Carboxylic Acids Carboxyl Group Carboxylic acids contain the
Carboxylic Acids
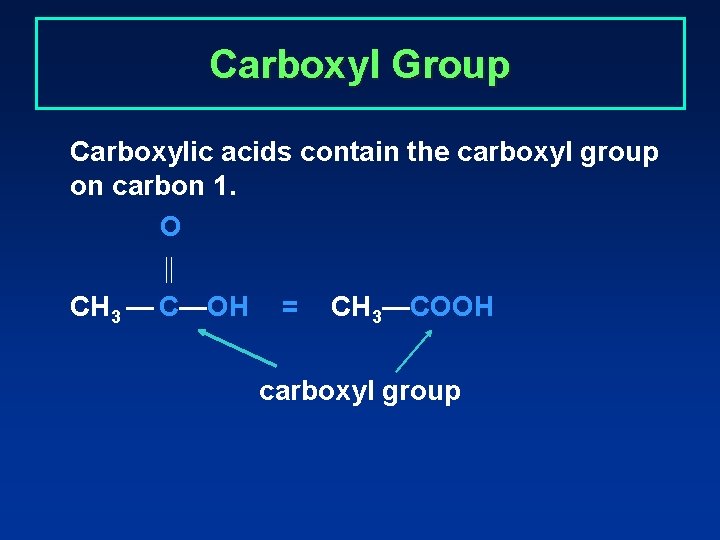
Carboxyl Group Carboxylic acids contain the carboxyl group on carbon 1. O CH 3 — C—OH = CH 3—COOH carboxyl group

Naming Carboxylic Acids Formula IUPAC alkan -oic acid Common prefix – ic acid HCOOH methanoic acid formic acid CH 3 COOH ethanoic acid acetic acid CH 3 CH 2 COOH propanoic acid propionic acid CH 3 CH 2 COOH butanoic acid butyric acid

Carboxylic Acids l Abundant and widely distributed in nature, many having a Greek or Latin word describing their origin – acetic acid (ethanoic acid) from acetum, meaning vinegar – many that were isolated from fats are called fatty acids l Examples on page 249

Naming Rules • Identify longest chain • (IUPAC) Number carboxyl carbon as 1 • (Common) Assign , , to carbon atoms adjacent to carboxyl carbon CH 3 | CH 3 — CH—CH 2 —COOH IUPAC 3 -methylbutanoic acid Common -methylbutryic acid

Learning Check Give IUPAC and common names: A. CH 3 COOH CH 3 | B. CH 3 CHCOOH

Solution A. CH 3 COOH ethanoic acid; acetic acid CH 3 | B. CH 3 CHCOOH 2 -methylpropanoic acid; -methylpropionic acid
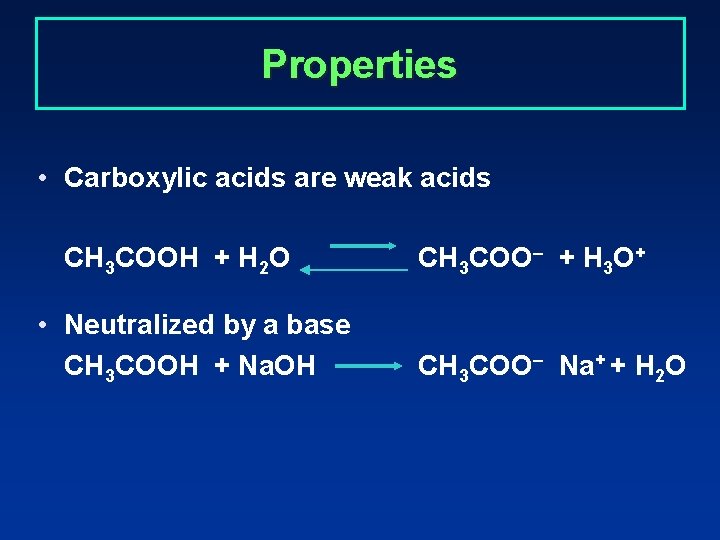
Properties • Carboxylic acids are weak acids CH 3 COOH + H 2 O • Neutralized by a base CH 3 COOH + Na. OH CH 3 COO– + H 3 O+ CH 3 COO– Na+ + H 2 O
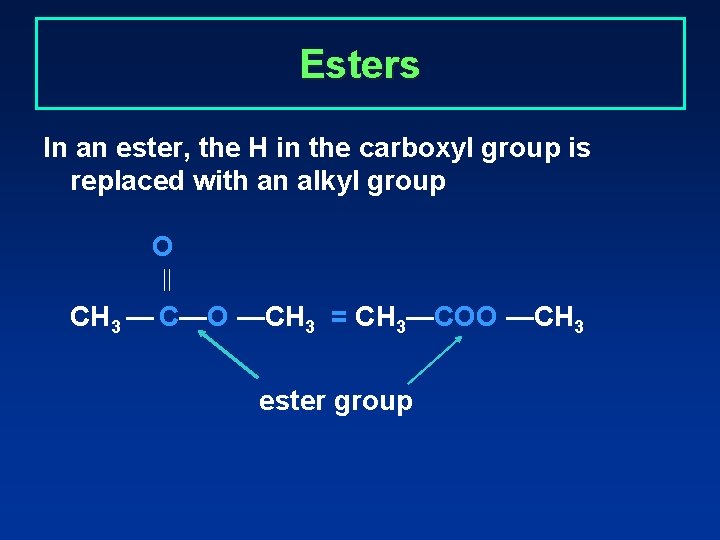
Esters In an ester, the H in the carboxyl group is replaced with an alkyl group O CH 3 — C—O —CH 3 = CH 3—COO —CH 3 ester group

Esters l Although polar, they do not form hydrogen bonds (reason: there is no hydrogen bonded to a highly electronegative atom!) – thus, much lower b. p. than the hydrogen-bonded carboxylic acids they came from

Esters in Plants Esters give flowers and fruits their pleasant fragances and flavors.

Esters l Can be prepared from a carboxylic acid an alcohol; usually a trace of mineral acid added as catalyst (because acids are dehydrating agents) l Note equation on bottom p. 345

Esterification • Reaction of a carboxylic acid and alcohol • Acid catalyst O H+ CH 3 — C—OH + HO—CH 2 CH 3 O CH 3 — C—O—CH 2 CH 3 + H 2 O

Esters l Naming? It has 2 words: – 1 st: alkyl attached to single bonded oxygen from alcohol – 2 nd: take the acid name, remove the -ic acid, add -ate l example on bottom p. 345
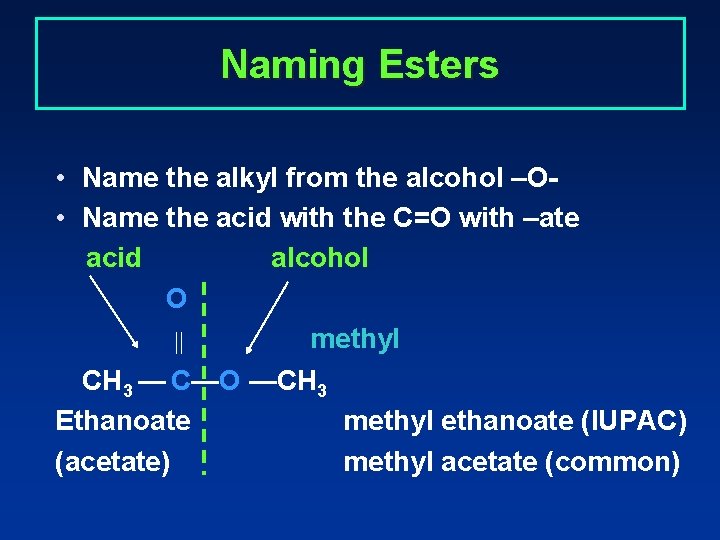
Naming Esters • Name the alkyl from the alcohol –O • Name the acid with the C=O with –ate acid alcohol O methyl CH 3 — C—O —CH 3 Ethanoate methyl ethanoate (IUPAC) (acetate) methyl acetate (common)

Some Esters and Their Names Flavor/Odor Raspberries HCOOCH 2 CH 3 ethyl methanoate (IUPAC) ethyl formate (common) Pineapples CH 3 CH 2 COOCH 2 CH 3 ethyl butanoate (IUPAC) ethyl butyrate (common)
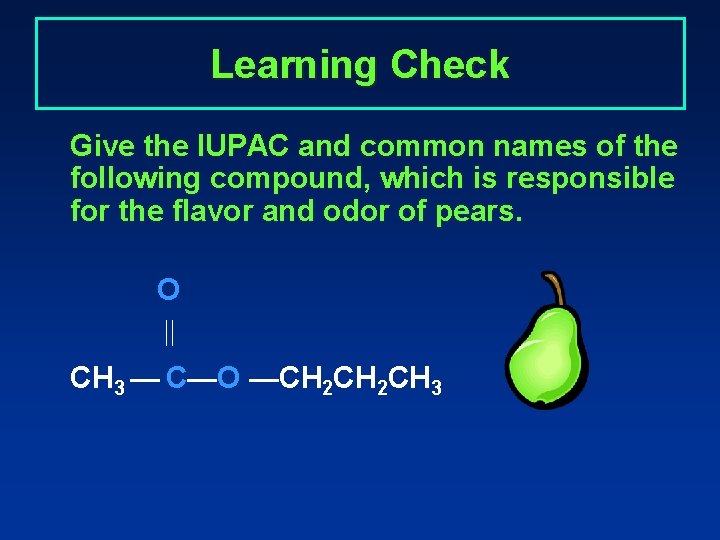
Learning Check Give the IUPAC and common names of the following compound, which is responsible for the flavor and odor of pears. O CH 3 — C—O —CH 2 CH 3
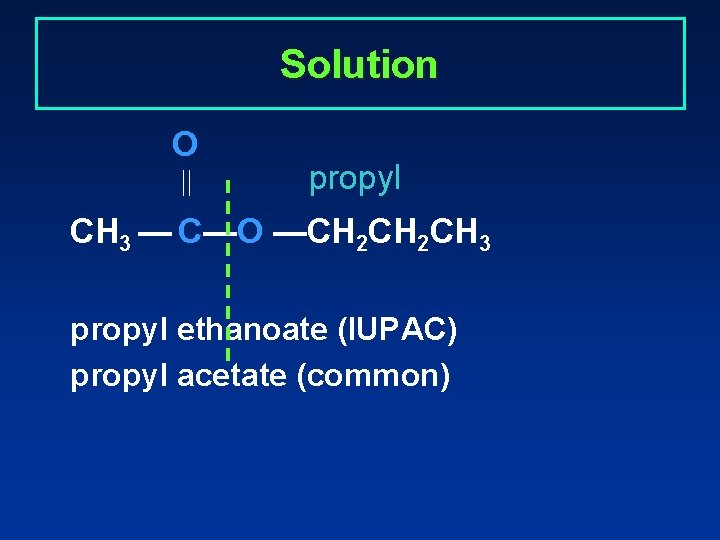
Solution O propyl CH 3 — C—O —CH 2 CH 3 propyl ethanoate (IUPAC) propyl acetate (common)

Learning Check Draw the structure of the following compounds: A. 3 -bromobutanoic acid B. Ethyl propionoate

Solution A. 3 -bromobutanoic acid Br | CH 3 CHCH 2 COOH B. Ethyl propionoate O CH 3 CH 2 COCH 2 CH 3 CH 2 COOCH 2 CH 3
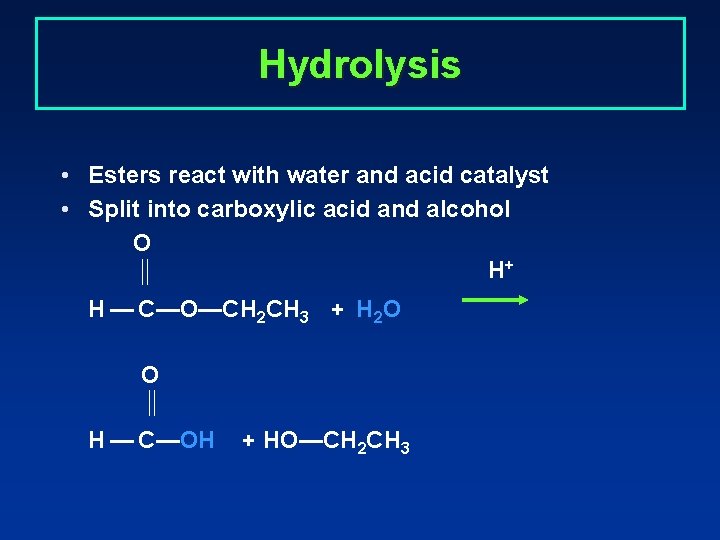
Hydrolysis • Esters react with water and acid catalyst • Split into carboxylic acid and alcohol O H+ H — C—O—CH 2 CH 3 + H 2 O O H — C—OH + HO—CH 2 CH 3

Saponification • Esters react with a bases • Produce the salt of the carboxylic acid and alcohol O CH 3 C—OCH 2 CH 3 + Na. OH O CH 3 C—O– Na+ salt of carboxylic acid + HOCH 2 CH 3

Learning Check Write the equation for the reaction of propionic acid and methyl alcohol in the presence of an acid catalyst.
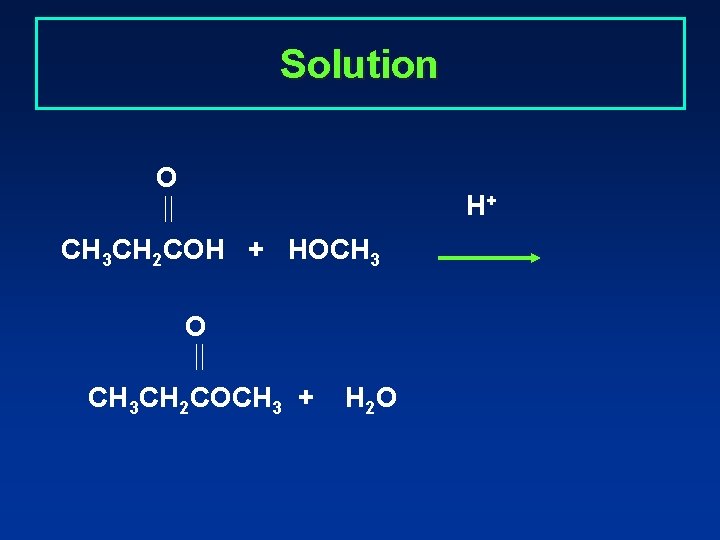
Solution O H+ CH 3 CH 2 COH + HOCH 3 O CH 3 CH 2 COCH 3 + H 2 O

Learning Check What are the organic products when methylacetate reacts with A. Water and an acid catalyst? B. KOH?

Solution A. O CH 3 COH + HOCH 3 O B. CH 3 CO– K+ + HOCH 3
- Slides: 26