Carboxylic Acids Esters Amines and Amides Amines Reactions

Carboxylic Acids Esters, Amines and Amides Amines Reactions of Amines Amides Reactions of Amides 1
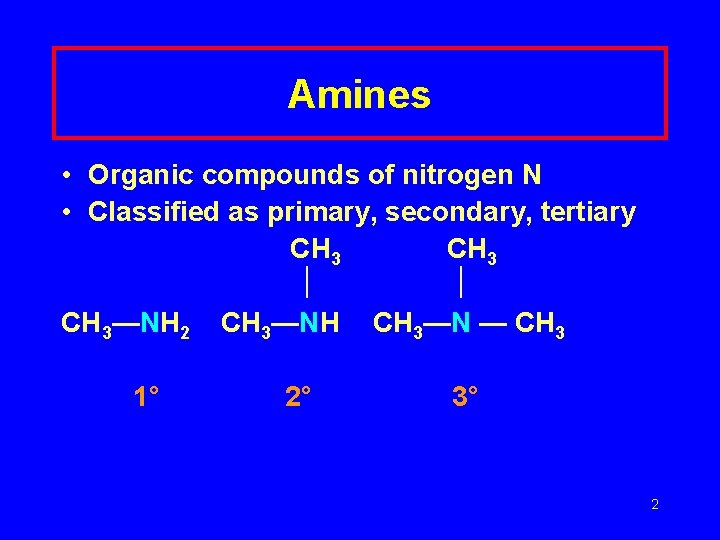
Amines • Organic compounds of nitrogen N • Classified as primary, secondary, tertiary CH 3—NH 2 CH 3—NH CH 3—N — CH 3 1° 2° 3° 2
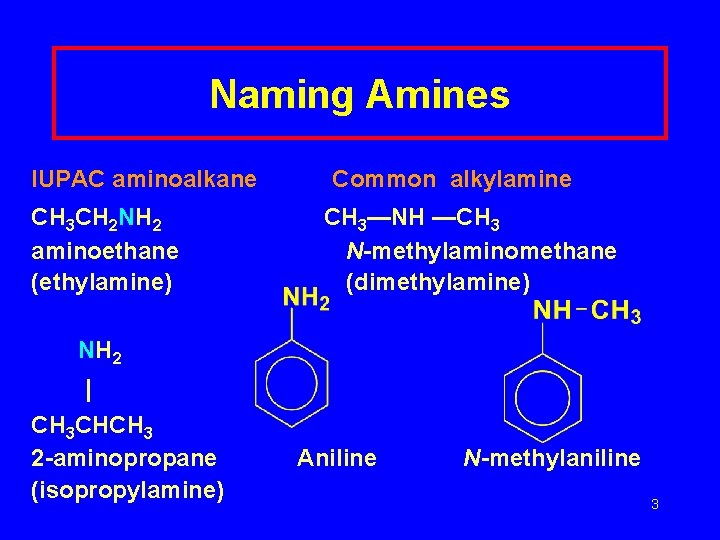
Naming Amines IUPAC aminoalkane CH 3 CH 2 NH 2 aminoethane (ethylamine) Common alkylamine CH 3—NH —CH 3 N-methylaminomethane (dimethylamine) NH 2 | CH 3 CHCH 3 2 -aminopropane (isopropylamine) Aniline N-methylaniline 3

Learning Check AM 1 Give the common name and classify and give the IUPAC name: A. CH 3 NHCH 2 CH 3 | B. CH 3 CH 2 NCH 3 4

Solution AM 1 A. CH 3 NHCH 2 CH 3 ethylmethylamine, 2°, N-methylaminoethane CH 3 | B. CH 3 CH 2 NCH 3 ethyldimethylamine, 3°, N, N-dimethylaminoethane 5
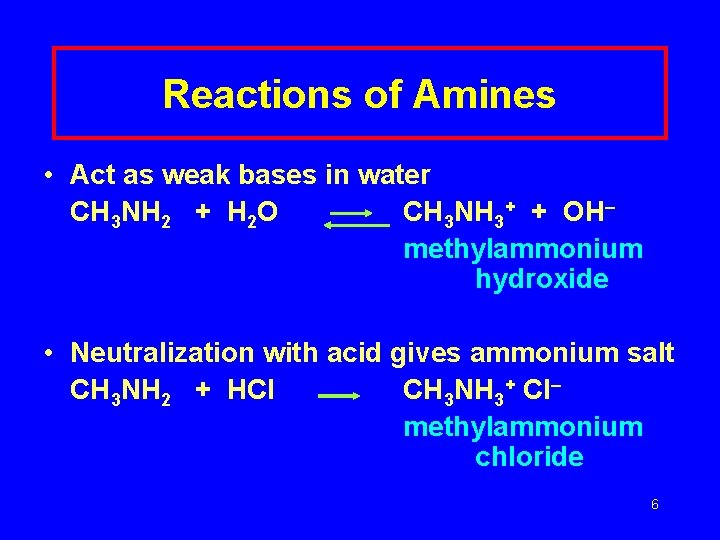
Reactions of Amines • Act as weak bases in water CH 3 NH 2 + H 2 O CH 3 NH 3+ + OH– methylammonium hydroxide • Neutralization with acid gives ammonium salt CH 3 NH 2 + HCl CH 3 NH 3+ Cl– methylammonium chloride 6

Alkaloids • Physiologically active nitrogen-containing compounds • Obtained from plants • Used as anesthetics, antidepressants, and stimulants • Many are addictive 7

Nicotine 8
Caffeine 9
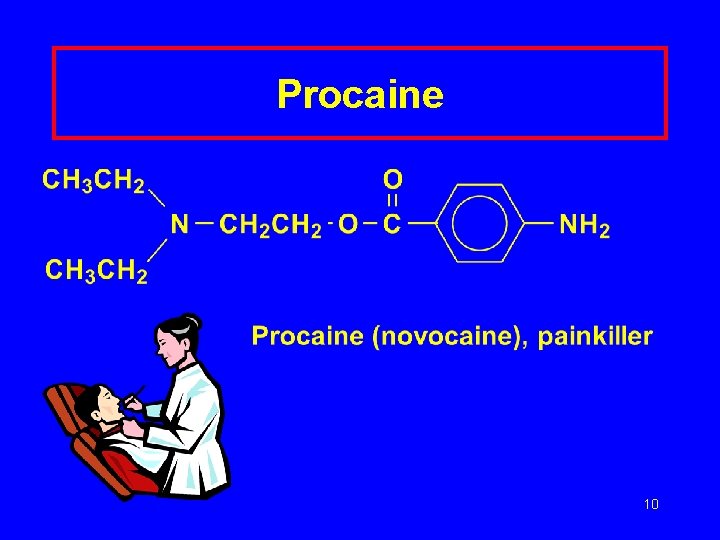
Procaine 10

Leaning Check AM 2 Write a structural formula for A. 2 -aminopentane B. 1, 3 -diaminocyclohexane 11
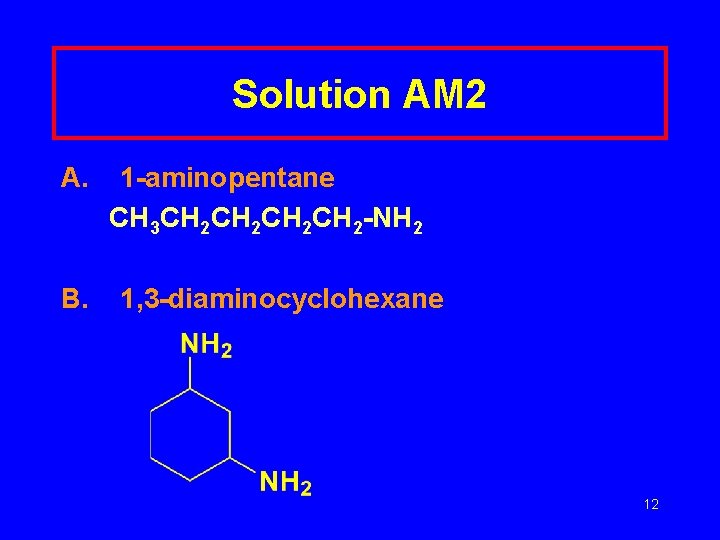
Solution AM 2 A. B. 1 -aminopentane CH 3 CH 2 CH 2 -NH 2 1, 3 -diaminocyclohexane 12
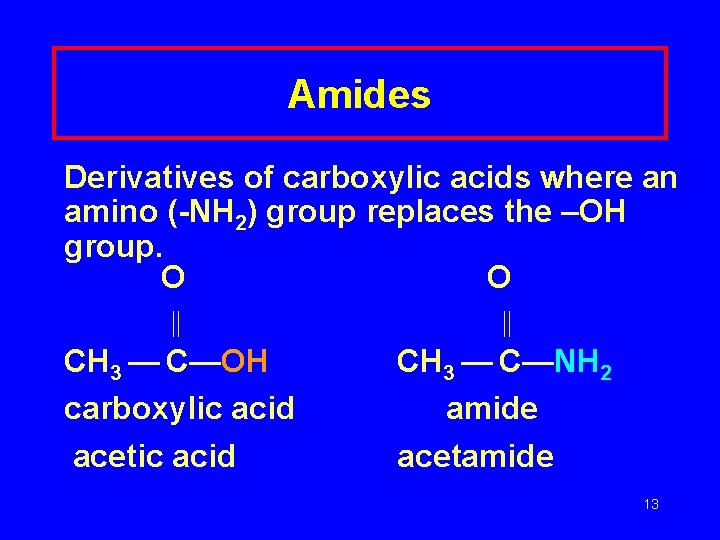
Amides Derivatives of carboxylic acids where an amino (-NH 2) group replaces the –OH group. O O CH 3 — C—OH carboxylic acid acetic acid CH 3 — C—NH 2 amide acetamide 13
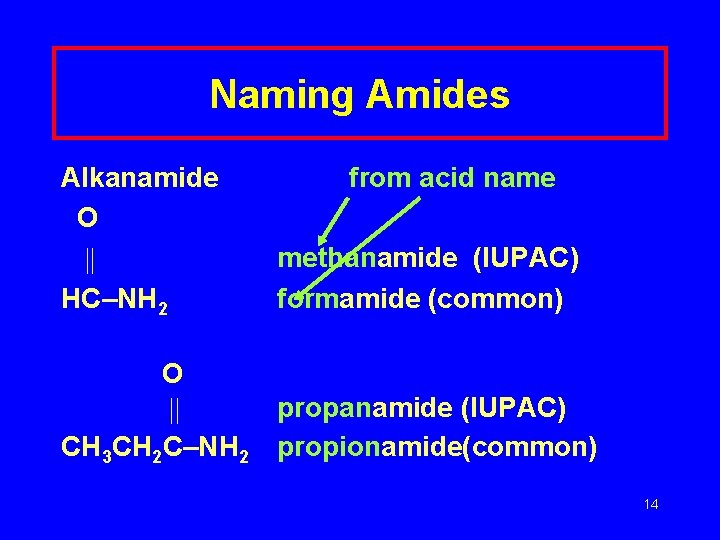
Naming Amides Alkanamide O HC–NH 2 from acid name methanamide (IUPAC) formamide (common) O CH 3 CH 2 C–NH 2 propanamide (IUPAC) propionamide(common) 14

Naming Amides with N-Groups O CH 3 C–NHCH 3 N-methylethanamide (IUPAC) N-methylacetamide (common) O CH 3 CH 2 C–N(CH 3)2 N, N-dimethylpropanamide N, N-dimethylpropionamide 15

Aromatic Amides 16

Learning Check AM 3 Name the following amides: O A. CH 3 CH 2 C–NH 2 B. O CH 3 C–N(CH 2 CH 3)2 17

Solution AM 3 O A. CH 3 CH 2 C–NH 2 butanamide; butryamide (common) O B. CH 3 C–N(CH 2 CH 3)2 N, N-diethylethanamide; N, N-diethylacetamide 18

Learning Check AM 4 Draw the structures of A. Pentanamide B. N-methylbutanamide 19
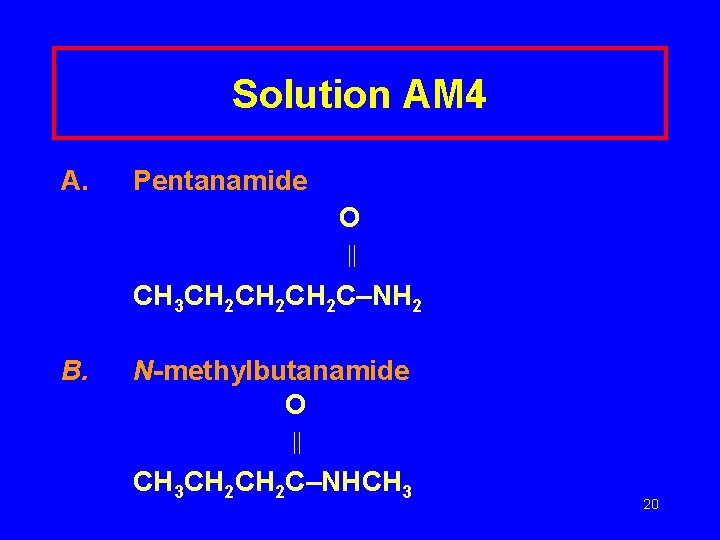
Solution AM 4 A. Pentanamide O CH 3 CH 2 CH 2 C–NH 2 B. N-methylbutanamide O CH 3 CH 2 C–NHCH 3 20

Reactions of Amides undergo acid hydrolysis base hydrolysis carboxylic acid ammonium salt of carboxylic acid an amine or ammonia 21
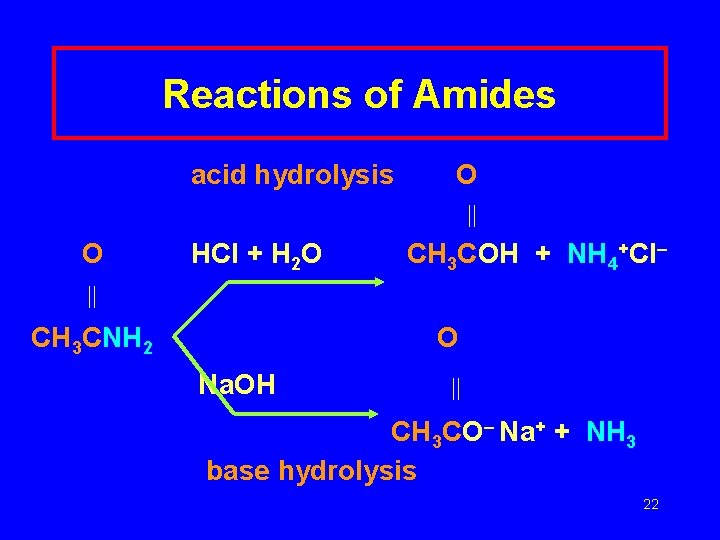
Reactions of Amides acid hydrolysis O O HCl + H 2 O CH 3 COH + NH 4+Cl– CH 3 CNH 2 O Na. OH CH 3 CO– Na+ + NH 3 base hydrolysis 22

Learning Check AM 5 Write the products of the hydrolysis of N- ethylpropanamide with Na. OH. 23

Solution AM 5 Hydrolysis of N-ethylpropanamide with Na. OH gives the following products. O CH 3 CH 2 CO– Na+ + CH 3 CH 2 NH 2 24
- Slides: 24