Carbonyl Condensation Reactions 1 Very General Reaction What

Carbonyl Condensation Reactions 1

Very General Reaction. • What is needed? – A Ketone or aldehyde – ONE reactant must have a-Hydrogens – Base (Na. OH) 2
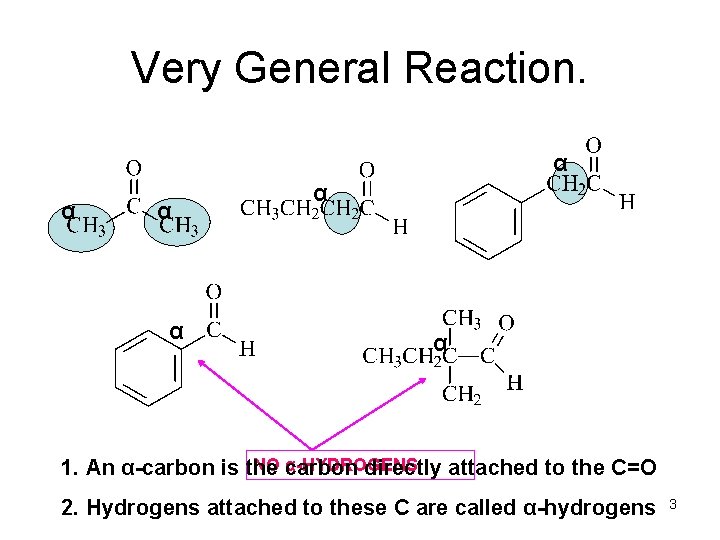
Very General Reaction. α α α NO carbon α-HYDROGENS 1. An α-carbon is the directly attached to the C=O 2. Hydrogens attached to these C are called α-hydrogens 3
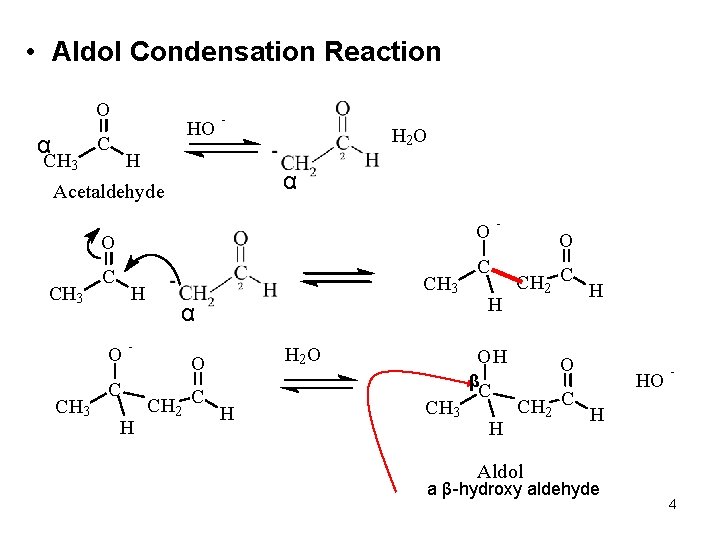
• Aldol Condensation Reaction O α CH 3 HO C - H H 2 O α Acetaldehyde O O CH 3 C H O CH 3 - C H CH 3 C H α O CH 2 C H H 2 O - O CH 2 C H OH β CH 3 C H O CH 2 C H HO - Aldol a β-hydroxy aldehyde 4
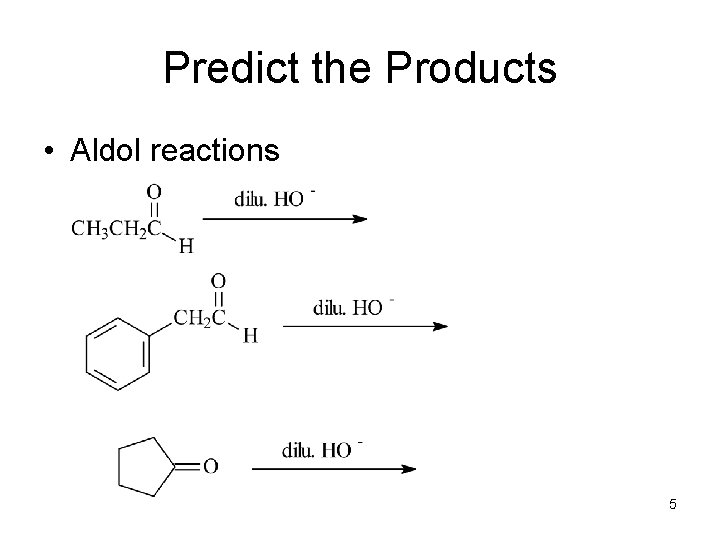
Predict the Products • Aldol reactions 5
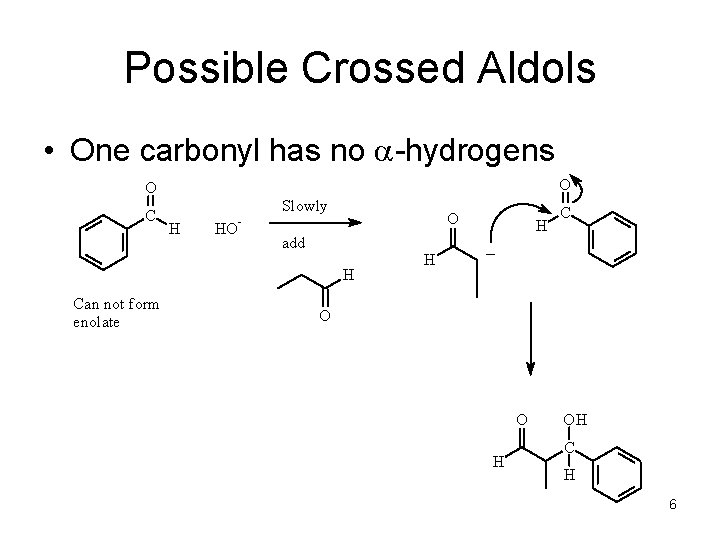
Possible Crossed Aldols • One carbonyl has no a-hydrogens O O HO add H Can not form enolate H C H H - Slowly O C O O H OH C H 6
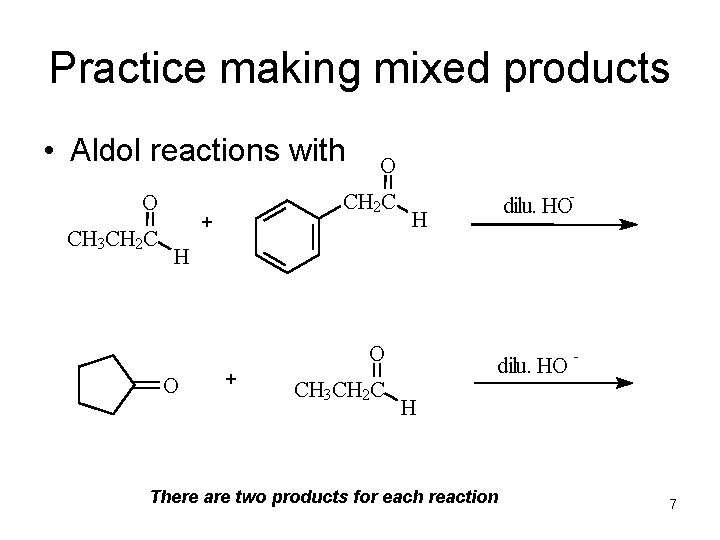
Practice making mixed products • Aldol reactions with O CH 3 CH 2 C O CH 2 C + - dilu. HO H H O O + CH 3 CH 2 C dilu. HO - H There are two products for each reaction 7
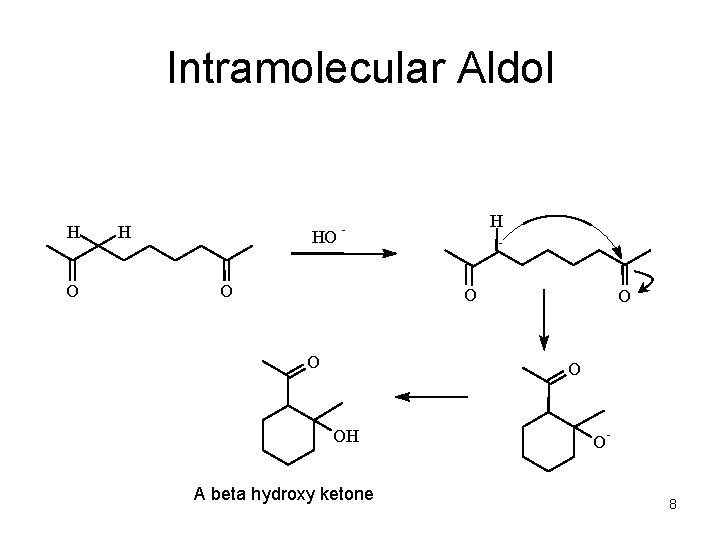
Intramolecular Aldol H O H HO H - O O OH A beta hydroxy ketone O - 8
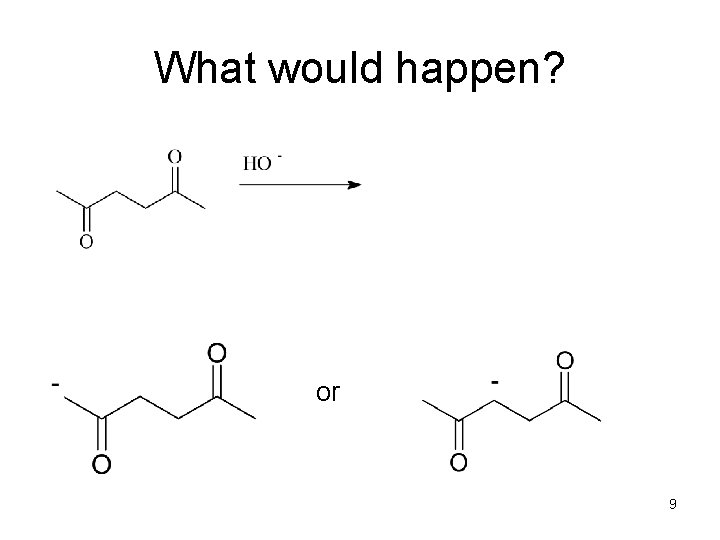
What would happen? or 9
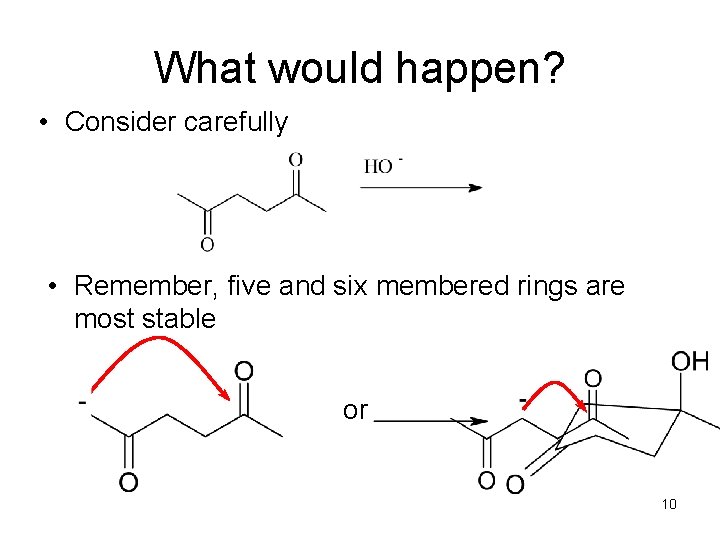
What would happen? • Consider carefully • Remember, five and six membered rings are most stable or 10
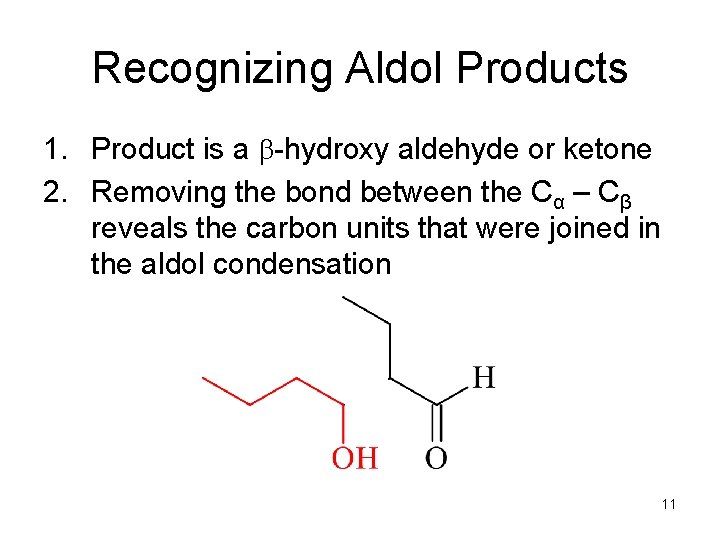
Recognizing Aldol Products 1. Product is a b-hydroxy aldehyde or ketone 2. Removing the bond between the Cα – Cβ reveals the carbon units that were joined in the aldol condensation 11
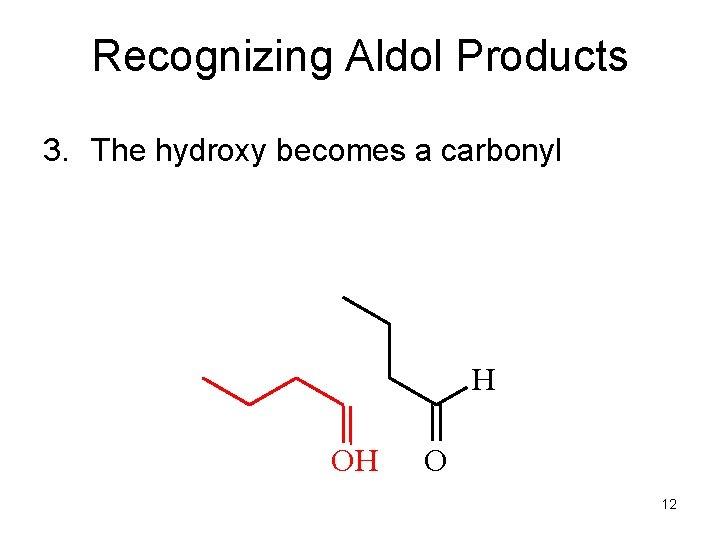
Recognizing Aldol Products 3. The hydroxy becomes a carbonyl H OH O 12
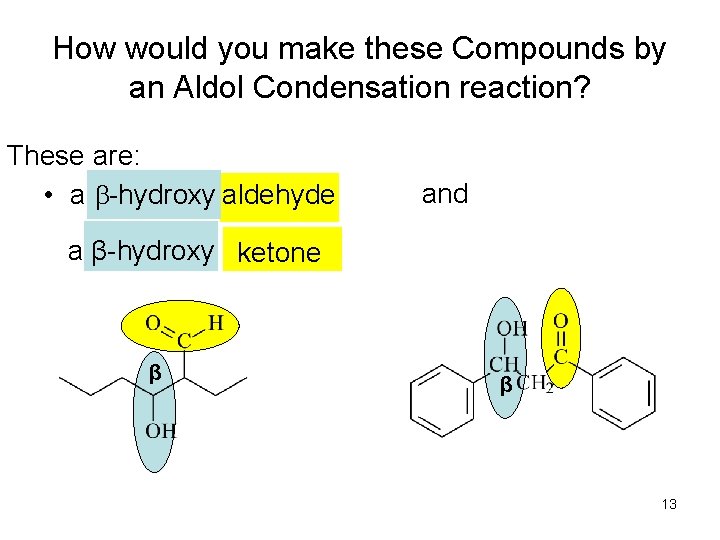
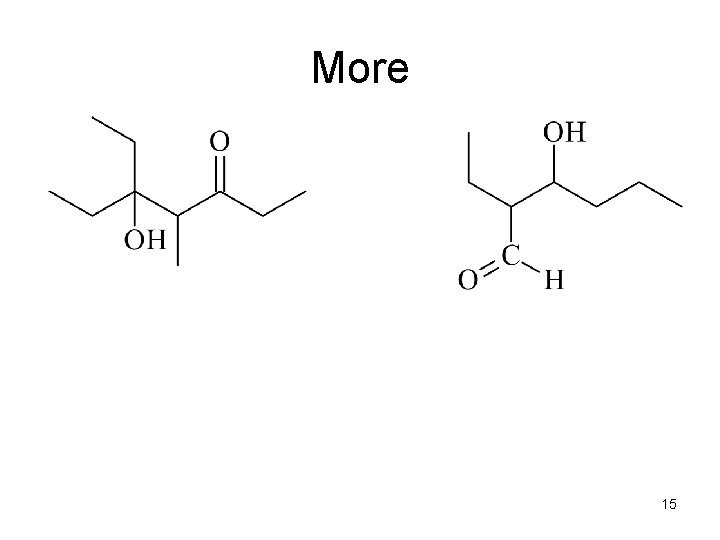
How would you make these Compounds by an Aldol Condensation reaction? These are: • a b-hydroxy aldehyde and a β-hydroxy ketone β β 13
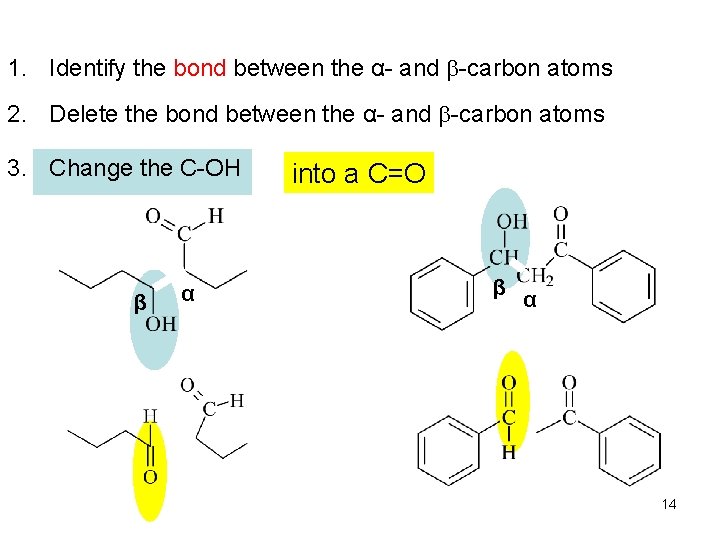
1. Identify the bond between the α- and b-carbon atoms 2. Delete the bond between the α- and b-carbon atoms 3. Change the C-OH β α into a C=O β α 14
More 15
- Slides: 15