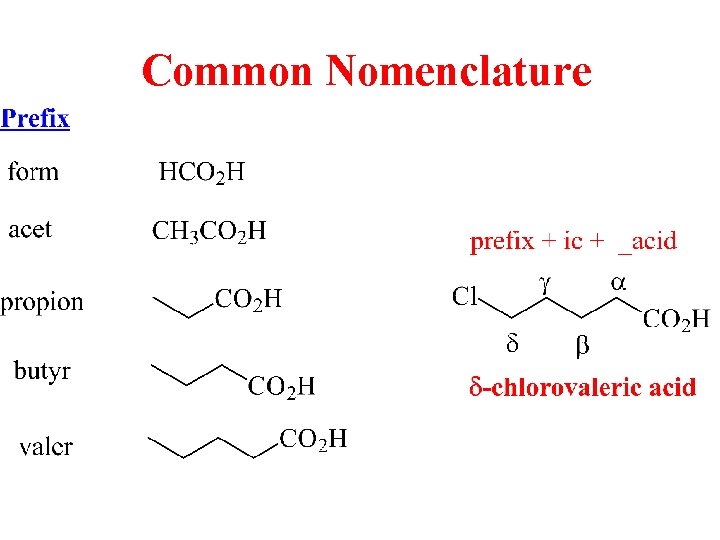
Common Nomenclature
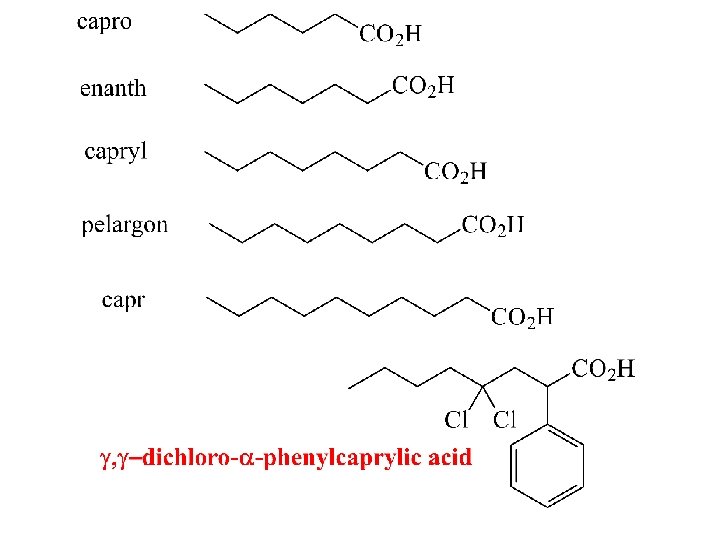
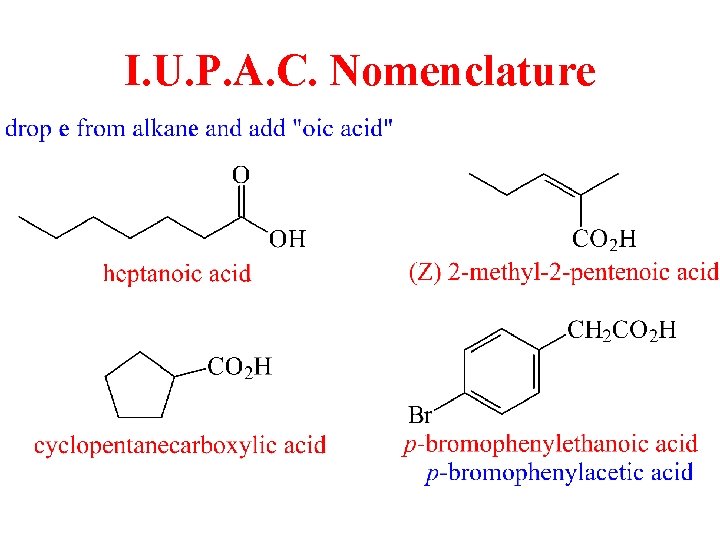
I. U. P. A. C. Nomenclature

Dicarboxylic Acids
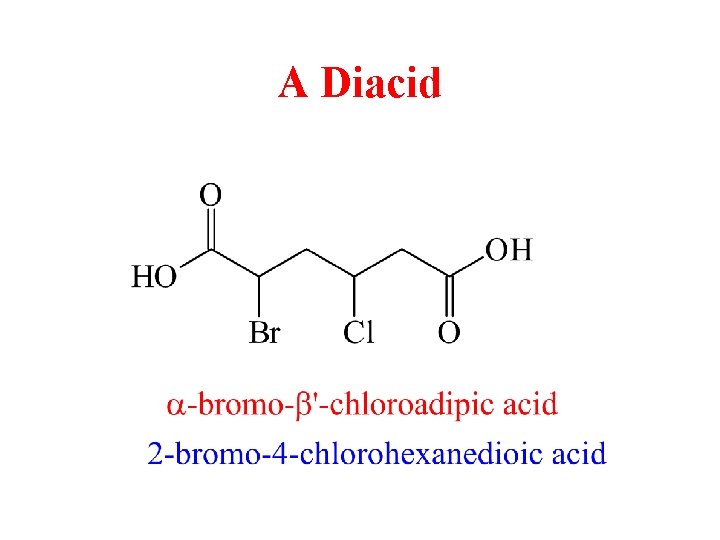
A Diacid
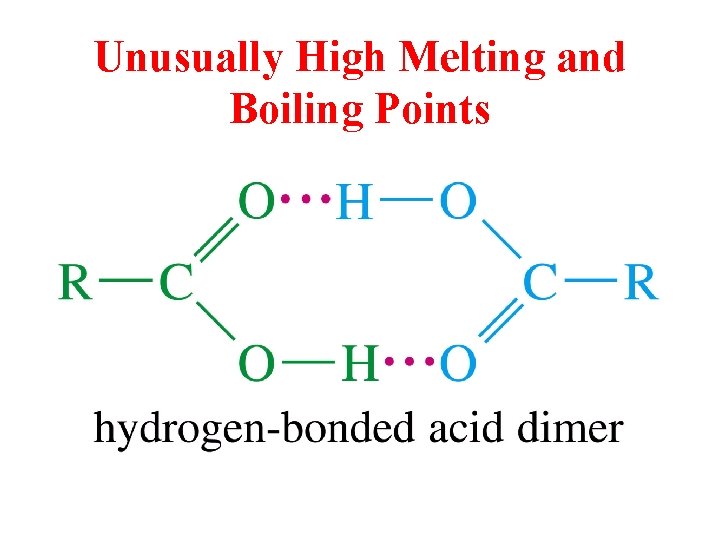
Unusually High Melting and Boiling Points

Boiling Points
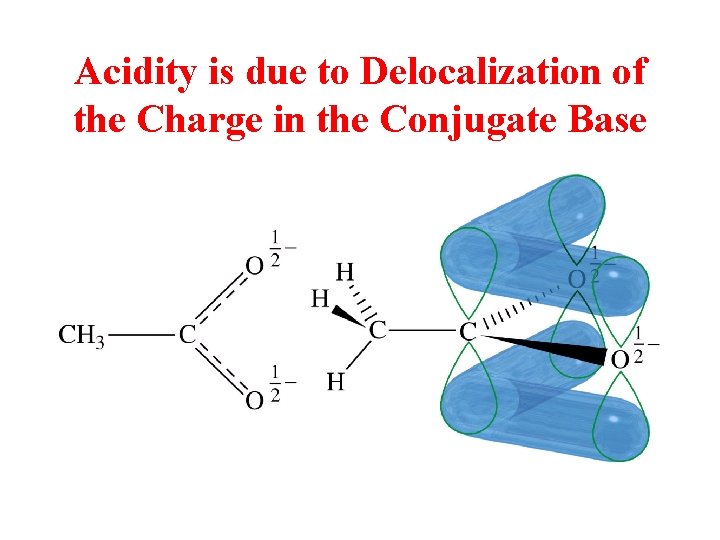
Acidity is due to Delocalization of the Charge in the Conjugate Base
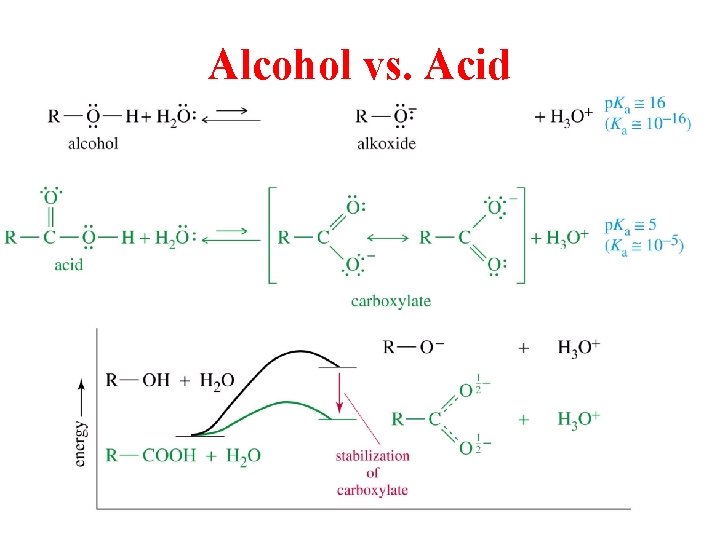
Alcohol vs. Acid
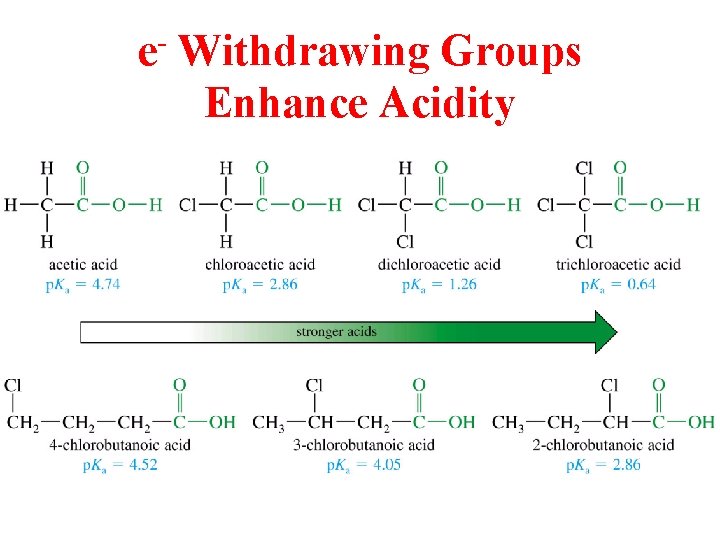
e Withdrawing Groups Enhance Acidity
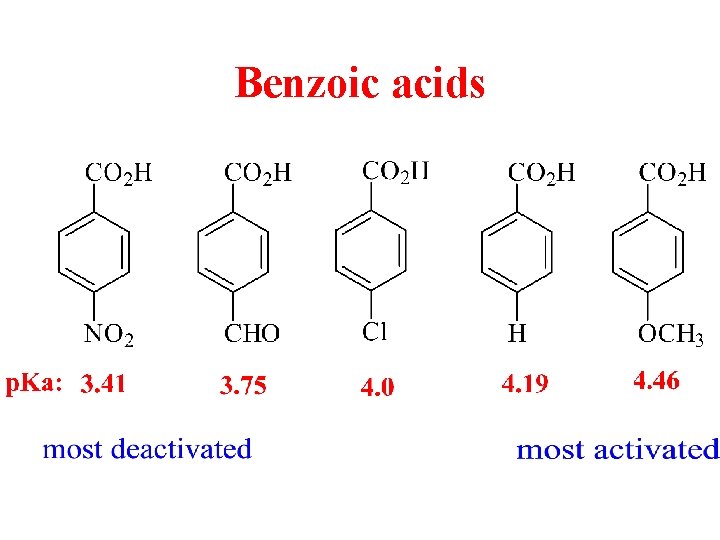
Benzoic acids
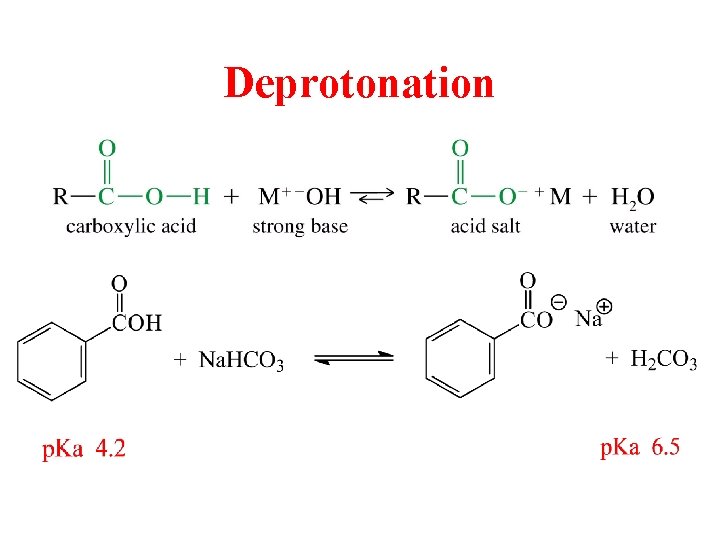
Deprotonation

Carboxylic Acid Preparations • Oxidation of 1 o alcohols and aldehydes with Na 2 Cr 2 O 7/H 2 SO 4 • KMn. O 4 oxidation of alkylbenzenes • Grignard reaction with CO 2 (Carboxylation) • Hydrolysis of a carboxylic derivative: e. g. acid chloride, acid anhydride, ester, amide, nitrile
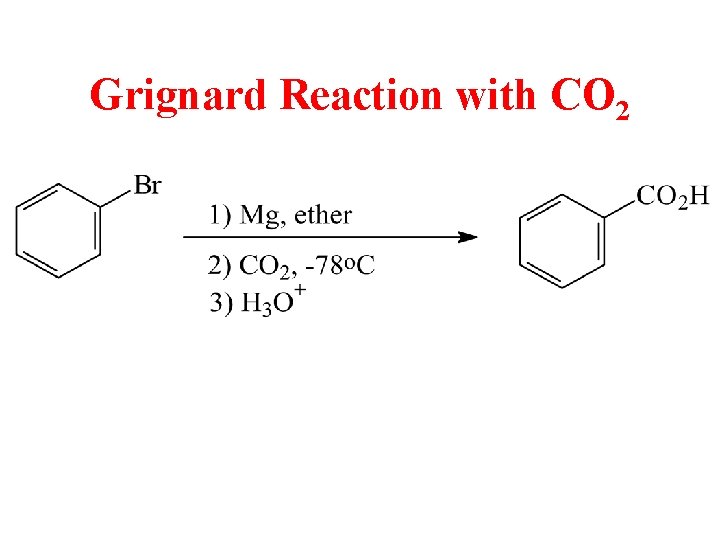
Grignard Reaction with CO 2
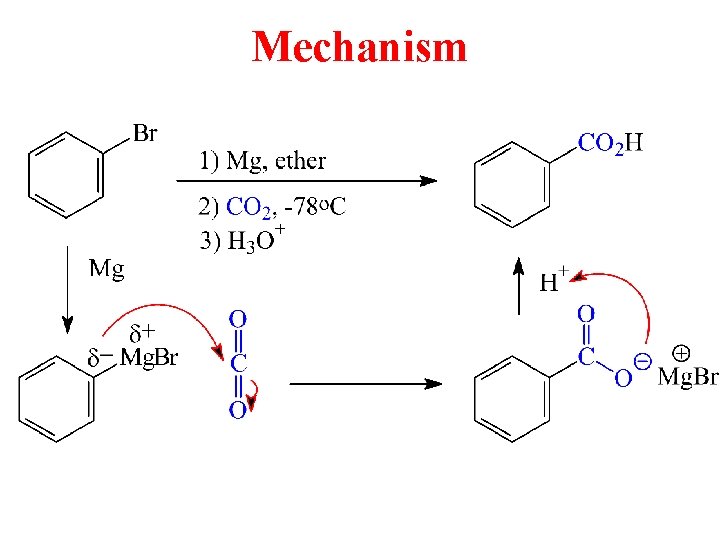
Mechanism
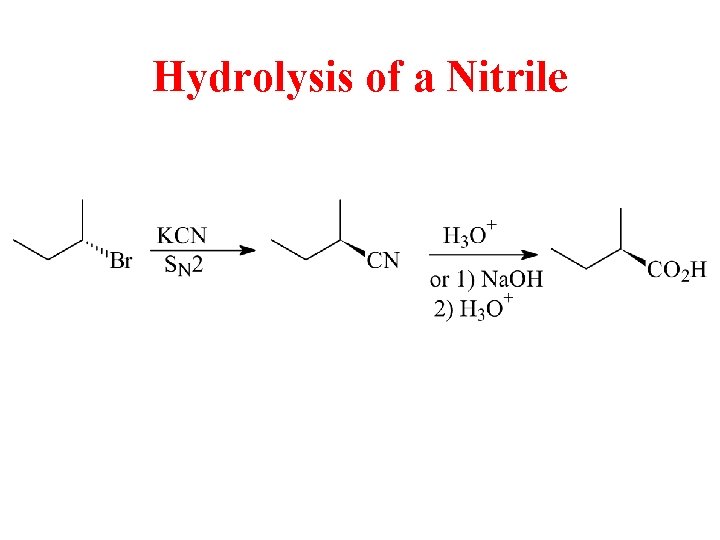
Hydrolysis of a Nitrile
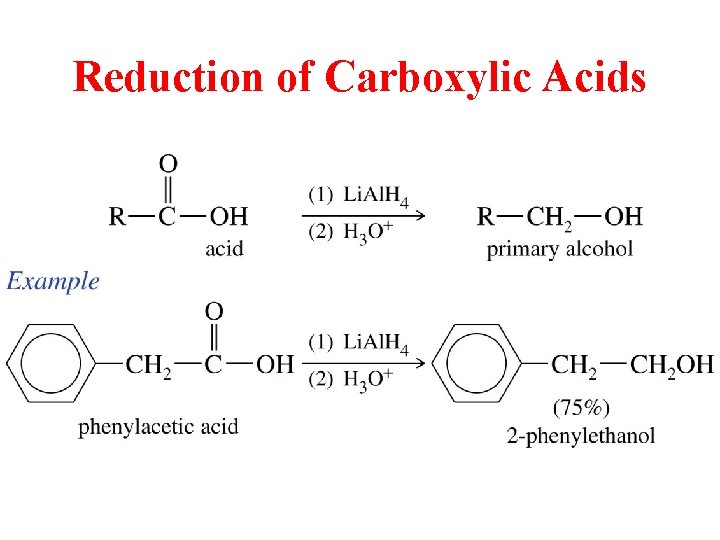
Reduction of Carboxylic Acids

Carboxylic Acid Derivatives Acid Chlorides Acid Anhydrides Esters Amides Nitriles Nucleophilic Acyl Substitution

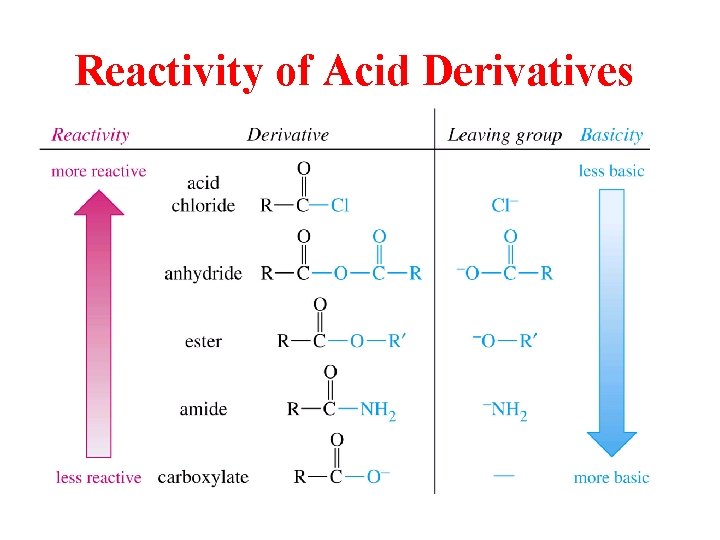
Reactivity of Acid Derivatives

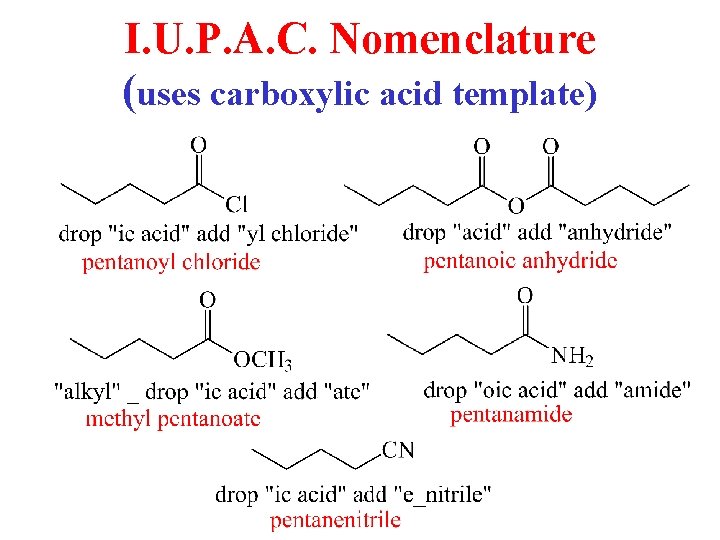
I. U. P. A. C. Nomenclature (uses carboxylic acid template)
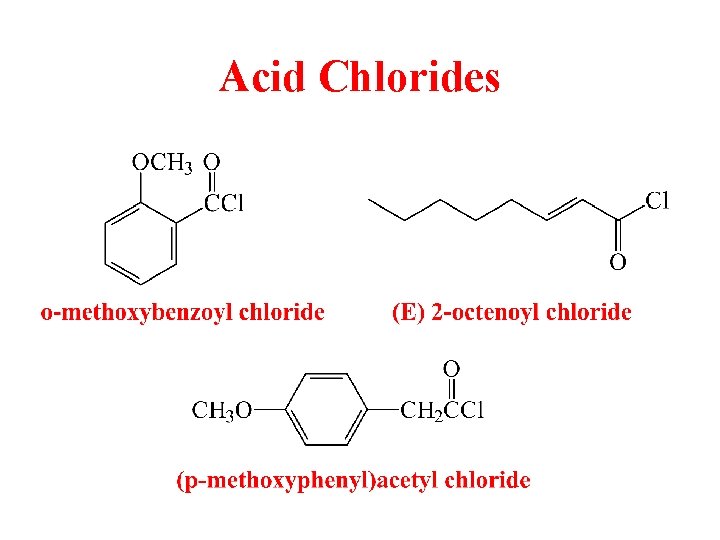
Acid Chlorides
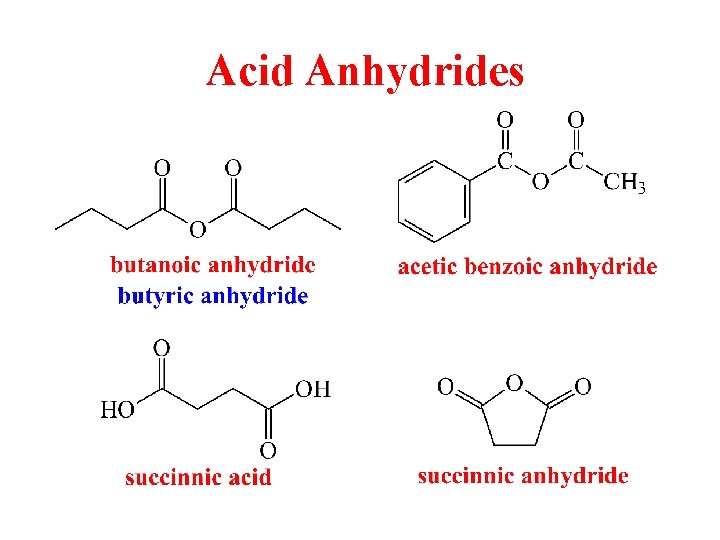
Acid Anhydrides
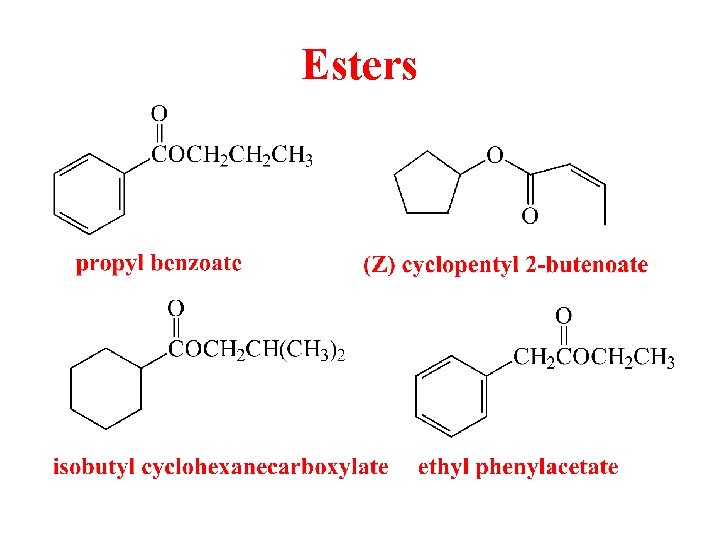
Esters
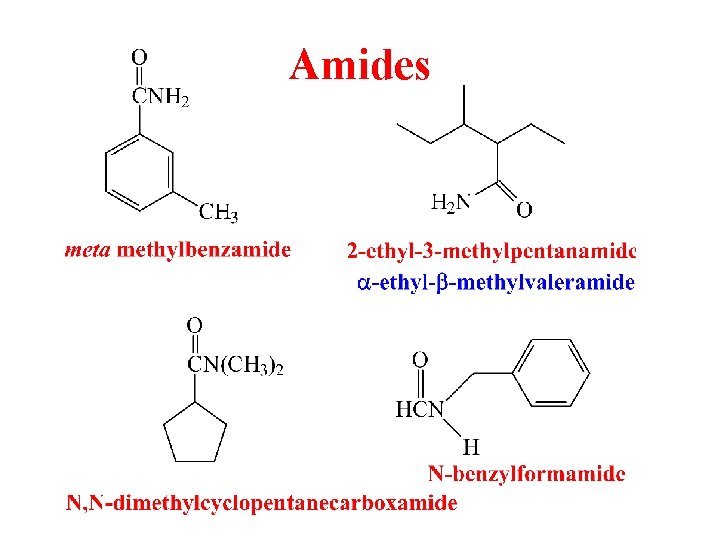
Amides
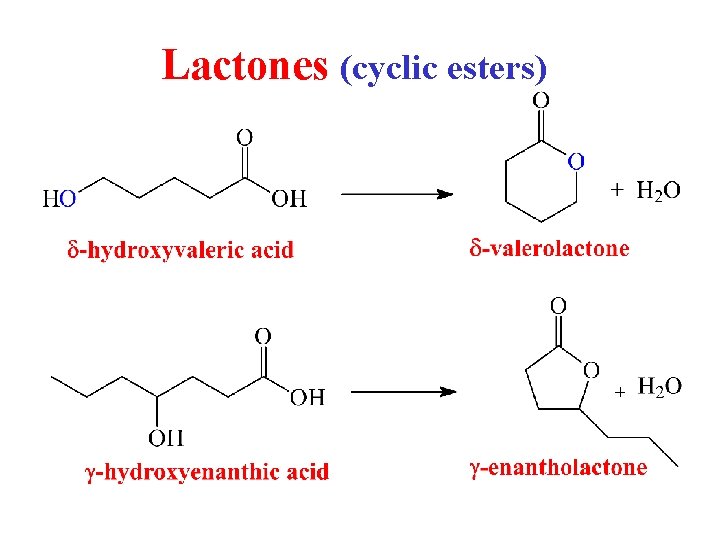
Lactones (cyclic esters)
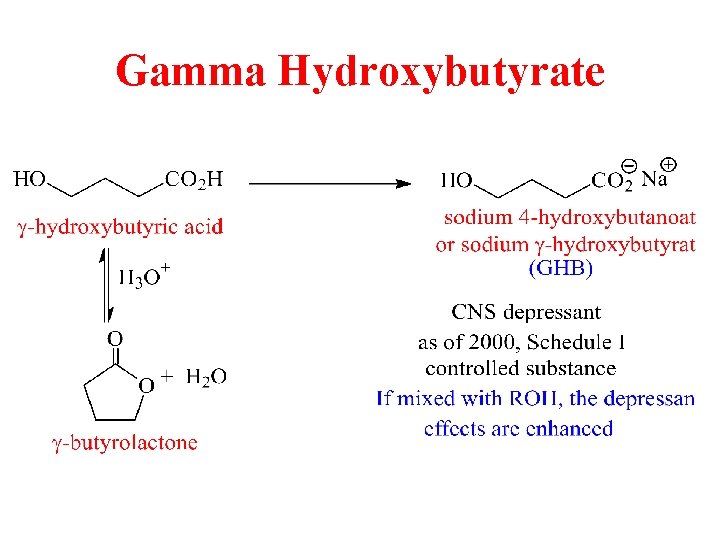
Gamma Hydroxybutyrate
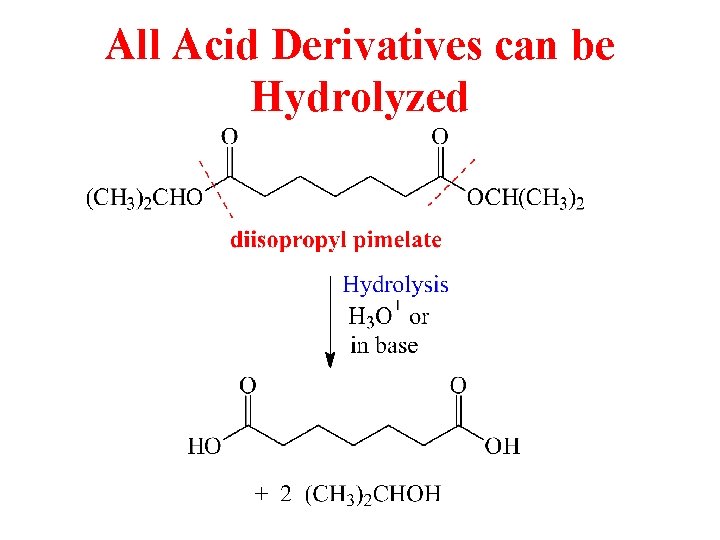
All Acid Derivatives can be Hydrolyzed
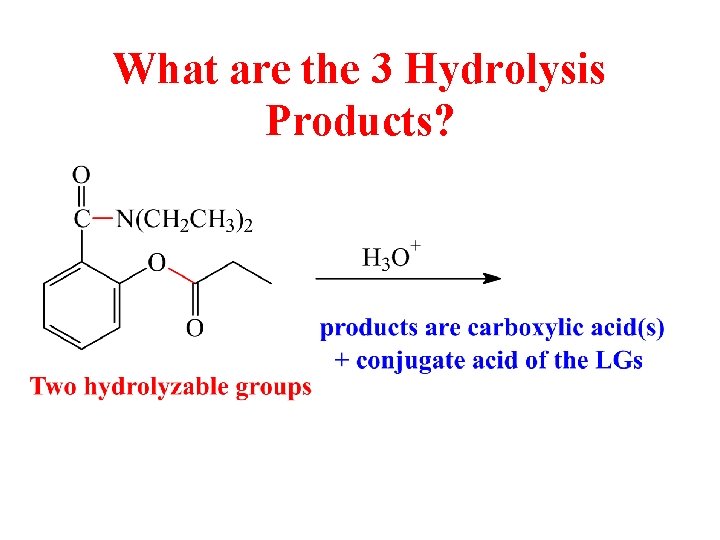
What are the 3 Hydrolysis Products?

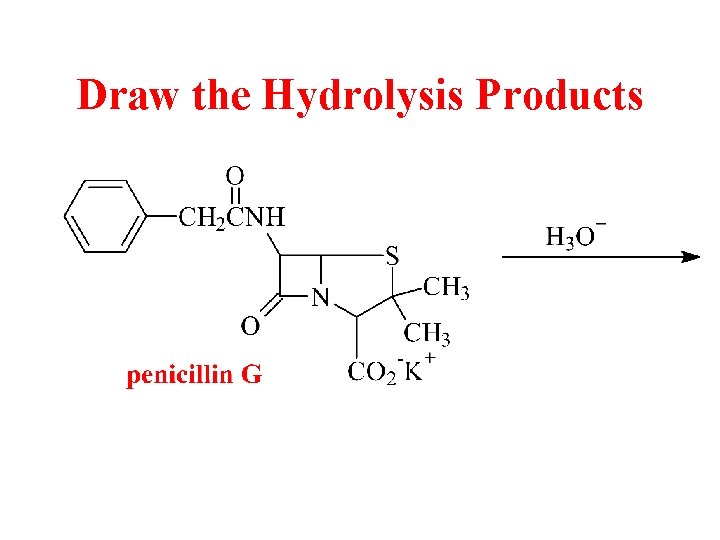
Draw the Hydrolysis Products
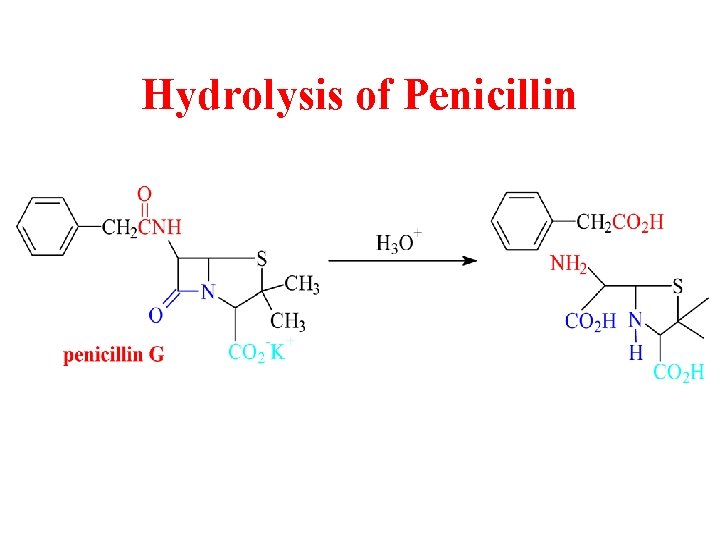
Hydrolysis of Penicillin
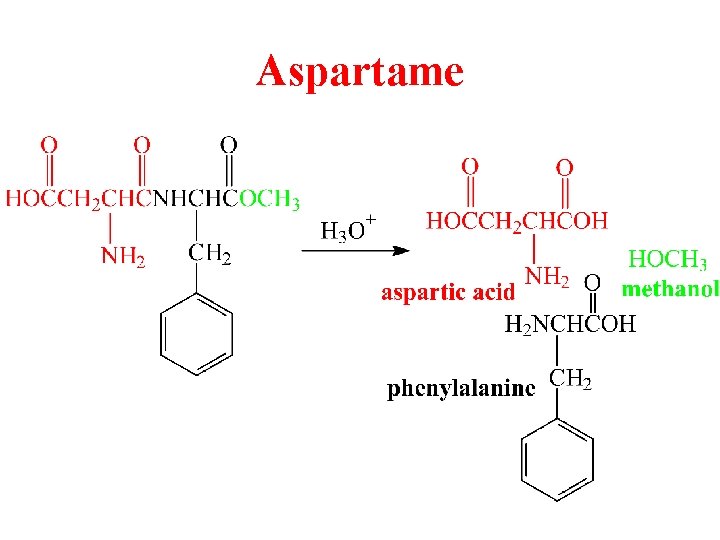
Aspartame
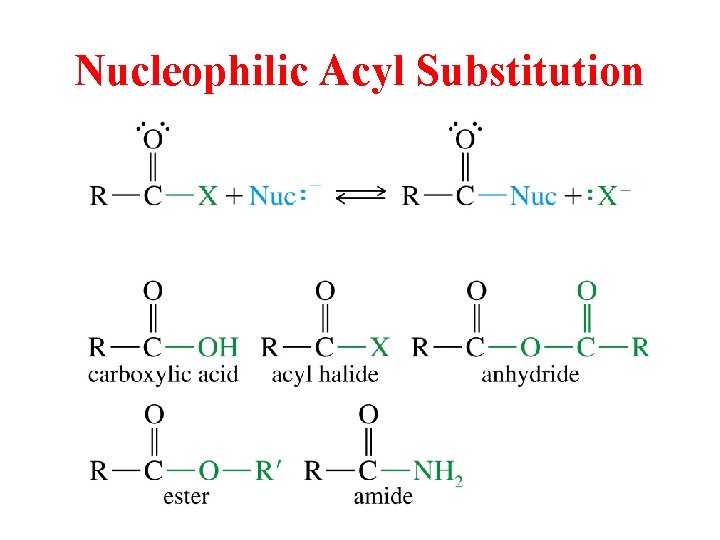
Nucleophilic Acyl Substitution
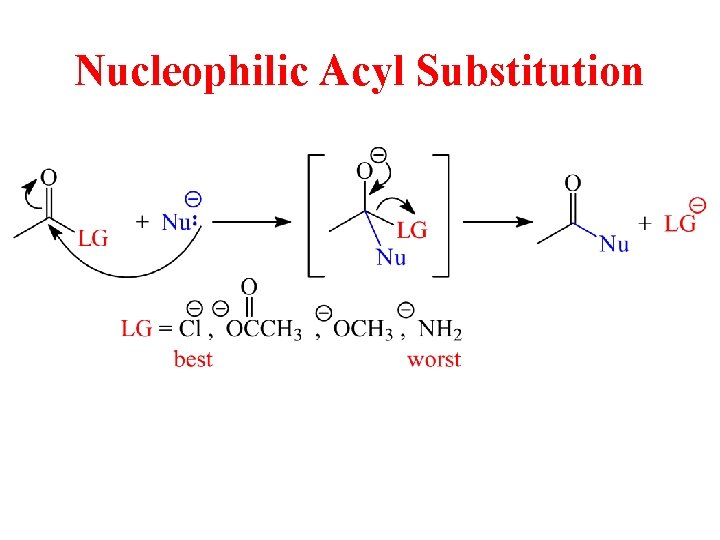
Nucleophilic Acyl Substitution
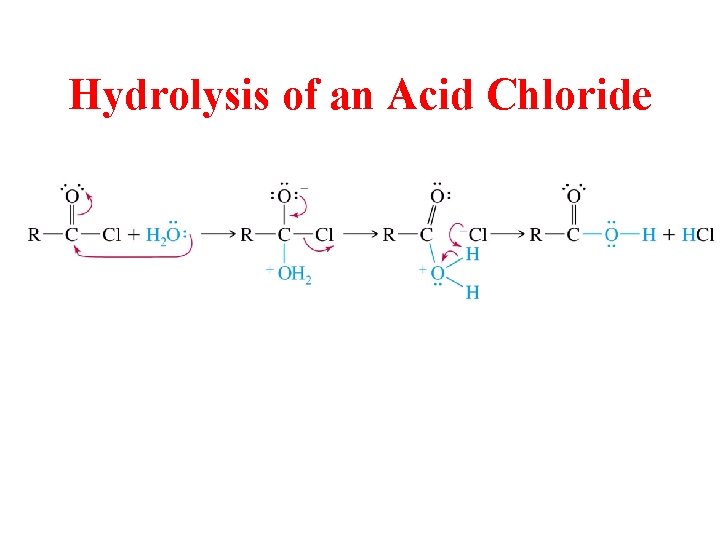
Hydrolysis of an Acid Chloride
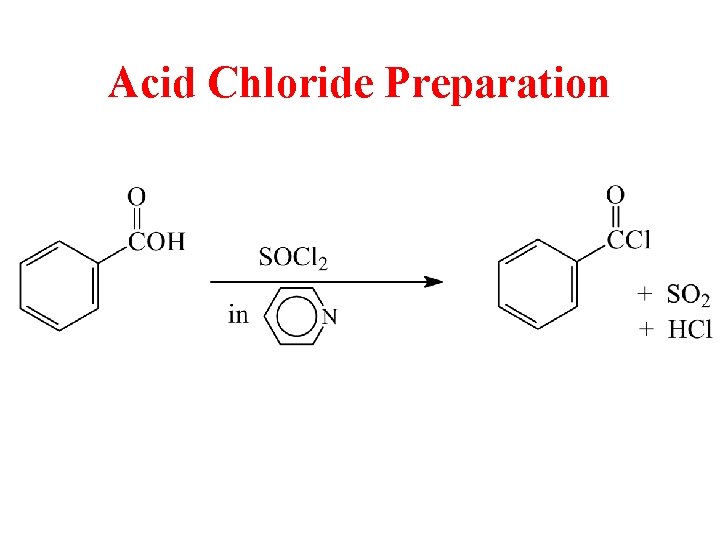
Acid Chloride Preparation

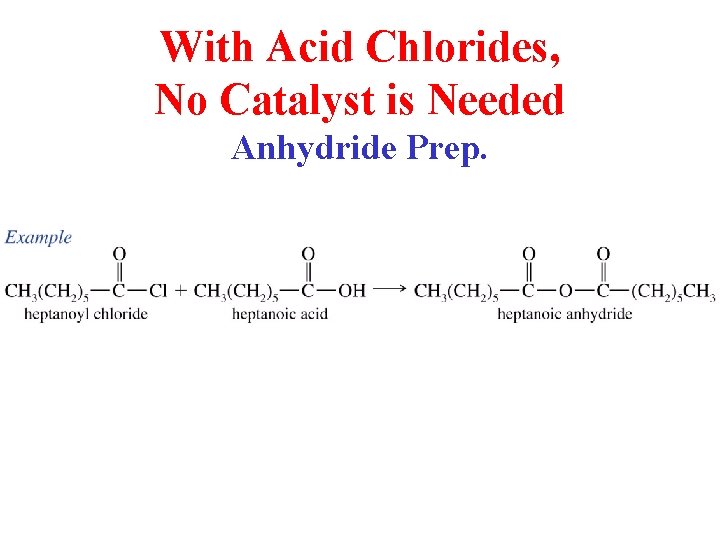
With Acid Chlorides, No Catalyst is Needed Anhydride Prep.

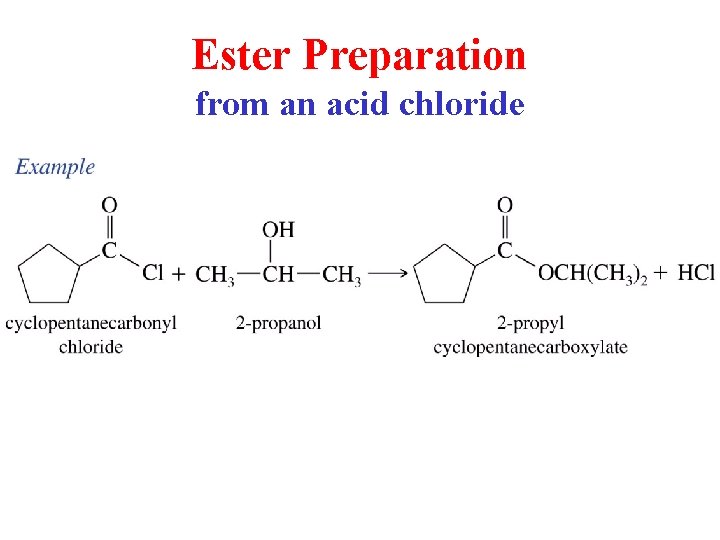
Ester Preparation from an acid chloride
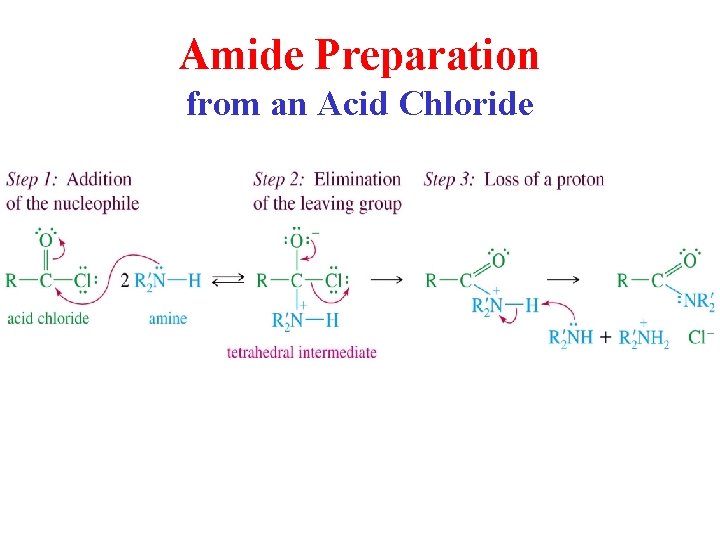
Amide Preparation from an Acid Chloride
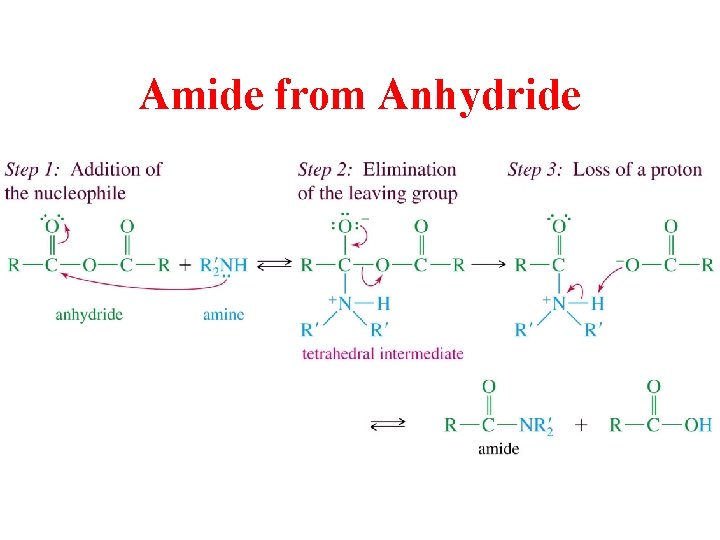
Amide from Anhydride
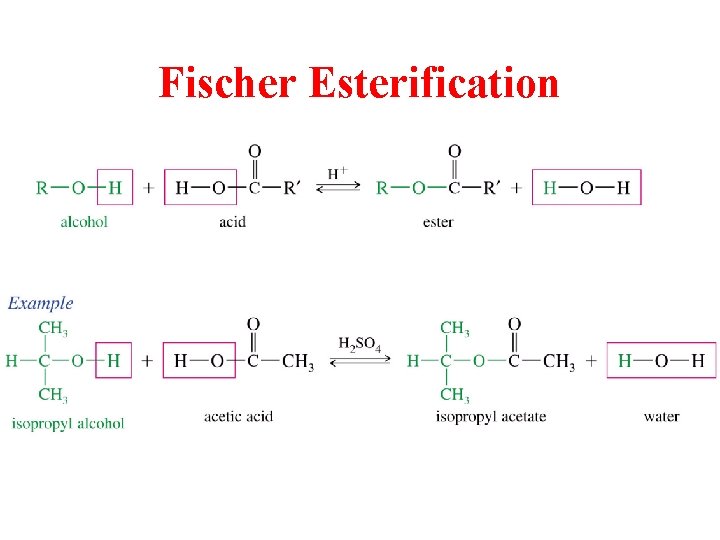
Fischer Esterification
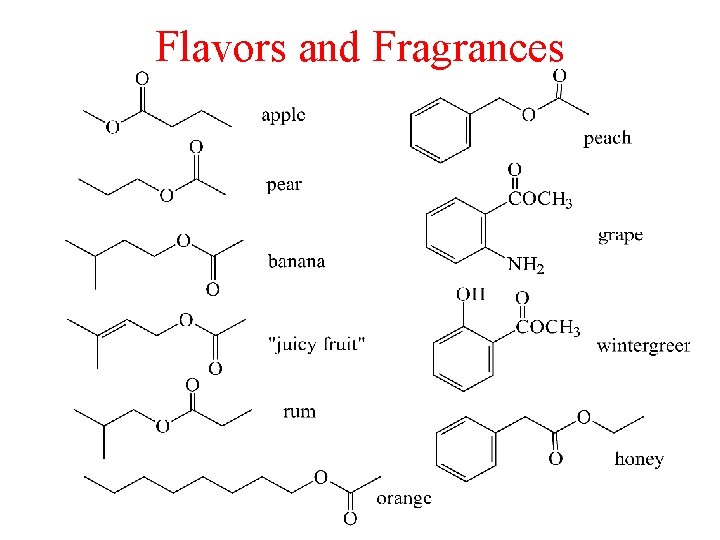
Flavors and Fragrances
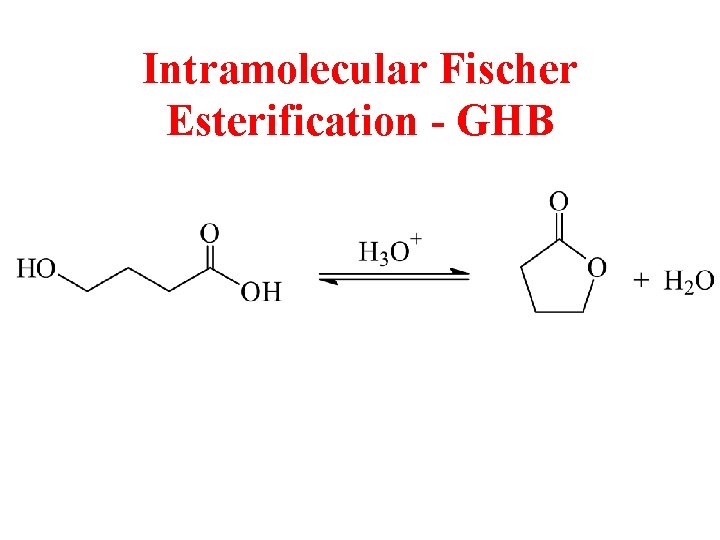
Intramolecular Fischer Esterification - GHB

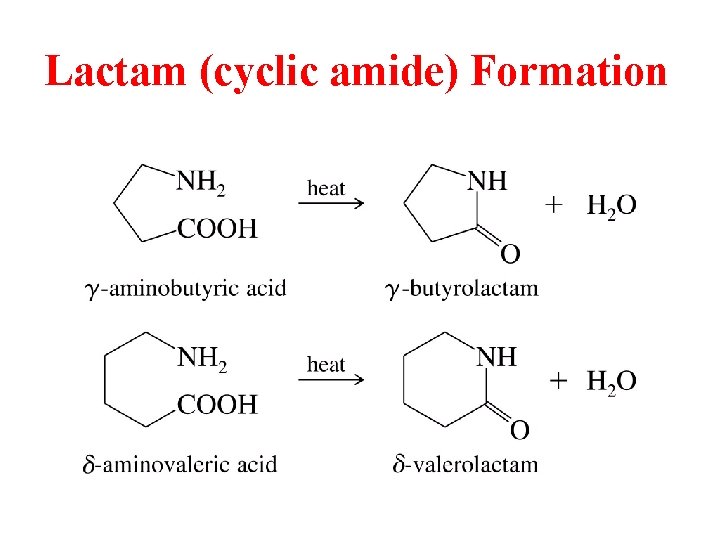
Lactam (cyclic amide) Formation
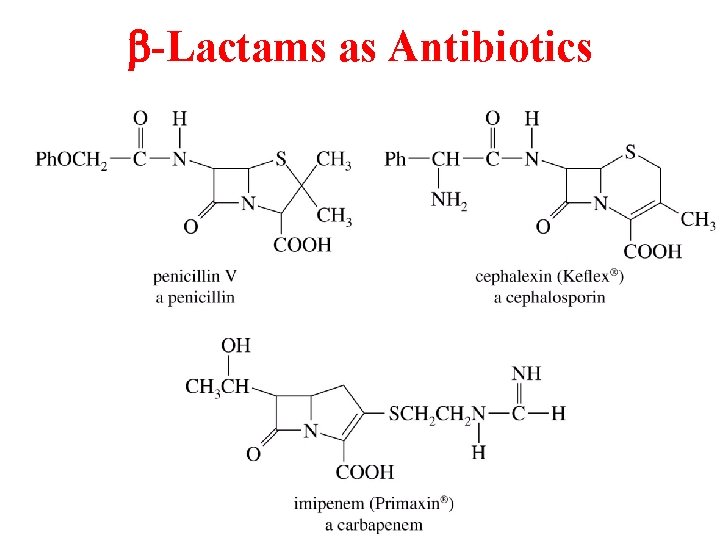
b-Lactams as Antibiotics
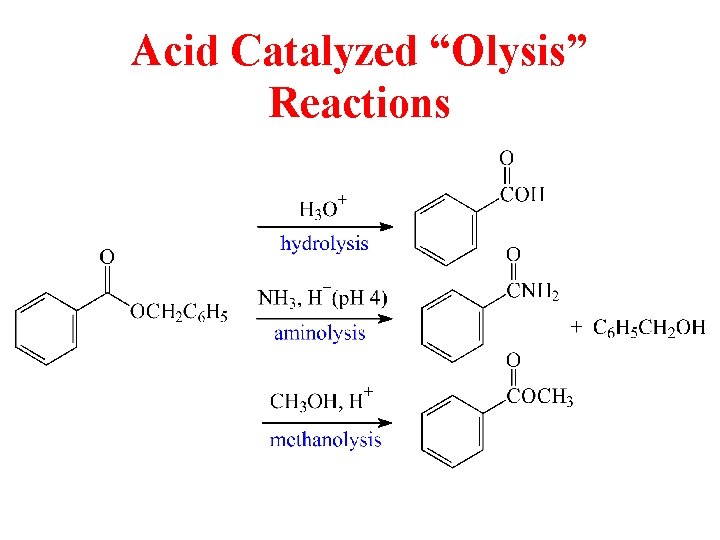
Acid Catalyzed “Olysis” Reactions
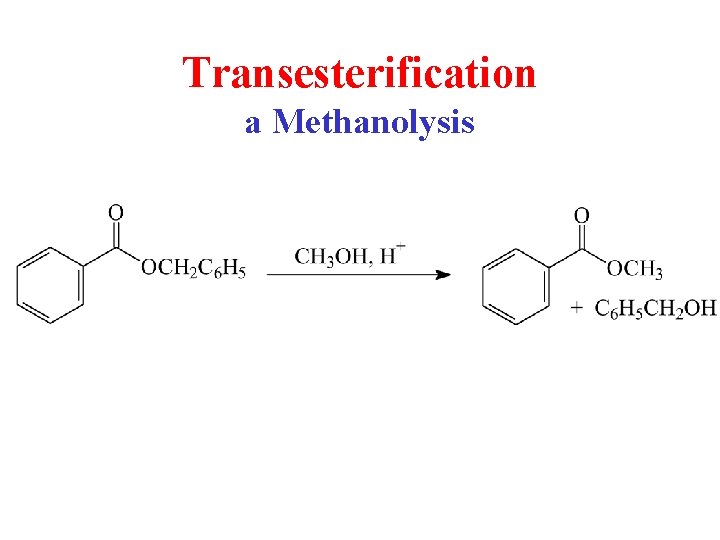
Transesterification a Methanolysis

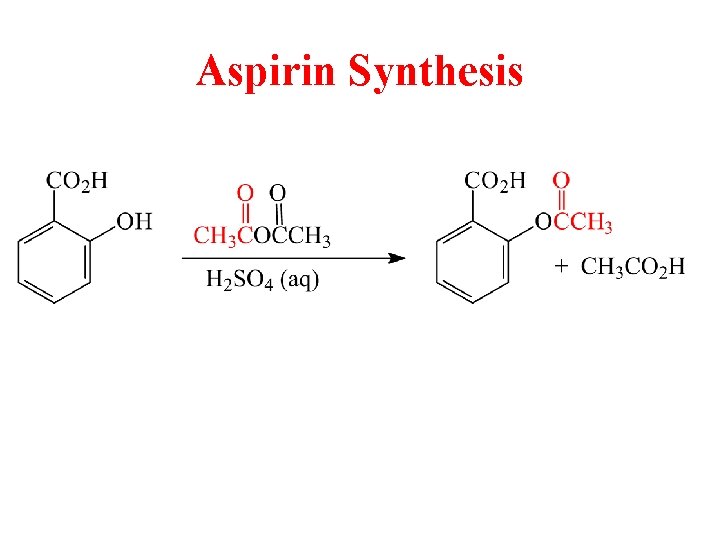
Aspirin Synthesis

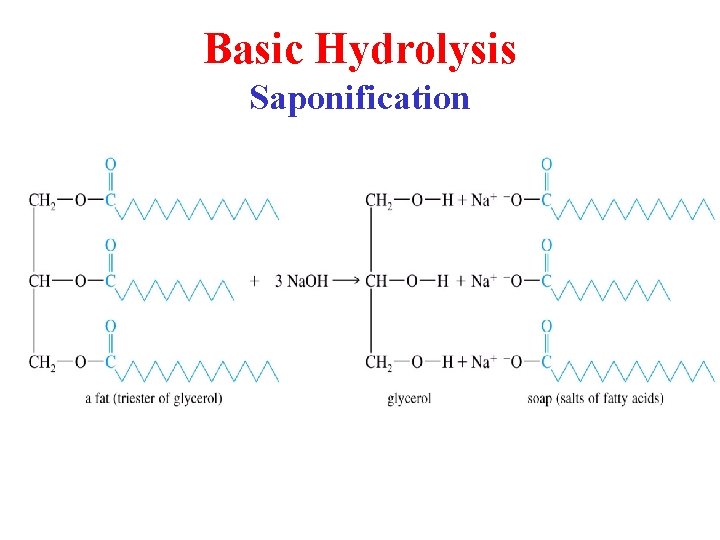
Basic Hydrolysis Saponification
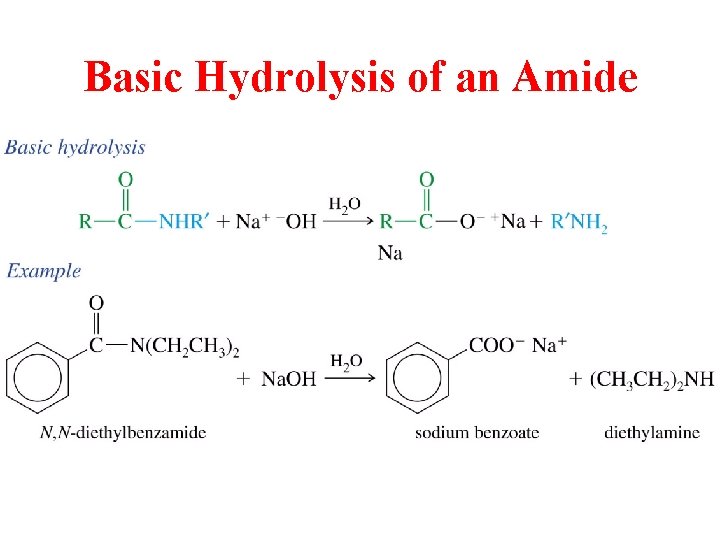
Basic Hydrolysis of an Amide
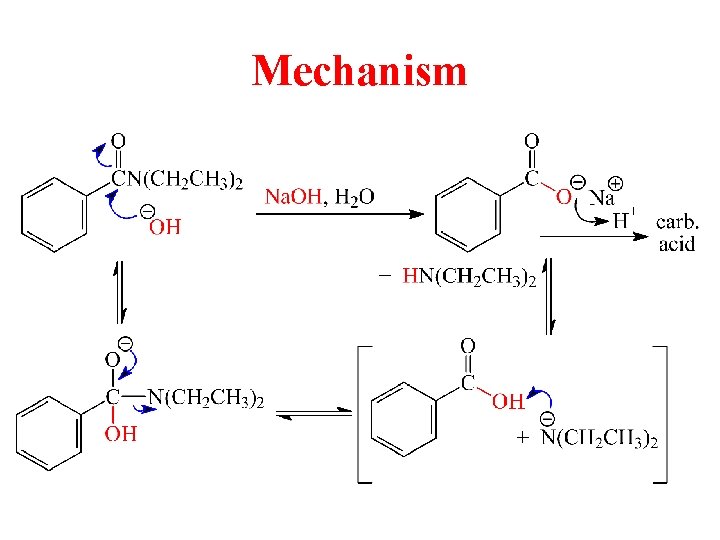
Mechanism
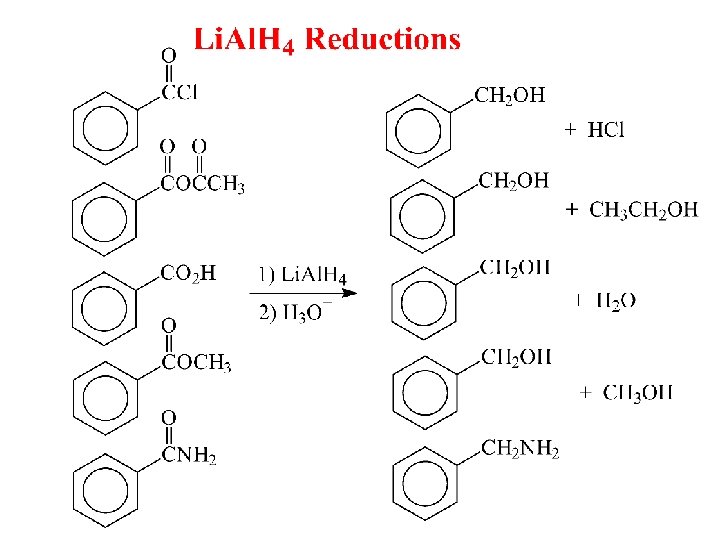
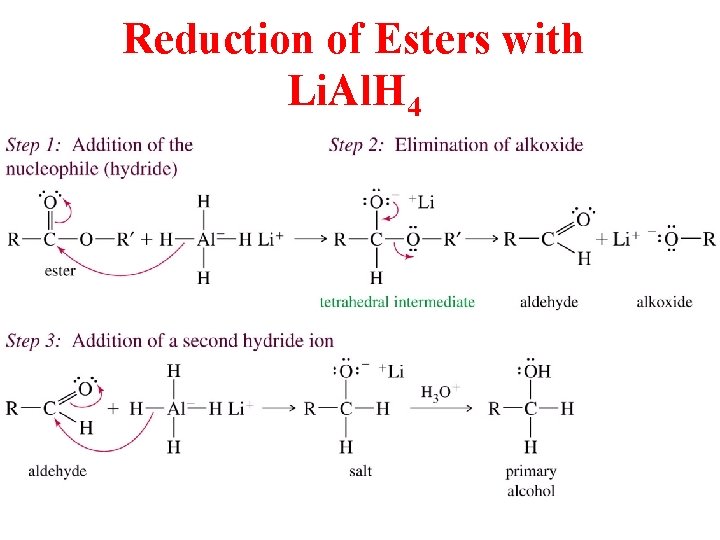
Reduction of Esters with Li. Al. H 4
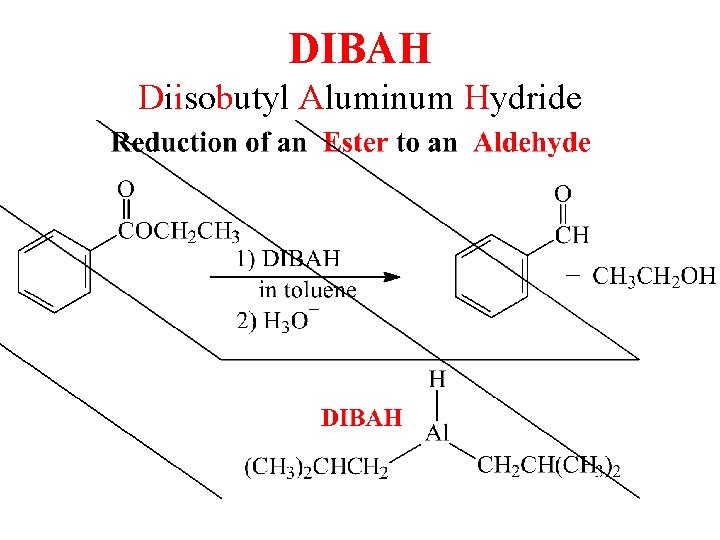
DIBAH Diisobutyl Aluminum Hydride
- Slides: 67