Carboxylic Acids And Their Derivatives 1435 1436 2014

Carboxylic Acids And Their Derivatives 1435 -1436 2014 -2015 1

Carboxylic Acids And Their Derivatives Chapter nine introduces carboxylic acids and their derivatives. The chemistry is very similar to that of aldehydes and ketones because of the presence of the carbonyl group. The main topics in this chapter that the students should know and understand include: ØThe structure of carboxylic acids ØThe common and IUPAC nomenclature of carboxylic acids ØThe physical properties of carboxylic acids ØThe Factors affecting acidity of carboxylic acids. ØThe different ways to make carboxylic acids ØSalt formation reactions of carboxylic acids ØThe nucleophilic substitution reactions at the carbonyl carbon and the specific products formed in each case. Ø The chemistry of carboxylic acid derivatives 2

Carboxylic Acids And Their Derivatives Structure Of Carboxylic Acids Ø Carboxylic acids are organic acids contain one or more carboxyl group, which is a combination of carbonyl group C=O and hydroxyl group O-H Ø It is often written in condensed form as –CO 2 H or –COOH Carboxylic acids are classified as aliphatic R-COOH or aromatic Ar -COOH depending on the group bonded to the carboxylic group. Ø The simplest acid is formic acid R= H Ø The fatty acid is long chain aliphatic acids CH 3 -(CH 2)16 -COOH 3
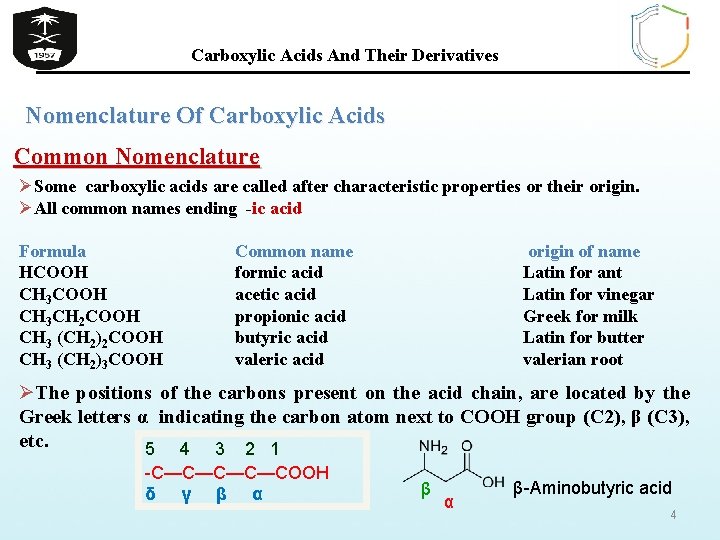
Carboxylic Acids And Their Derivatives Nomenclature Of Carboxylic Acids Common Nomenclature ØSome carboxylic acids are called after characteristic properties or their origin. ØAll common names ending -ic acid Formula HCOOH CH 3 CH 2 COOH CH 3 (CH 2)3 COOH Common name formic acid acetic acid propionic acid butyric acid valeric acid origin of name Latin for ant Latin for vinegar Greek for milk Latin for butter valerian root ØThe positions of the carbons present on the acid chain, are located by the Greek letters α indicating the carbon atom next to COOH group (C 2), β (C 3), etc. 5 4 3 2 1 -C—C—COOH δ γ β α β-Aminobutyric acid 4
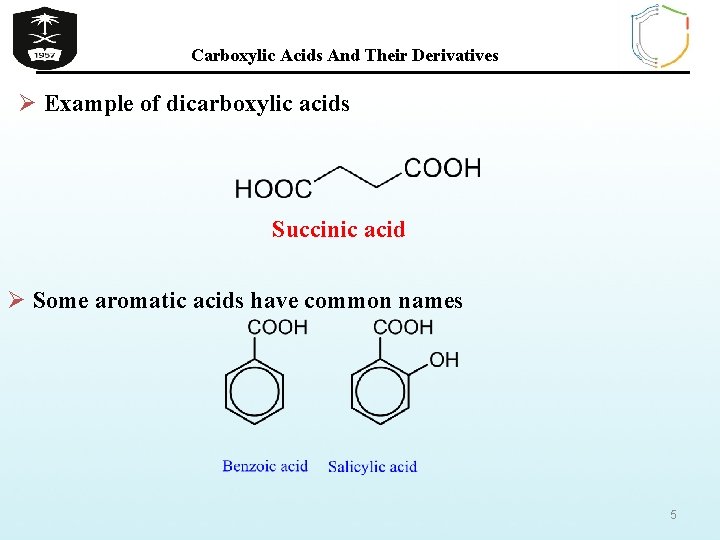
Carboxylic Acids And Their Derivatives Ø Example of dicarboxylic acids Succinic acid Ø Some aromatic acids have common names 5
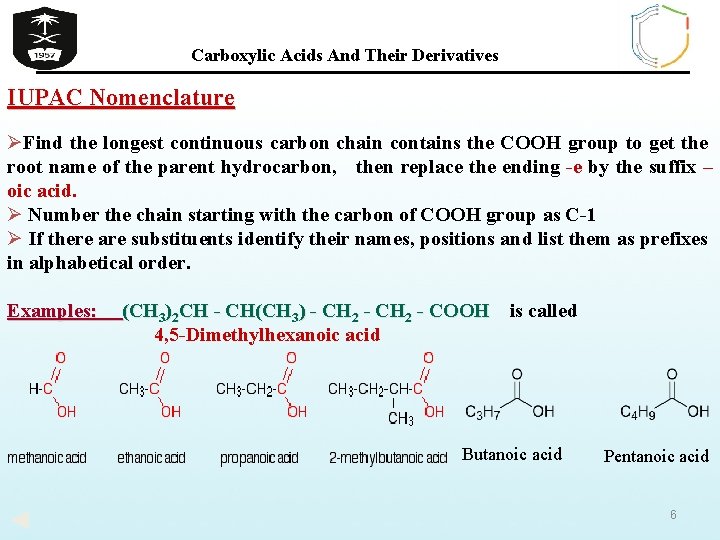
Carboxylic Acids And Their Derivatives IUPAC Nomenclature ØFind the longest continuous carbon chain contains the COOH group to get the root name of the parent hydrocarbon, then replace the ending -e by the suffix – oic acid. Ø Number the chain starting with the carbon of COOH group as C-1 Ø If there are substituents identify their names, positions and list them as prefixes in alphabetical order. Examples: (CH 3)2 CH - CH(CH 3) - CH 2 - COOH is called 4, 5 -Dimethylhexanoic acid Butanoic acid Pentanoic acid 6
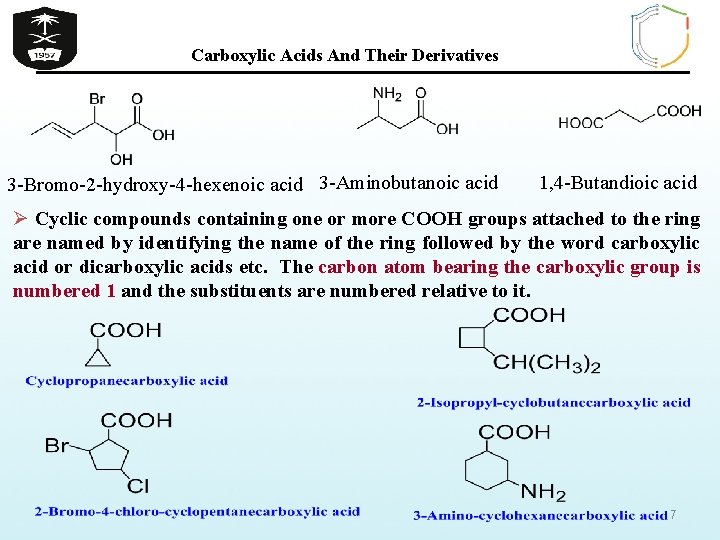
Carboxylic Acids And Their Derivatives 3 -Bromo-2 -hydroxy-4 -hexenoic acid 3 -Aminobutanoic acid 1, 4 -Butandioic acid Ø Cyclic compounds containing one or more COOH groups attached to the ring are named by identifying the name of the ring followed by the word carboxylic acid or dicarboxylic acids etc. The carbon atom bearing the carboxylic group is numbered 1 and the substituents are numbered relative to it. 7
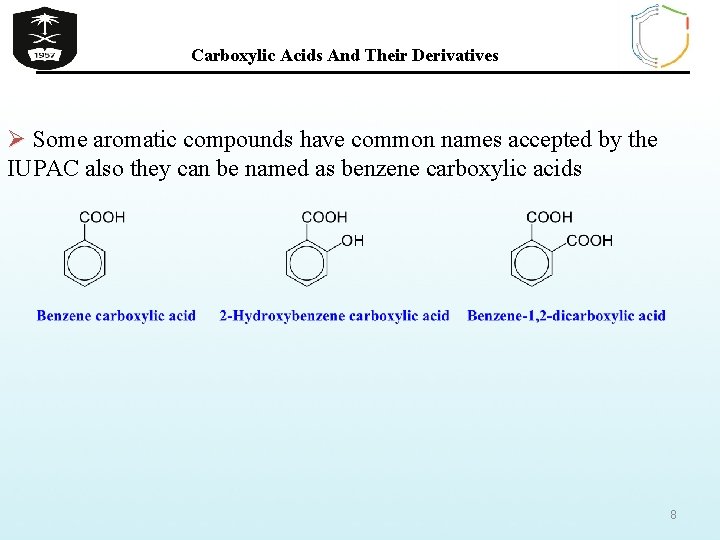
Carboxylic Acids And Their Derivatives Ø Some aromatic compounds have common names accepted by the IUPAC also they can be named as benzene carboxylic acids 8
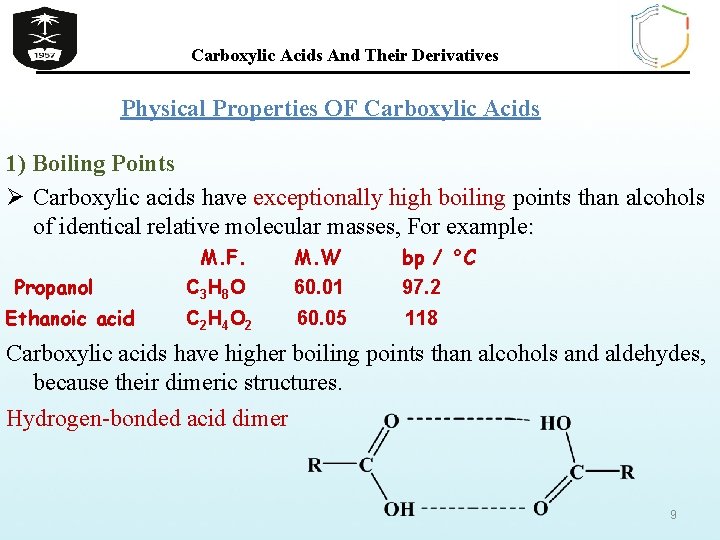
Carboxylic Acids And Their Derivatives Physical Properties OF Carboxylic Acids 1) Boiling Points Ø Carboxylic acids have exceptionally high boiling points than alcohols of identical relative molecular masses, For example: Propanol Ethanoic acid M. F. M. W bp / °C C 3 H 8 O 60. 01 97. 2 C 2 H 4 O 2 60. 05 118 Carboxylic acids have higher boiling points than alcohols and aldehydes, because their dimeric structures. Hydrogen-bonded acid dimer 9
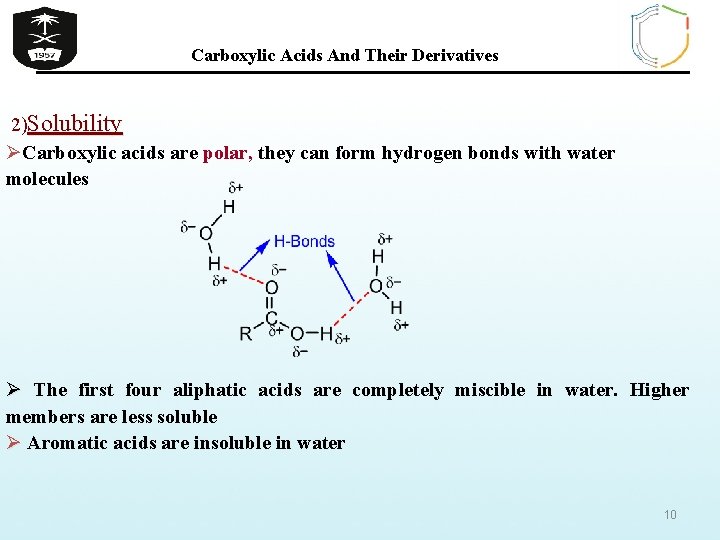
Carboxylic Acids And Their Derivatives 2)Solubility ØCarboxylic acids are polar, they can form hydrogen bonds with water molecules Ø The first four aliphatic acids are completely miscible in water. Higher members are less soluble Ø Aromatic acids are insoluble in water 10
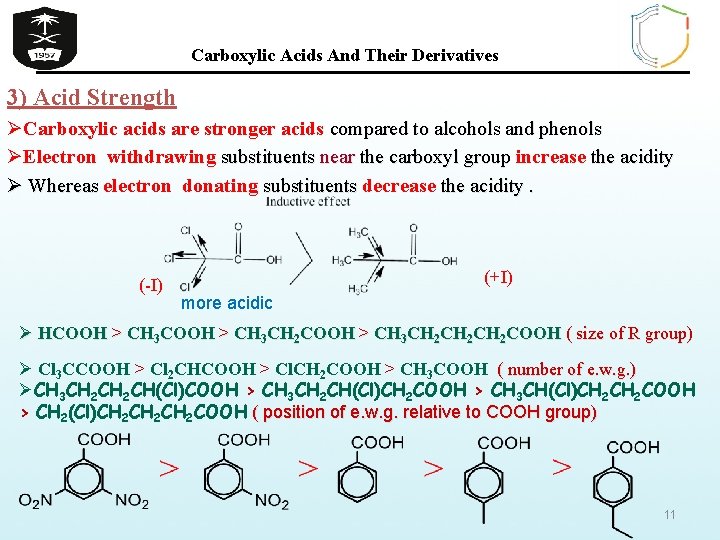
Carboxylic Acids And Their Derivatives 3) Acid Strength ØCarboxylic acids are stronger acids compared to alcohols and phenols ØElectron withdrawing substituents near the carboxyl group increase the acidity Ø Whereas electron donating substituents decrease the acidity. (-I) (+I) more acidic Ø HCOOH > CH 3 CH 2 COOH > CH 3 CH 2 CH 2 COOH ( size of R group) Ø Cl 3 CCOOH > Cl 2 CHCOOH > Cl. CH 2 COOH > CH 3 COOH ( number of e. w. g. ) ØCH 3 CH 2 CH(Cl)COOH > CH 3 CH 2 CH(Cl)CH 2 COOH > CH 3 CH(Cl)CH 2 COOH > CH 2(Cl)CH 2 CH 2 COOH ( position of e. w. g. relative to COOH group) 11
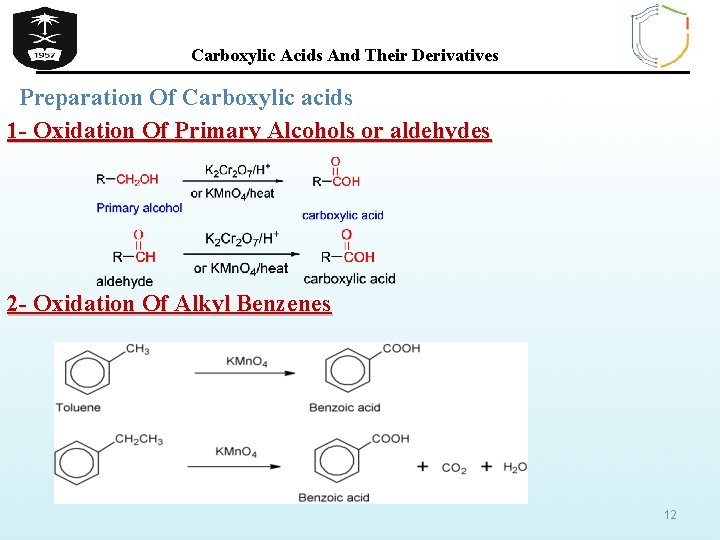
Carboxylic Acids And Their Derivatives Preparation Of Carboxylic acids 1 - Oxidation Of Primary Alcohols or aldehydes 2 - Oxidation Of Alkyl Benzenes 12
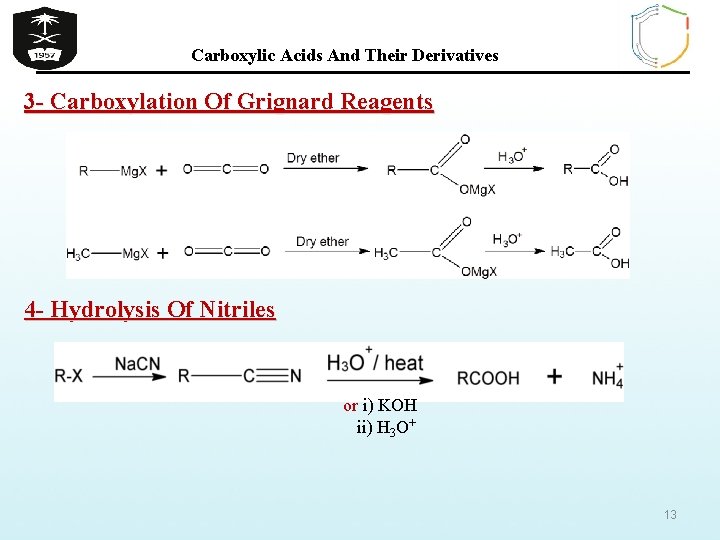
Carboxylic Acids And Their Derivatives 3 - Carboxylation Of Grignard Reagents 4 - Hydrolysis Of Nitriles or i) KOH ii) H 3 O+ 13
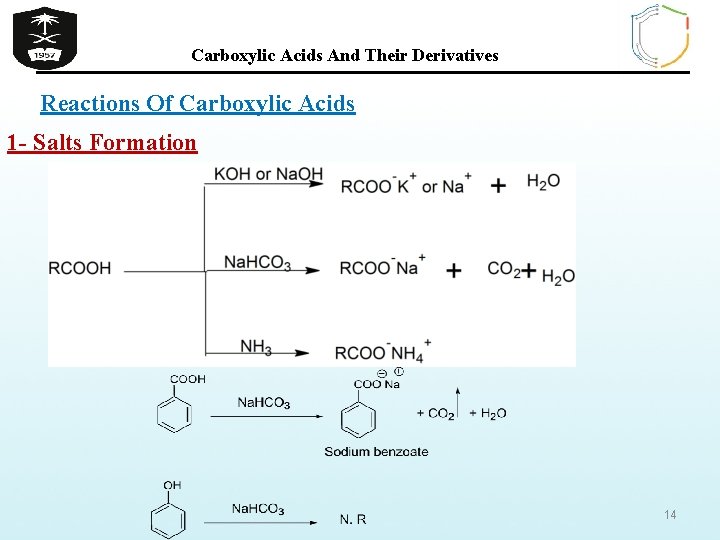
Carboxylic Acids And Their Derivatives Reactions Of Carboxylic Acids 1 - Salts Formation 14
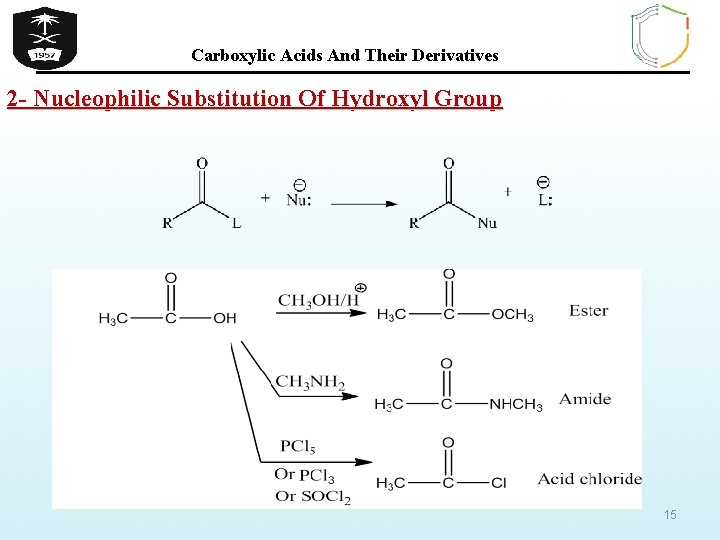
Carboxylic Acids And Their Derivatives 2 - Nucleophilic Substitution Of Hydroxyl Group 15
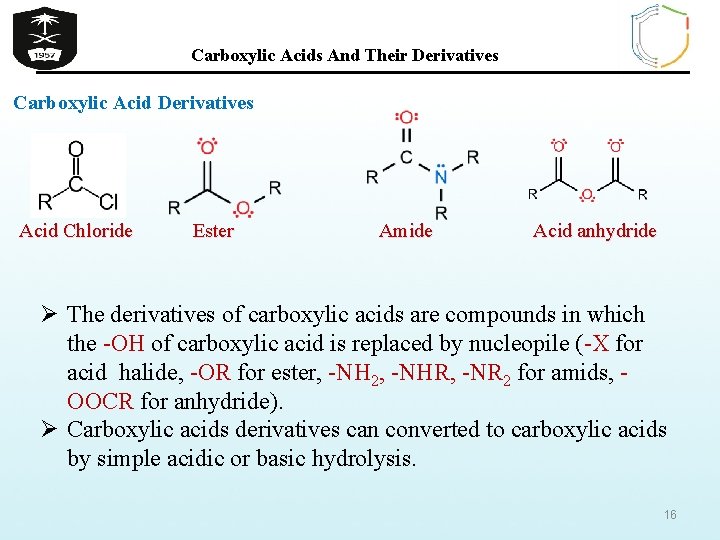
Carboxylic Acids And Their Derivatives Carboxylic Acid Derivatives Acid Chloride Ester Amide Acid anhydride Ø The derivatives of carboxylic acids are compounds in which the -OH of carboxylic acid is replaced by nucleopile (-X for acid halide, -OR for ester, -NH 2, -NHR, -NR 2 for amids, OOCR for anhydride). Ø Carboxylic acids derivatives can converted to carboxylic acids by simple acidic or basic hydrolysis. 16
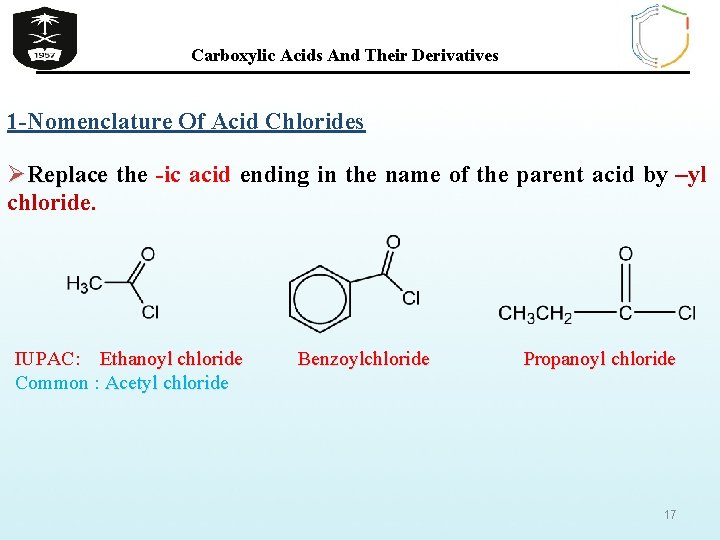
Carboxylic Acids And Their Derivatives 1 -Nomenclature Of Acid Chlorides ØReplace the -ic acid ending in the name of the parent acid by –yl chloride. IUPAC: Ethanoyl chloride Common : Acetyl chloride Benzoylchloride Propanoyl chloride 17
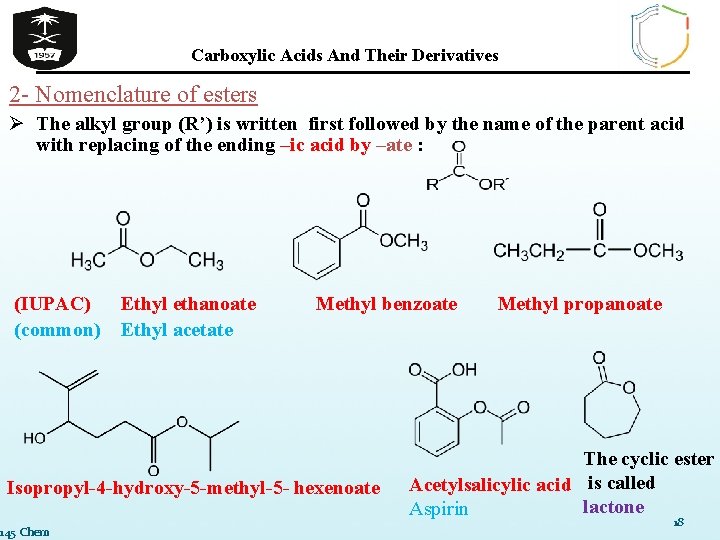
Carboxylic Acids And Their Derivatives 2 - Nomenclature of esters Ø The alkyl group (R’) is written first followed by the name of the parent acid with replacing of the ending –ic acid by –ate : (IUPAC) Ethyl ethanoate (common) Ethyl acetate Methyl benzoate Isopropyl-4 -hydroxy-5 -methyl-5 - hexenoate 145 Chem Methyl propanoate The cyclic ester Acetylsalicylic acid is called lactone Aspirin 18
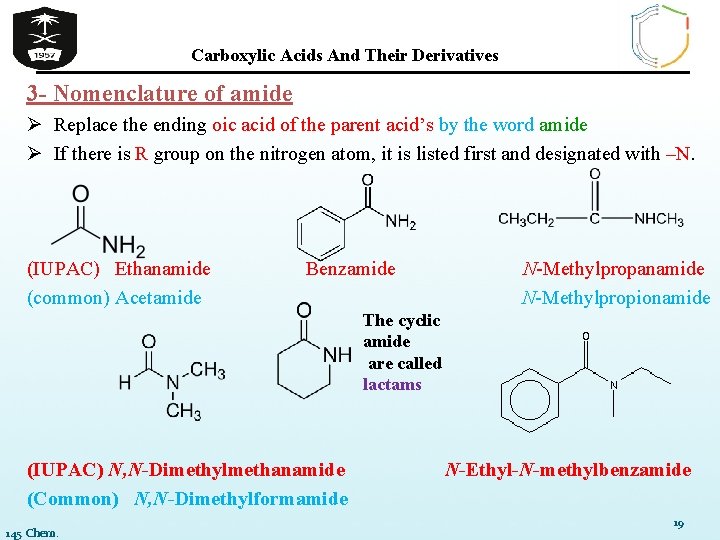
Carboxylic Acids And Their Derivatives 3 - Nomenclature of amide Ø Replace the ending oic acid of the parent acid’s by the word amide Ø If there is R group on the nitrogen atom, it is listed first and designated with –N. (IUPAC) Ethanamide (common) Acetamide Benzamide N-Methylpropanamide N-Methylpropionamide The cyclic amide are called lactams (IUPAC) N, N-Dimethylmethanamide (Common) N, N-Dimethylformamide 145 Chem. N-Ethyl-N-methylbenzamide 19
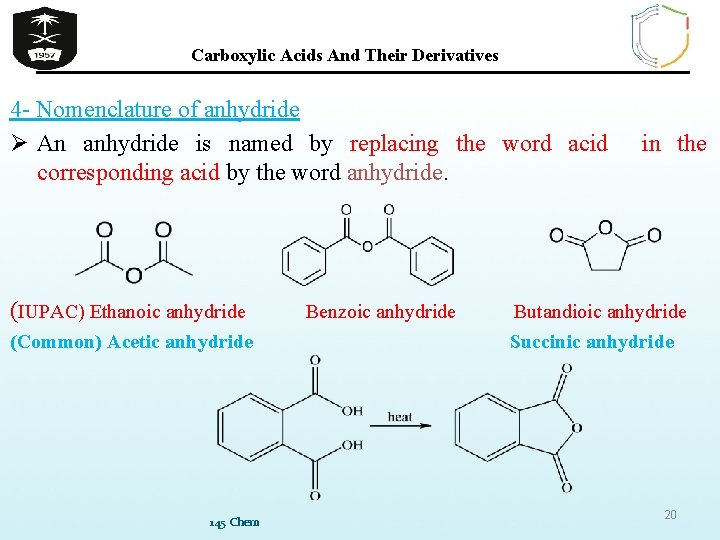
Carboxylic Acids And Their Derivatives 4 - Nomenclature of anhydride Ø An anhydride is named by replacing the word acid corresponding acid by the word anhydride. (IUPAC) Ethanoic anhydride (Common) Acetic anhydride 145 Chem Benzoic anhydride in the Butandioic anhydride Succinic anhydride 20
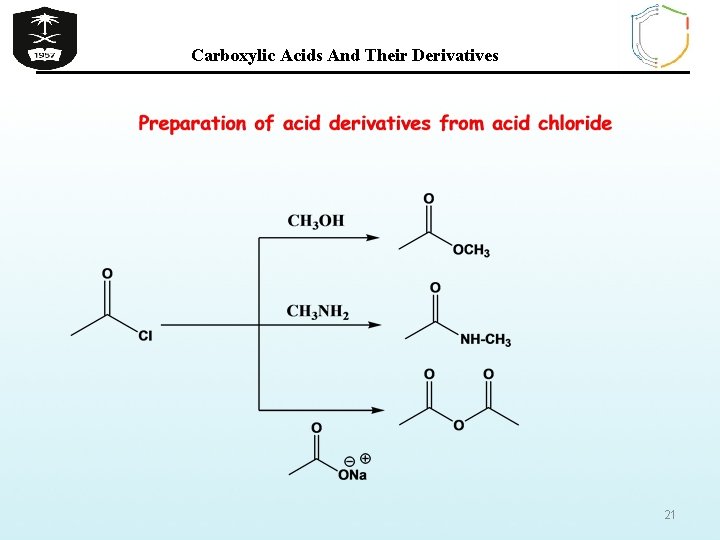
Carboxylic Acids And Their Derivatives 21
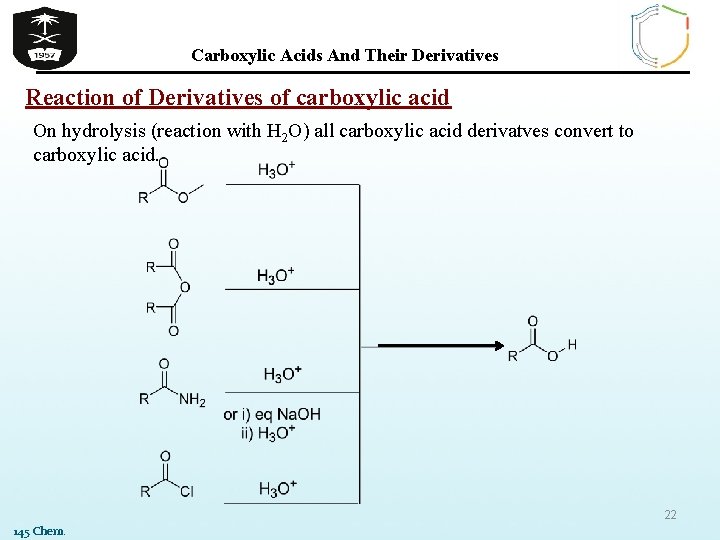
Carboxylic Acids And Their Derivatives Reaction of Derivatives of carboxylic acid On hydrolysis (reaction with H 2 O) all carboxylic acid derivatves convert to carboxylic acid. 22 145 Chem.
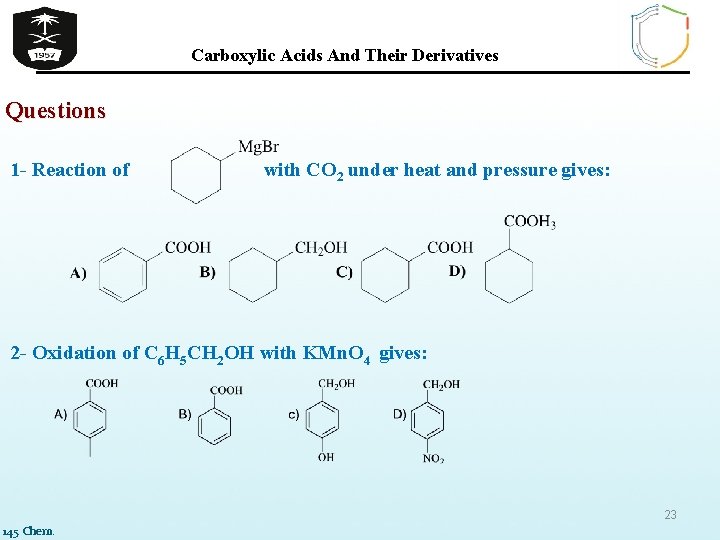
Carboxylic Acids And Their Derivatives Questions 1 - Reaction of with CO 2 under heat and pressure gives: 2 - Oxidation of C 6 H 5 CH 2 OH with KMn. O 4 gives: 23 145 Chem.

Carboxylic Acids And Their Derivatives 3 - The most acidic compound is? 4 - The common name of this compound A) N, N-Dimethylacetamide C) N, N-Dimethylpropionamide is? B) N, N-Dimethyl propanamide D) N, N-Dimethylbutanamide 5 - Acid halide react with ammonia to give? A) Amines B)Amides 145 Chem C) Phenols D) Alcohols 24

Carboxylic Acids And Their Derivatives Thank You for your kind attention ! Questions? Comments 25
- Slides: 25