CARBOXYLIC ACIDS C 2 7 Alcohols Carboxylic Acids

CARBOXYLIC ACIDS C 2. 7 Alcohols & Carboxylic Acids
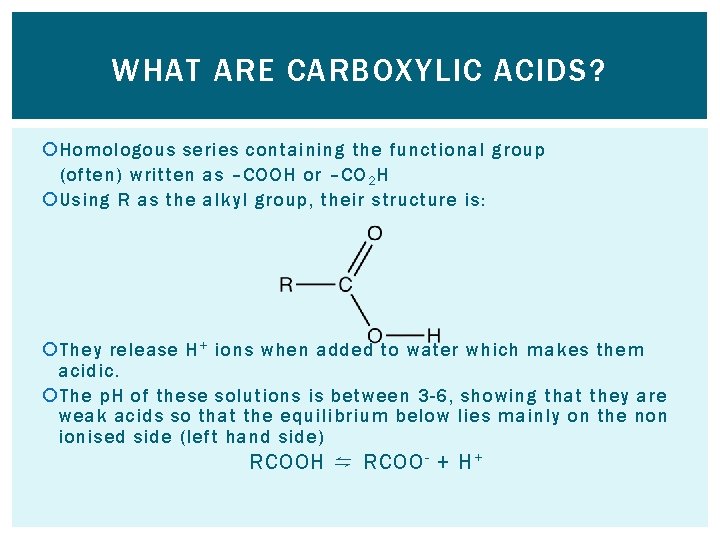
WHAT ARE CARBOXYLIC ACIDS? Homologous series containing the functional group (often) written as –COOH or –CO 2 H Using R as the alkyl group, their structure is: They release H + ions when added to water which makes them acidic. The p. H of these solutions is between 3 -6, showing that they are weak acids so that the equilibrium below lies mainly on the non ionised side (left hand side) RCOOH ⇋ RCOO - + H +

REACTIONS OF CARBOXYLIC ACIDS – AS AN ACID q General equations: Carboxylic acid + Base Salt + Water Carboxylic acid + Carbonate Salt + carbon dioxide + water Carboxylic acid + Hydrogen Carbonate Salt + carbon dioxide + water q Both soluble bases and solid bases neutralise carboxylic acids q Both reactions would show a change in p. H q The carbonate reactions could be tested by adding limewater (turns milky with CO 2 ) q You need to be able to write word and symbol equations for each of these reaction along with state symbols!

REACTIONS OF CARBOXYLIC ACIDS – AS AN ACID q General equations: Carboxylic acid + Base Salt + Water Carboxylic acid + Carbonate Salt + carbon dioxide + water Carboxylic acid + Hydrogen Carbonate Salt + carbon dioxide + water Word equation examples: e. g. Ethanoic acid + Sodium hydroxide Sodium ethanoate + water e. g. Propanoic acid + Calcium carbonate Calcium ethanoate + carbon dioxide + water e. g. Methanoic acid + Copper oxide Copper methanoate + water e. g. Propanoic acid + Zinc carbonate Zinc propanoate + water

REACTIONS OF CARBOXYLIC ACIDS – AS AN ACID q General equations: Carboxylic acid + Base Salt + Water Carboxylic acid + Carbonate Salt + carbon dioxide + water Carboxylic acid + Hydrogen Carbonate Salt + carbon dioxide + water Symbol equation examples: e. g. Ethanoic acid + Sodium hydroxide Sodium ethanoate + water CH 3 COOH(aq) + Na. OH(aq) CH 3 COONa(aq) + H 2 O(l) e. g. Propanoic acid + Calcium carbonate Calcium ethanoate + carbon dioxide + water CH 3 CH 2 COOH(aq) + Na. OH(aq) CH 3 CH 2 COONa(aq) + CO 2(g) + H 2 O(l) e. g. Methanoic acid + Copper oxide Copper methanoate + water 2 HCOOH(aq) + Ca. O(s) (HCOO)2 Ca(aq) + CO 2(g) + H 2 O(l) e. g. Propanoic acid + Zinc carbonate Zinc propanoate + water CH 3 CH 2 COOH(aq) + Zn. CO 3(aq) (CH 3 CH 2 COO)2 Zn(aq) + CO 2(g) + H 2 O(l)

KNOWLEDGE CHECK 1. Write a word equation for the reaction between: a) Calcium carbonate (Ca. CO 3 ) and Ethanoic acid b) Potassium hydrogen carbonate (KHCO 3 ) and methanoic acid c) Propanoic acid and Sodium Hydroxide d) Pentanoic acid and Sodium Hydroxide 2. Write symbol equations for each of the above reactions including state symbols.
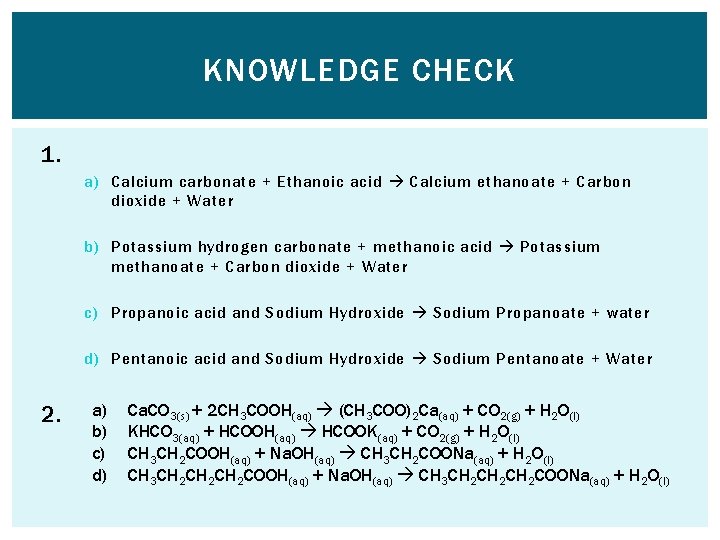
KNOWLEDGE CHECK 1. a) Calcium carbonate + Ethanoic acid Calcium ethanoate + Carbon dioxide + Water b) Potassium hydrogen carbonate + methanoic acid Potassium methanoate + Carbon dioxide + Water c) Propanoic acid and Sodium Hydroxide Sodium Propanoate + water d) Pentanoic acid and Sodium Hydroxide Sodium Pentanoate + Water 2. a) b) c) d) Ca. CO 3(s) + 2 CH 3 COOH(aq) (CH 3 COO)2 Ca(aq) + CO 2(g) + H 2 O(l) KHCO 3(aq) + HCOOH(aq) HCOOK(aq) + CO 2(g) + H 2 O(l) CH 3 CH 2 COOH(aq) + Na. OH(aq) CH 3 CH 2 COONa(aq) + H 2 O(l) CH 3 CH 2 CH 2 COOH(aq) + Na. OH(aq) CH 3 CH 2 CH 2 COONa(aq) + H 2 O(l)

REACTIONS OF CARBOXYLIC ACIDS – ESTERIFICATION q General equations: Carboxy lic acid + alcohol ⇋ Ester + Water q Conditions: Heat and concentrated sulfuric acid catalyst q You need to be able to write word, symbol and displayed equations for this reaction. e. g. Ethanoic acid + Ethanol Ethyl ethanoate + Water CH 3 COOH + C 2 H 5 OH ⇋ CH 3 COOC 2 H 5 + H 2 O Worked examples: Ethanoic acid + Methanol Propanoic acid + Ethanol
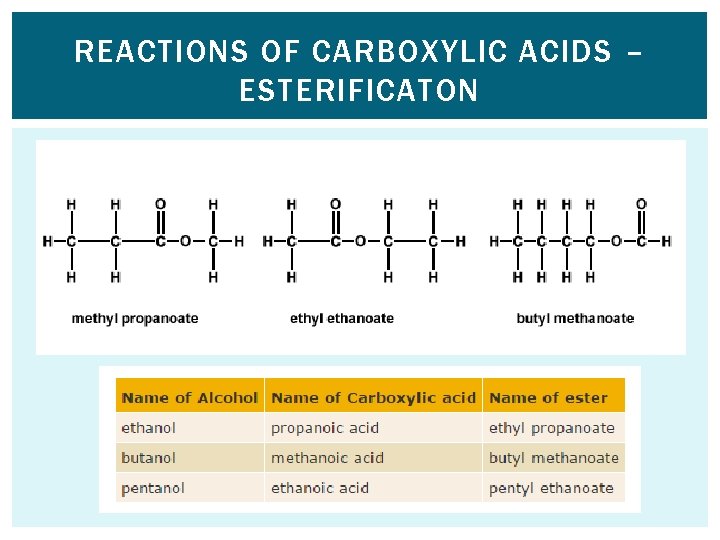
REACTIONS OF CARBOXYLIC ACIDS – ESTERIFICATON

REACTIONS OF CARBOXYLIC ACIDS – ESTERIFICATON The ester can be separated from the reaction mixture by distillation because it will have a different boiling point.

KNOWLEDGE CHECK 1. Write an equation to show the reaction that occurs when 2 -chloro-3 -methylbutanoic acid, CH 3 CH(CH 3 )CHCl. COOH, reacts with butan-2 -ol. 2. Concentrated sulphuric acid is added to the reaction mixture. What is the purpose of the sulphuric acid? 3. Why can the ester be distilled off, leaving the other organic substances in the reaction flask?

ANSWERS 1. a) b) Sulfuric acid acts as a catalyst 2. The ester has the lowest boiling point. Alcohols, carboxylic acids and water all contain the –OH group and can hydrogen bond. This increases their boiling point. Esters cannot hydrogen bond.
- Slides: 12