Carboplatin and Creatinine Have we got it right

Carboplatin and Creatinine Have we got it right? Dr Emma Cattell Medical Oncologist, Musgrove Park Sept 2015

Background • In Feb 2013, it was noted that there had been a general reduction in serum creatinine levels. • Most patients appeared to have dropped approximately 20 µmol/l. • Contacted Biochemistry - ? Reason

Measuring serum creatinine • Historically – colorimetric ‘Jaffe’ method used. • This was calibrated against a reference method, but remained concerns over specificity. • Interactions – bilirubin, drugs etc • New ‘enzymatic’ method which should give reference method results. • All UK labs assessed on performance against ref method values- so likely to be a UK wide/international issue

Impact • Instigating dose reductions for renally cleared drugs eg cisplatin and capecitabine. • Calculation of carboplatin doses which are based on the calvert formula – Dose = Target AUC x(GFR+25).

International concerns • In October 2010 the NCI (National Cancer Institute) Cancer Therapy Evaluation programme recognised the issue and published a letter recommending that carboplatin doses were capped at a GFR of 125 ml/min. AUC Max carboplatin dose 4 600 5 750 6 900 • GOG also recommended using a minimum creatinine of 0. 7 mg/dl(? 61 umol/l) , using pre-op not post-op Cr and adjusted ideal not actual body weight if BMI>25
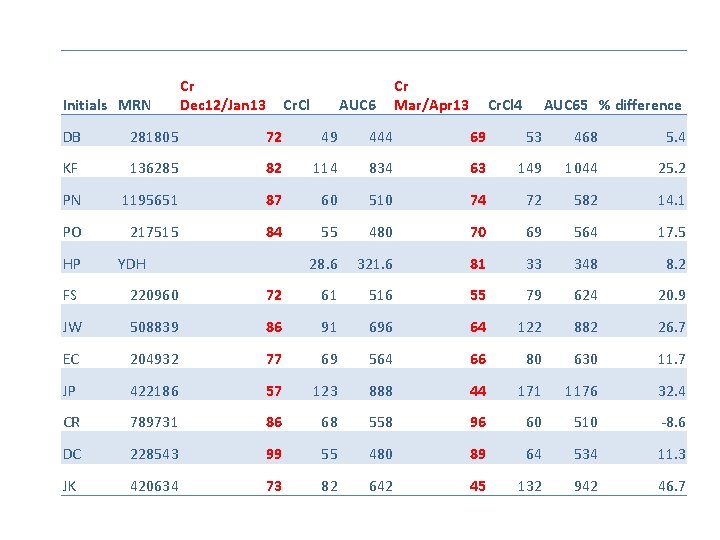
Initials MRN Cr Dec 12/Jan 13 Cr. Cl AUC 6 Cr Mar/Apr 13 Cr. Cl 4 AUC 65 % difference DB 281805 72 49 444 69 53 468 5. 4 KF 136285 82 114 834 63 149 1044 25. 2 PN 1195651 87 60 510 74 72 582 14. 1 PO 217515 84 55 480 70 69 564 17. 5 28. 6 321. 6 81 33 348 8. 2 HP YDH FS 220960 72 61 516 55 79 624 20. 9 JW 508839 86 91 696 64 122 882 26. 7 EC 204932 77 69 564 66 80 630 11. 7 JP 422186 57 123 888 44 171 1176 32. 4 CR 789731 86 68 558 96 60 510 -8. 6 DC 228543 99 55 480 89 64 534 11. 3 JK 420634 73 82 642 45 132 942 46. 7

PREVIOUS AUDIT • Following these concerns, we increased the number of patients having an isotope GFR (esp for ICON 8 patients. ) • Looked at all the patients who had an isotope GFR from March to Dec 2013, and calculated the dose of carboplatin they would have received using cockcroft and gault calculated clearance, at AUC 5 and AUC 6. • Also looked at alternatives such as wright formula.
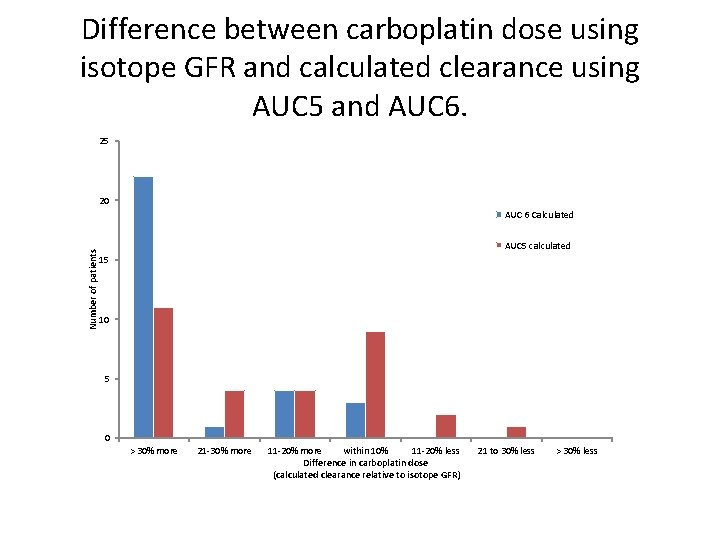
Difference between carboplatin dose using isotope GFR and calculated clearance using AUC 5 and AUC 6. 25 20 Number of patients AUC 6 Calculated AUC 5 calculated 15 10 5 0 > 30% more 21 -30% more 11 -20% more within 10% 11 -20% less Difference in carboplatin dose (calculated clearance relative to isotope GFR) 21 to 30% less > 30% less

Actions • For most patients, esp with low creatinine, consider starting at AUC 5 and increase to AUC 6 according to toleration. • Cap Cr. Cl at 125 ml/min • Audit of toxicity in pts having carboplatin. • Consider Isotope GFR (for all!!? ? , if Cr <50)

AUDIT OF CARBOPLATIN DOSES BEFORE AND AFTER THE CHANGE IN CREATININE MEASUREMENTS. • All patients with a gynae cancer who commenced carboplatin based chemotherapy for 2 time periods. – 1 st June 2011 to 1 st June 2012 – 1 st April 2013 to 1 st April 2014 Assay changed Feb 2013 • Excluded patients on ICON 8, as all had an isotope GFR.

Data collected • • Age of patient Primary Line of therapy. Interval debulk Response Dose of carboplatin. Dose Reductions Dose Delays.
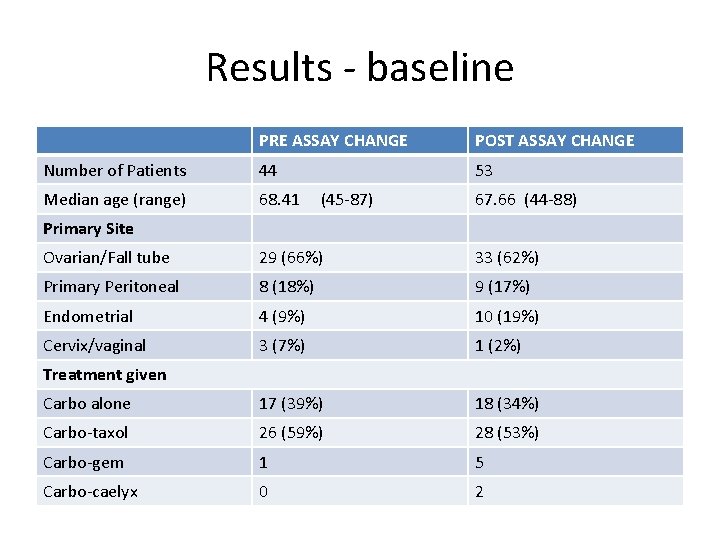
Results - baseline PRE ASSAY CHANGE POST ASSAY CHANGE Number of Patients 44 53 Median age (range) 68. 41 (45 -87) 67. 66 (44 -88) Primary Site Ovarian/Fall tube 29 (66%) 33 (62%) Primary Peritoneal 8 (18%) 9 (17%) Endometrial 4 (9%) 10 (19%) Cervix/vaginal 3 (7%) 1 (2%) Carbo alone 17 (39%) 18 (34%) Carbo-taxol 26 (59%) 28 (53%) Carbo-gem 1 5 Carbo-caelyx 0 2 Treatment given
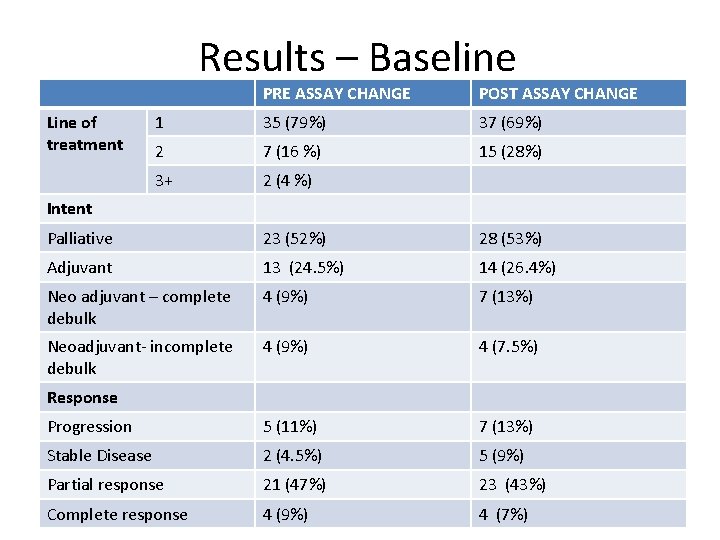
Results – Baseline Line of treatment PRE ASSAY CHANGE POST ASSAY CHANGE 1 35 (79%) 37 (69%) 2 7 (16 %) 15 (28%) 3+ 2 (4 %) Intent Palliative 23 (52%) 28 (53%) Adjuvant 13 (24. 5%) 14 (26. 4%) Neo adjuvant – complete debulk 4 (9%) 7 (13%) Neoadjuvant- incomplete debulk 4 (9%) 4 (7. 5%) Progression 5 (11%) 7 (13%) Stable Disease 2 (4. 5%) 5 (9%) Partial response 21 (47%) 23 (43%) Complete response 4 (9%) 4 (7%) Response
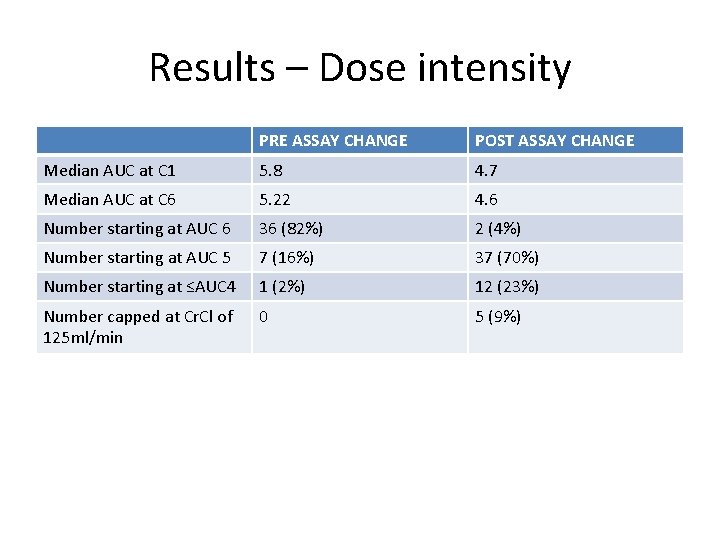
Results – Dose intensity PRE ASSAY CHANGE POST ASSAY CHANGE Median AUC at C 1 5. 8 4. 7 Median AUC at C 6 5. 22 4. 6 Number starting at AUC 6 36 (82%) 2 (4%) Number starting at AUC 5 7 (16%) 37 (70%) Number starting at ≤AUC 4 1 (2%) 12 (23%) Number capped at Cr. Cl of 125 ml/min 0 5 (9%)
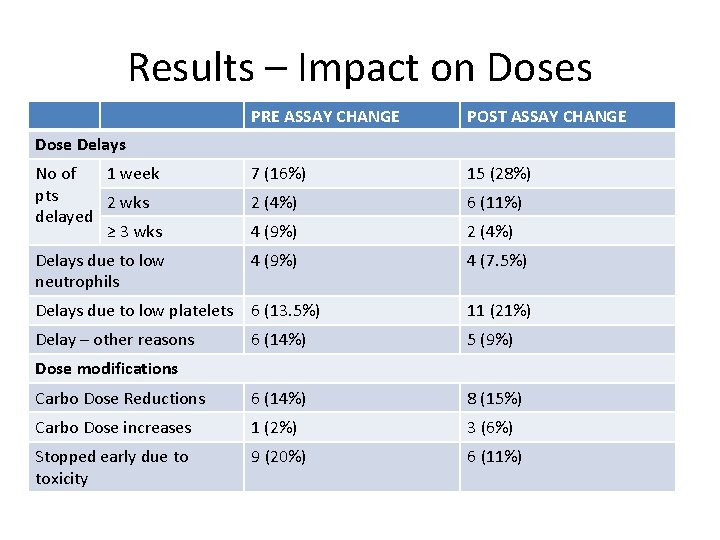
Results – Impact on Doses PRE ASSAY CHANGE POST ASSAY CHANGE No of 1 week pts 2 wks delayed ≥ 3 wks 7 (16%) 15 (28%) 2 (4%) 6 (11%) 4 (9%) 2 (4%) Delays due to low neutrophils 4 (9%) 4 (7. 5%) Delays due to low platelets 6 (13. 5%) 11 (21%) Delay – other reasons 6 (14%) 5 (9%) Carbo Dose Reductions 6 (14%) 8 (15%) Carbo Dose increases 1 (2%) 3 (6%) Stopped early due to toxicity 9 (20%) 6 (11%) Dose Delays Dose modifications

Conclusions • Since the change in creatinine measurements, we have been far more likely to give AUC 5 than AUC 6 when prescribing carboplatin. • Allowing for differences in primaries and line of treatment, no obvious differences in response rates or interval debulking outcomes. • Similar dose reductions and delays. • Slightly less likely to stop early due to toxicity?

Discussion • Feels safe – but ? In line with other centres. • Small patient group - ? Look at progression free survival - ? Numbers to be meaningful. • Are we disadvantaging some pts. . ? Should we be increasing dose more often if tolerating
- Slides: 17