Carboxylic Acids Have hydroxyl group bonded to carbonyl
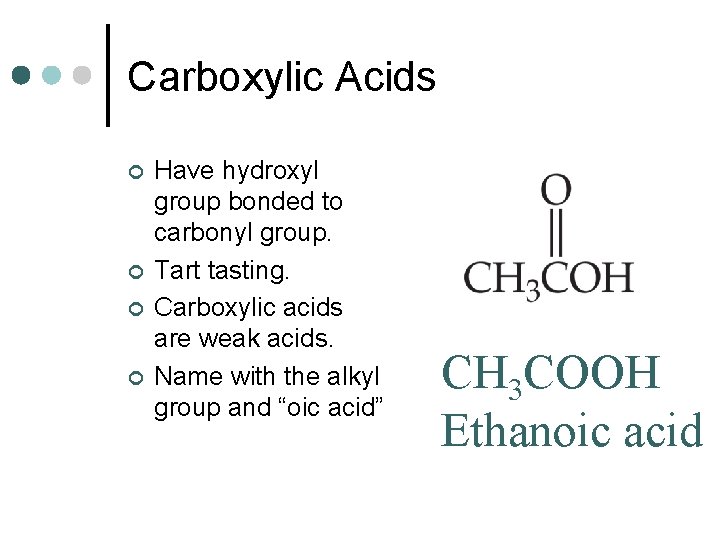
Carboxylic Acids ¢ ¢ Have hydroxyl group bonded to carbonyl group. Tart tasting. Carboxylic acids are weak acids. Name with the alkyl group and “oic acid” CH 3 COOH Ethanoic acid
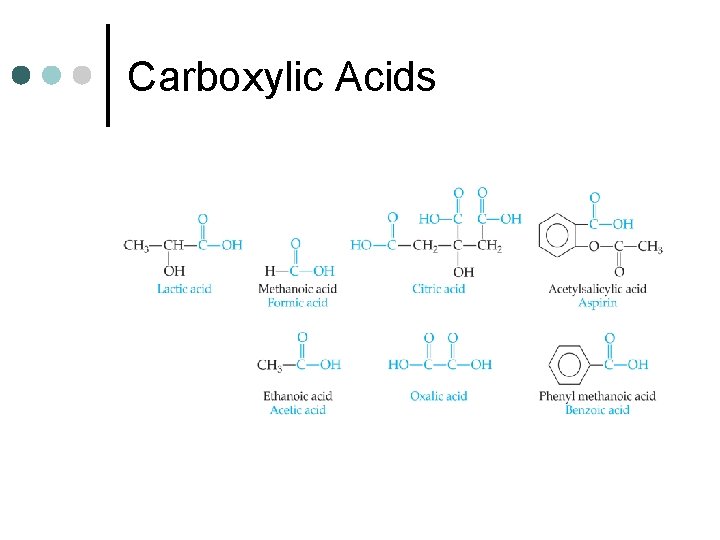
Carboxylic Acids
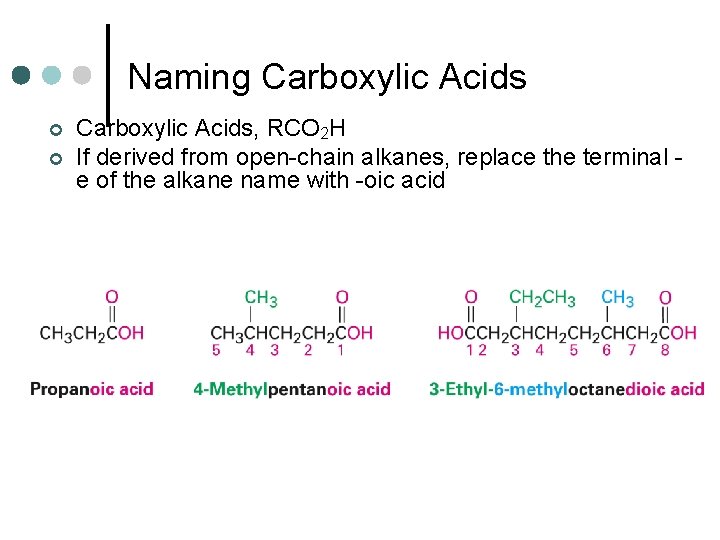
Naming Carboxylic Acids ¢ ¢ Carboxylic Acids, RCO 2 H If derived from open-chain alkanes, replace the terminal e of the alkane name with -oic acid
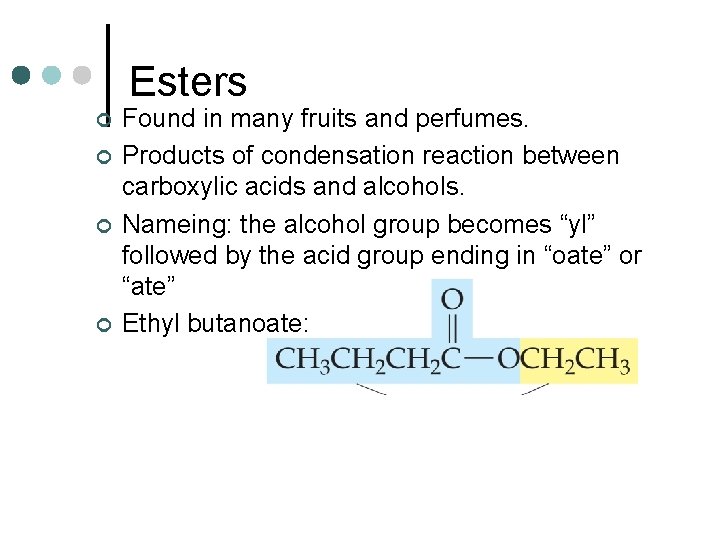
Esters ¢ ¢ Found in many fruits and perfumes. Products of condensation reaction between carboxylic acids and alcohols. Nameing: the alcohol group becomes “yl” followed by the acid group ending in “oate” or “ate” Ethyl butanoate:
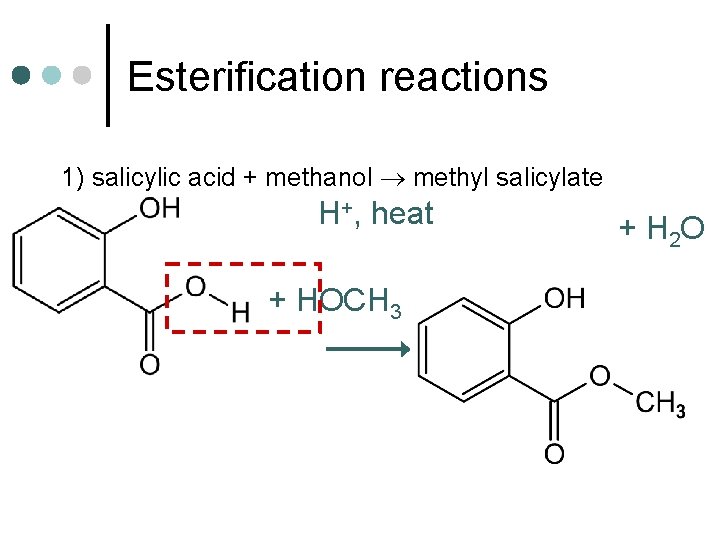
Esterification reactions 1) salicylic acid + methanol methyl salicylate H+, heat + HOCH 3 + H 2 O
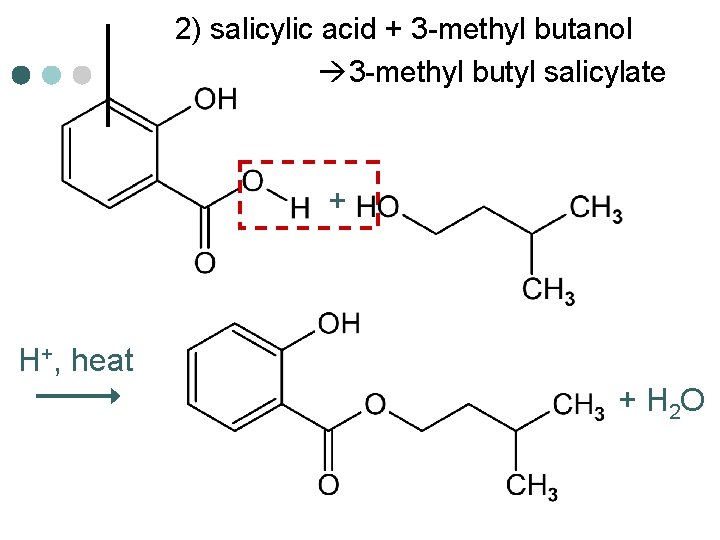
2) salicylic acid + 3 -methyl butanol 3 -methyl butyl salicylate + H+, heat + H 2 O
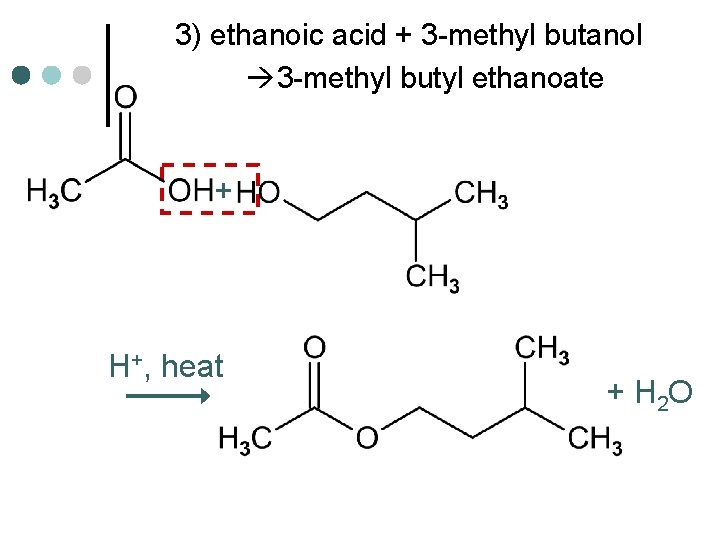
3) ethanoic acid + 3 -methyl butanol 3 -methyl butyl ethanoate + H+, heat + H 2 O
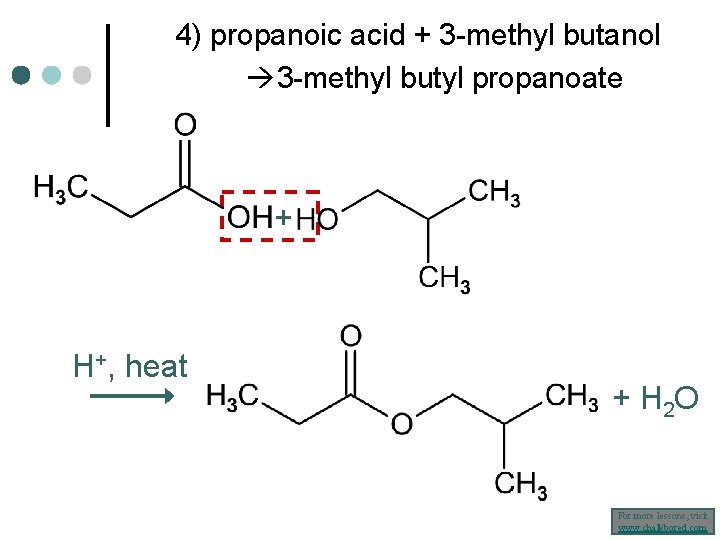
4) propanoic acid + 3 -methyl butanol 3 -methyl butyl propanoate + H+, heat + H 2 O For more lessons, visit www. chalkbored. com
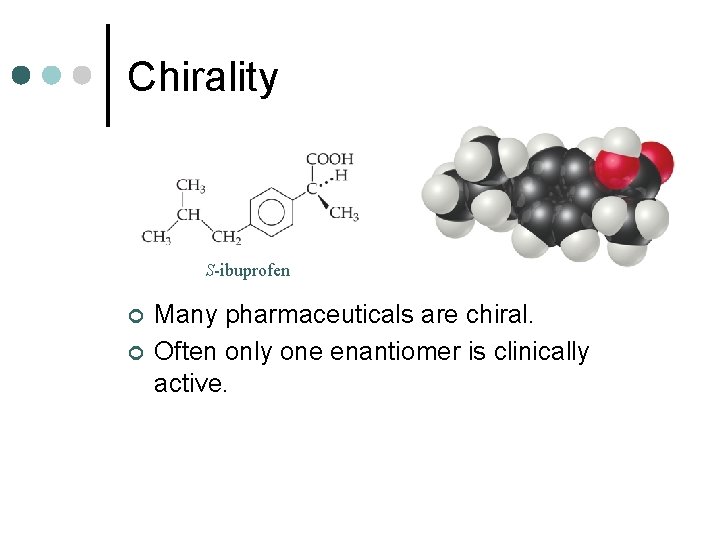
Chirality S-ibuprofen ¢ ¢ Many pharmaceuticals are chiral. Often only one enantiomer is clinically active.
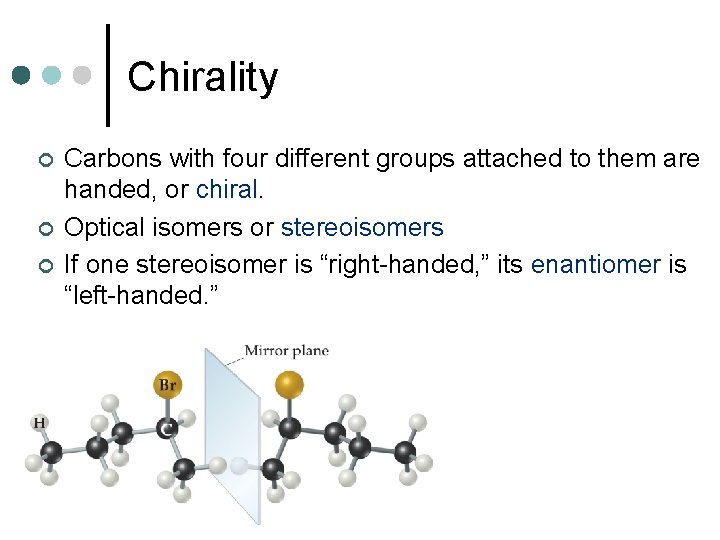
Chirality ¢ ¢ ¢ Carbons with four different groups attached to them are handed, or chiral. Optical isomers or stereoisomers If one stereoisomer is “right-handed, ” its enantiomer is “left-handed. ”
- Slides: 10