Carbonyl Chemistry Predict the products This is best

Carbonyl Chemistry Predict the products This is best viewed in the slide show view due to the animations. SO PLEASE DOWNLOAD IT AND USE POWERPOINT TO VIEW IT.
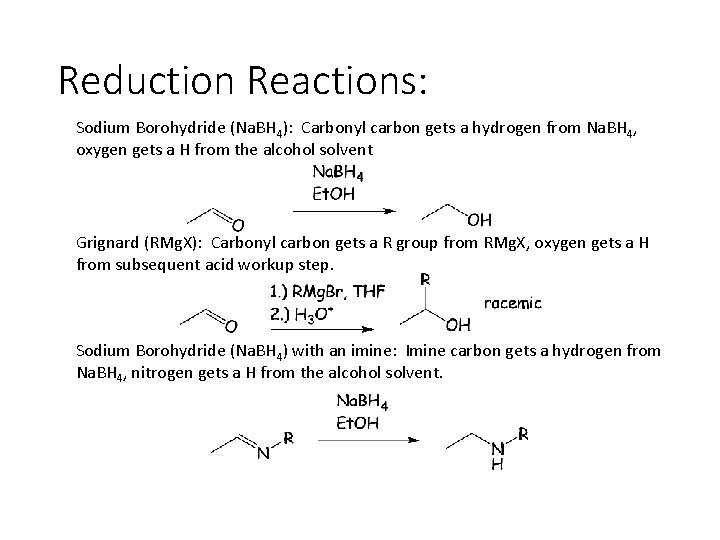
Reduction Reactions: Sodium Borohydride (Na. BH 4): Carbonyl carbon gets a hydrogen from Na. BH 4, oxygen gets a H from the alcohol solvent Grignard (RMg. X): Carbonyl carbon gets a R group from RMg. X, oxygen gets a H from subsequent acid workup step. Sodium Borohydride (Na. BH 4) with an imine: Imine carbon gets a hydrogen from Na. BH 4, nitrogen gets a H from the alcohol solvent.
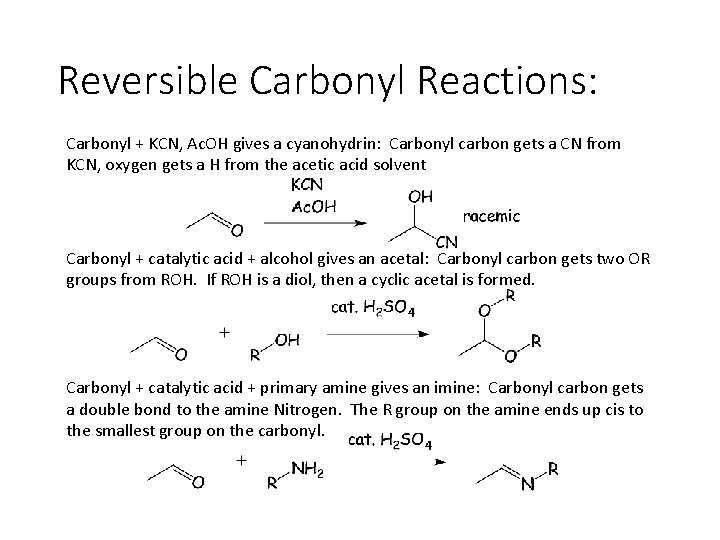
Reversible Carbonyl Reactions: Carbonyl + KCN, Ac. OH gives a cyanohydrin: Carbonyl carbon gets a CN from KCN, oxygen gets a H from the acetic acid solvent Carbonyl + catalytic acid + alcohol gives an acetal: Carbonyl carbon gets two OR groups from ROH. If ROH is a diol, then a cyclic acetal is formed. Carbonyl + catalytic acid + primary amine gives an imine: Carbonyl carbon gets a double bond to the amine Nitrogen. The R group on the amine ends up cis to the smallest group on the carbonyl.
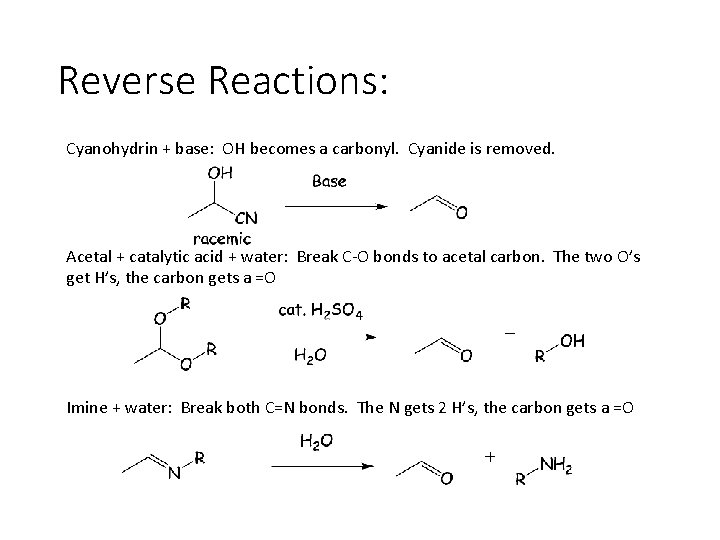
Reverse Reactions: Cyanohydrin + base: OH becomes a carbonyl. Cyanide is removed. Acetal + catalytic acid + water: Break C-O bonds to acetal carbon. The two O’s get H’s, the carbon gets a =O Imine + water: Break both C=N bonds. The N gets 2 H’s, the carbon gets a =O
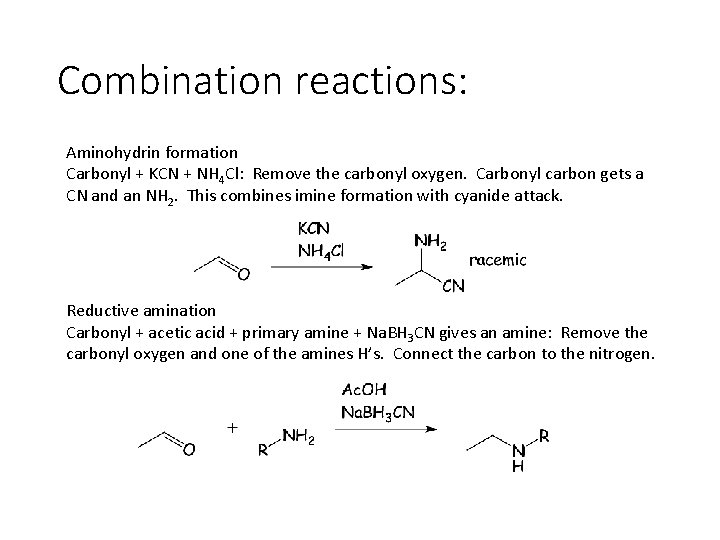
Combination reactions: Aminohydrin formation Carbonyl + KCN + NH 4 Cl: Remove the carbonyl oxygen. Carbonyl carbon gets a CN and an NH 2. This combines imine formation with cyanide attack. Reductive amination Carbonyl + acetic acid + primary amine + Na. BH 3 CN gives an amine: Remove the carbonyl oxygen and one of the amines H’s. Connect the carbon to the nitrogen.
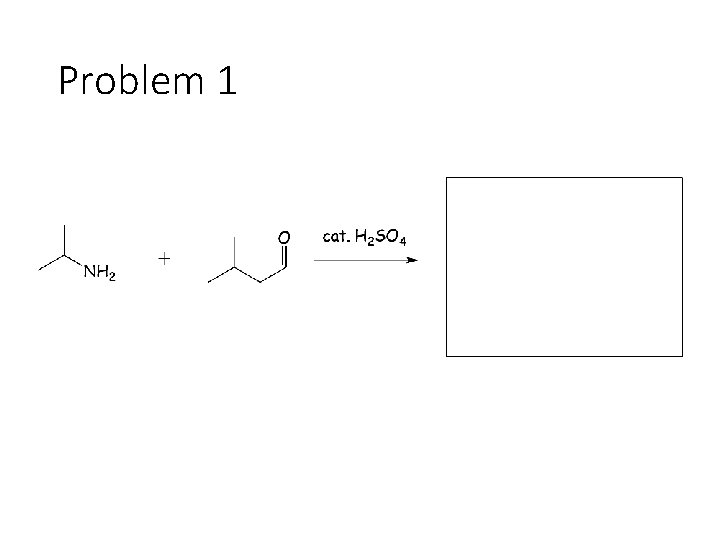
Problem 1
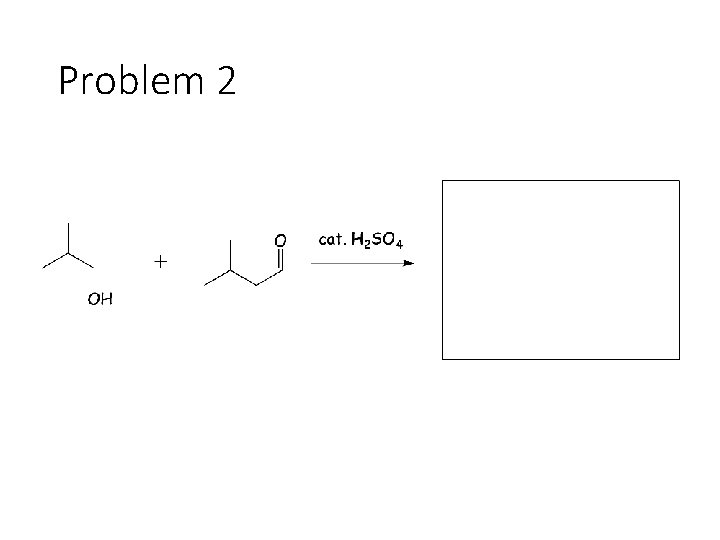
Problem 2
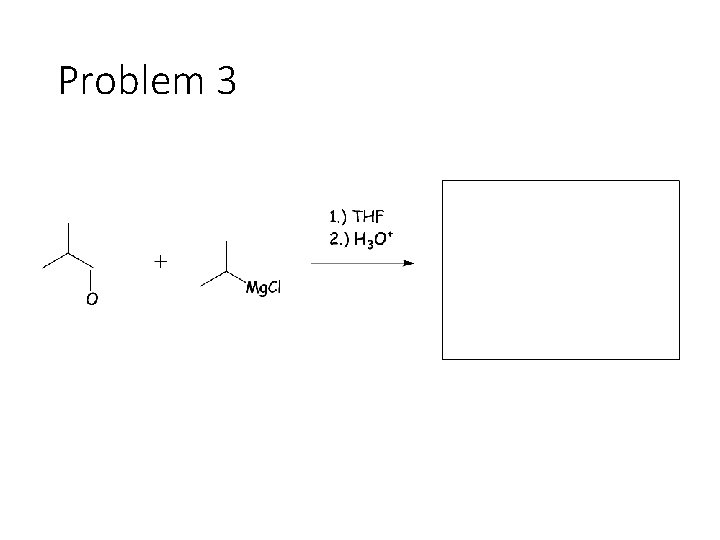
Problem 3
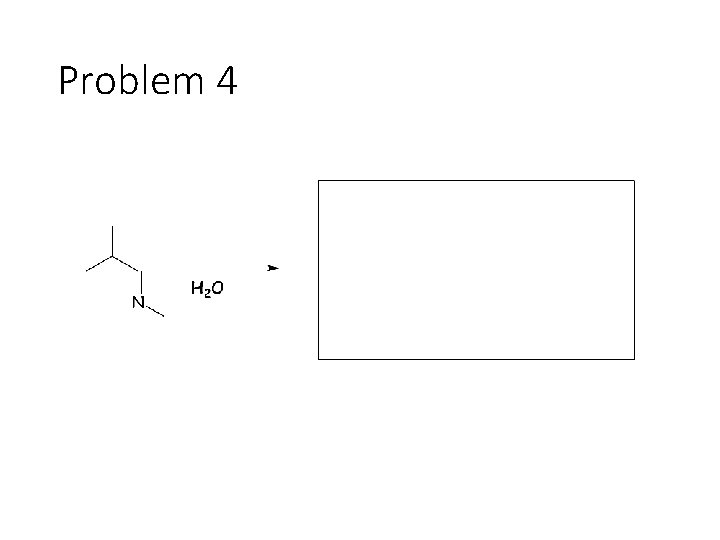
Problem 4
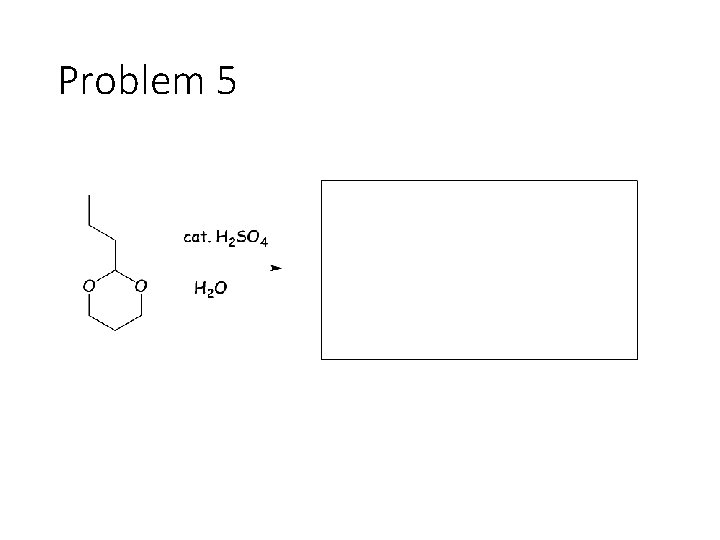
Problem 5
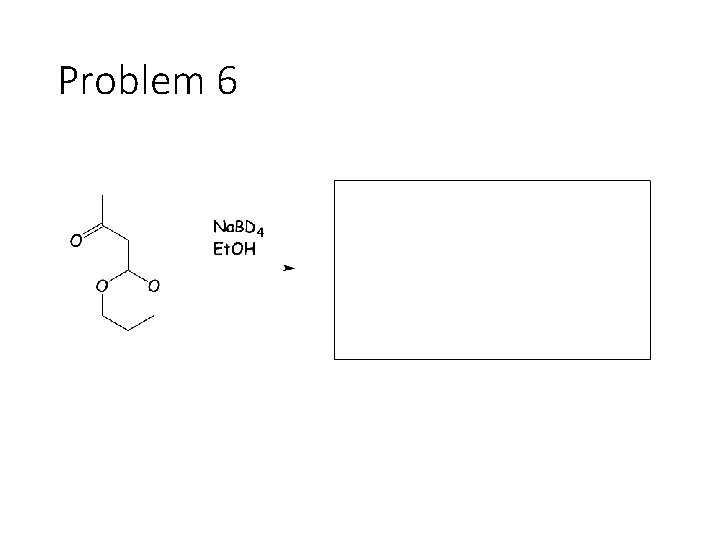
Problem 6
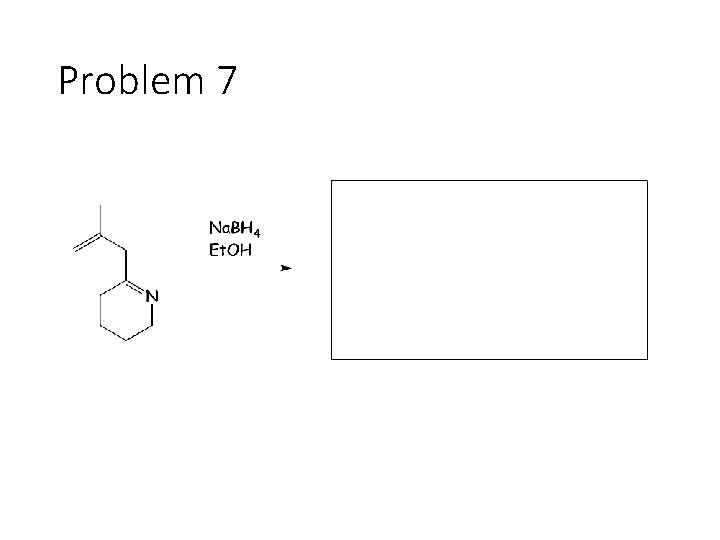
Problem 7
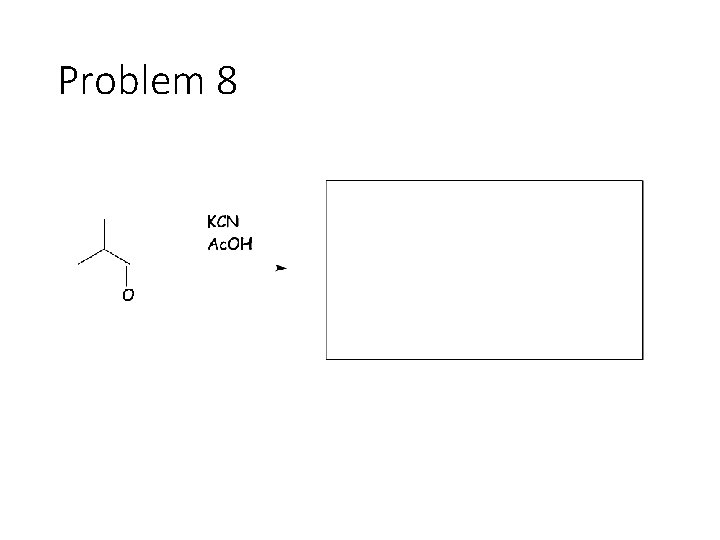
Problem 8
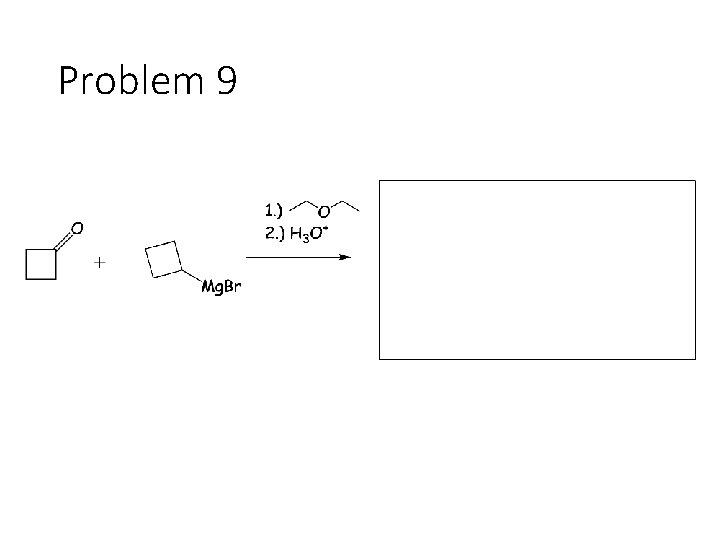
Problem 9
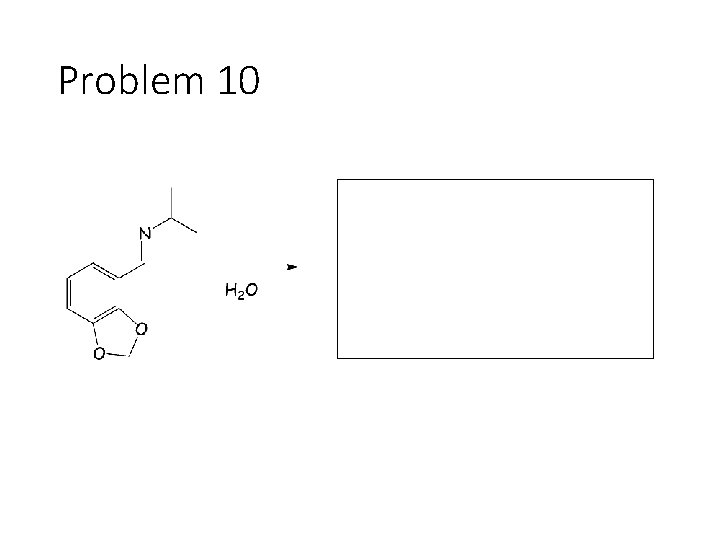
Problem 10
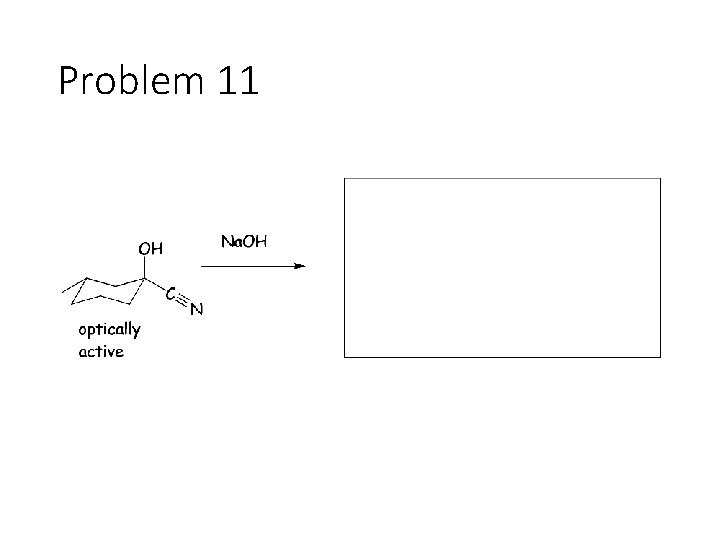
Problem 11
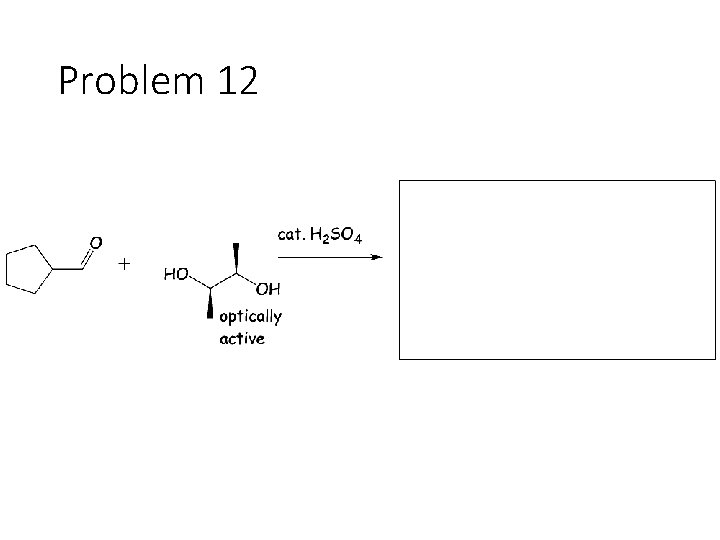
Problem 12
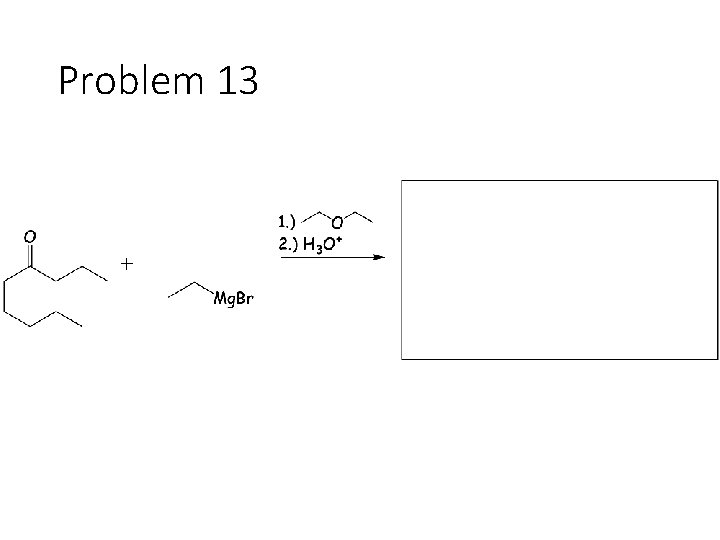
Problem 13
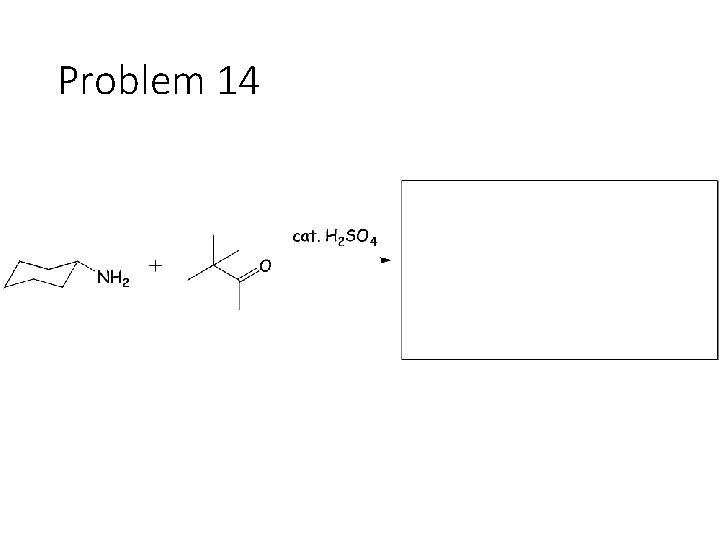
Problem 14
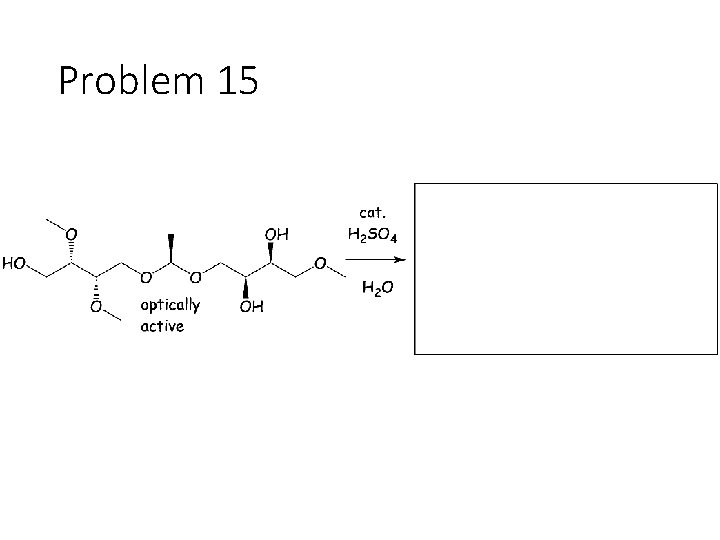
Problem 15
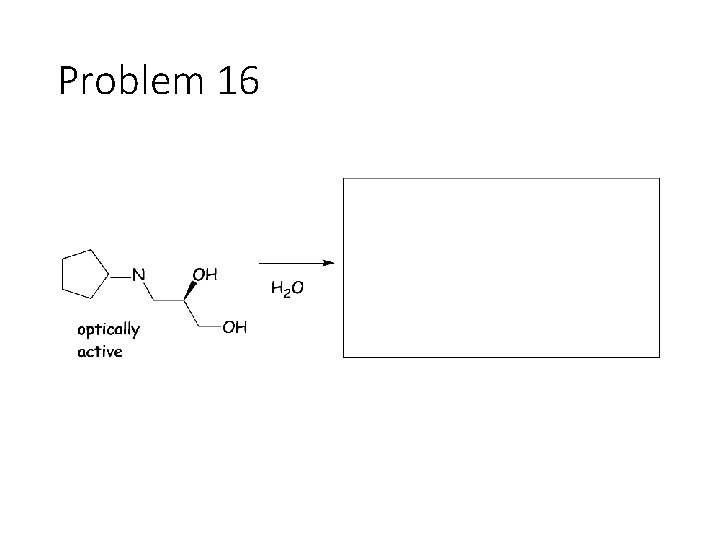
Problem 16
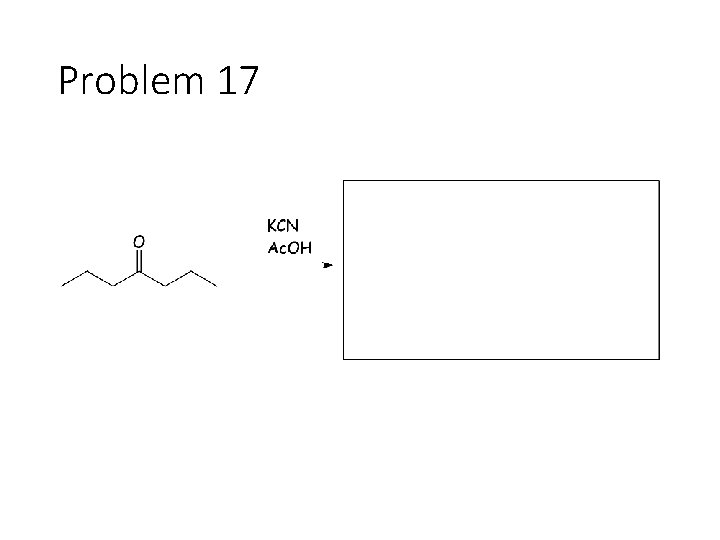
Problem 17
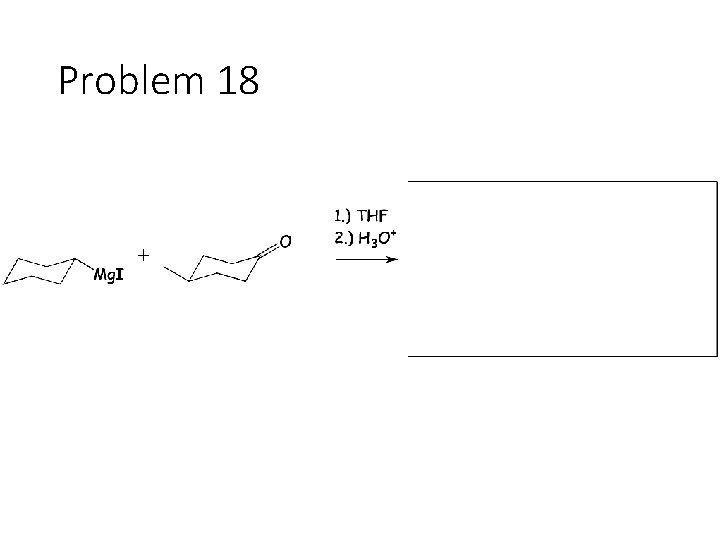
Problem 18
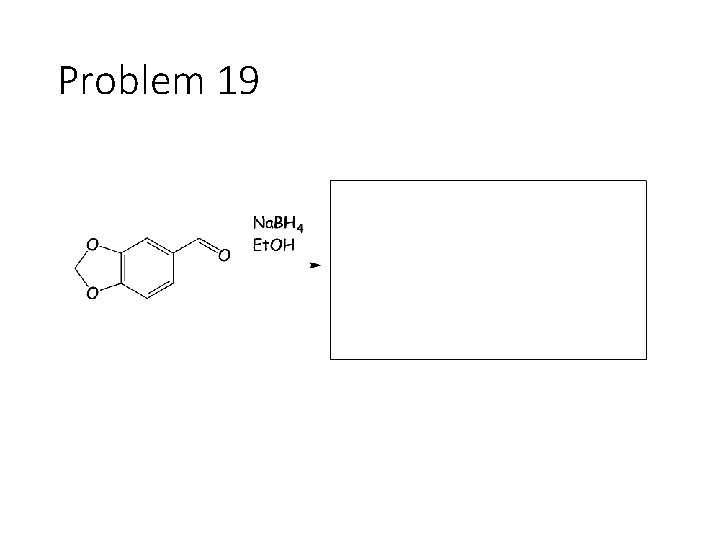
Problem 19
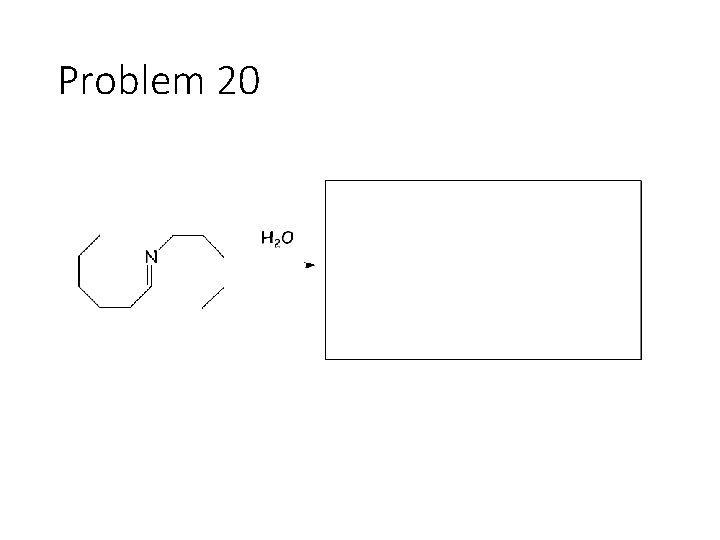
Problem 20
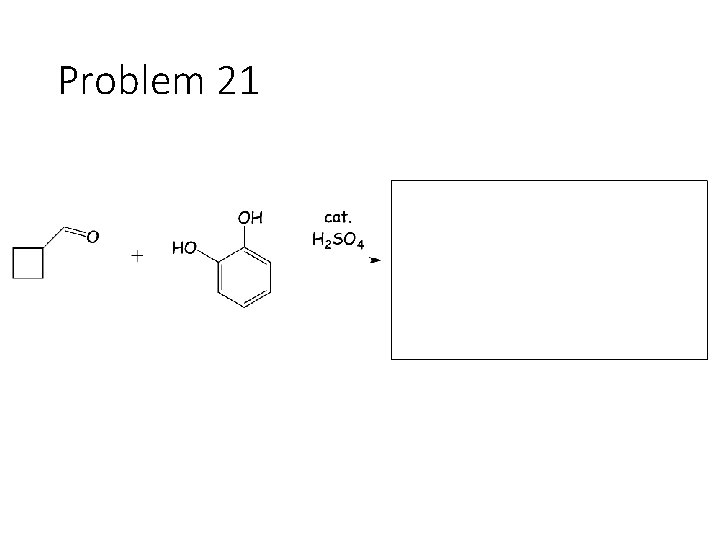
Problem 21
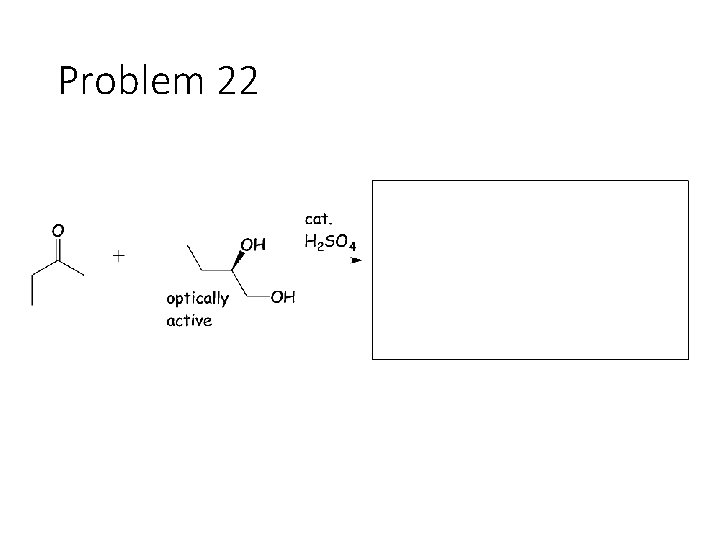
Problem 22
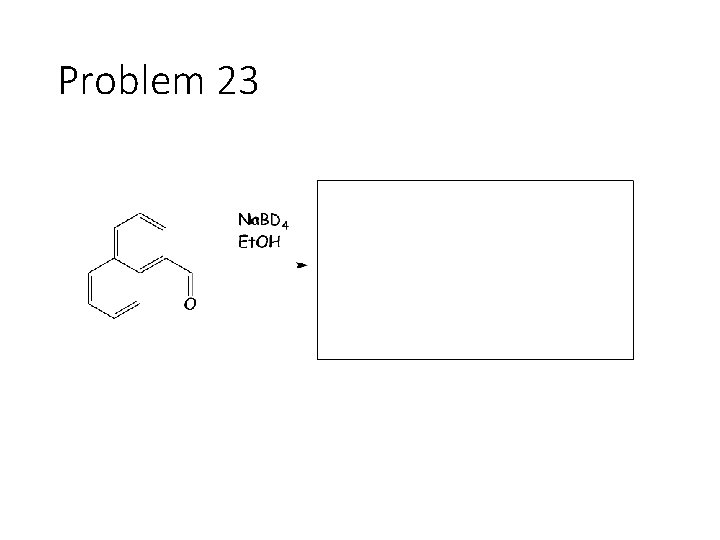
Problem 23
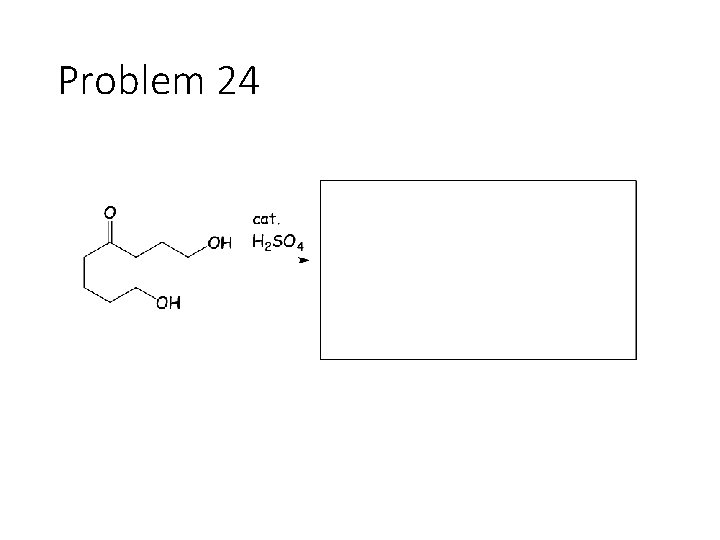
Problem 24
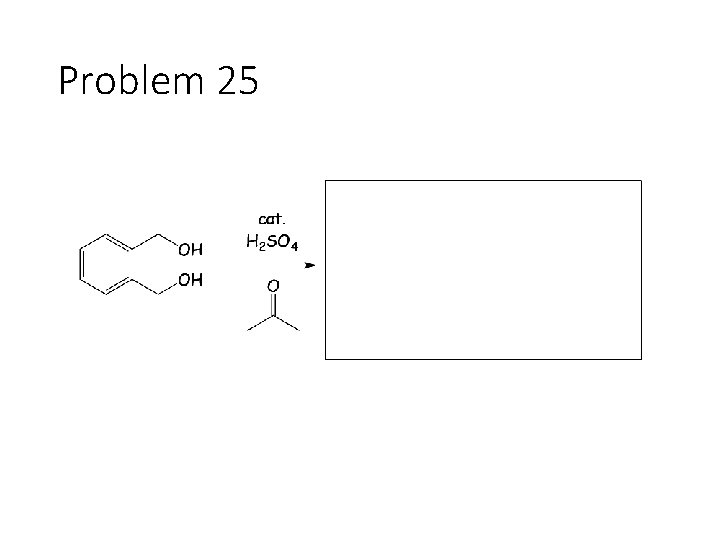
Problem 25
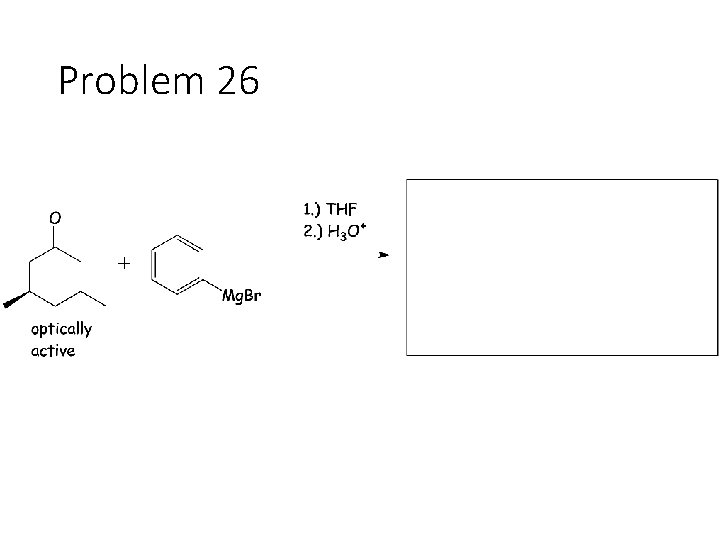
Problem 26
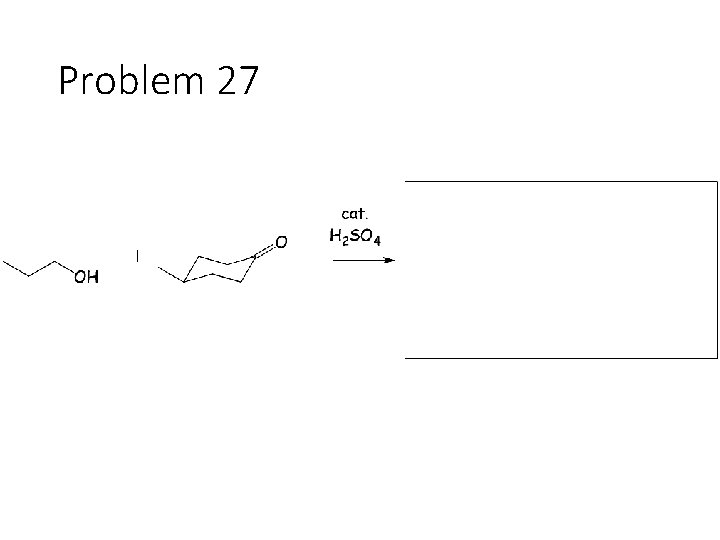
Problem 27
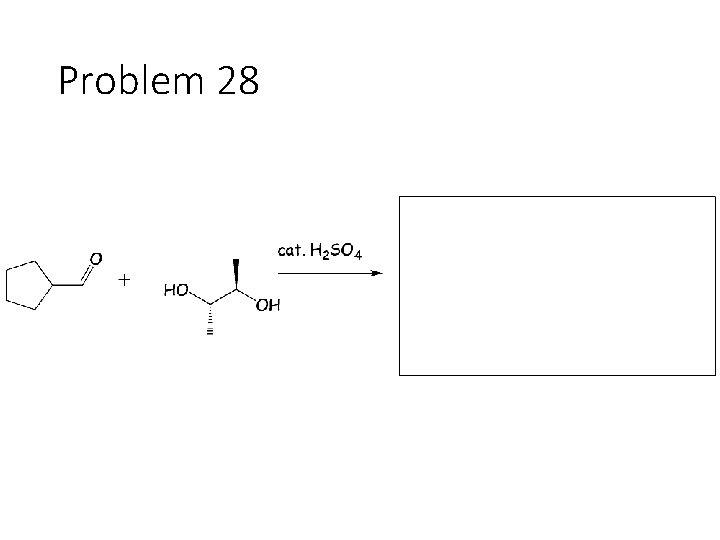
Problem 28
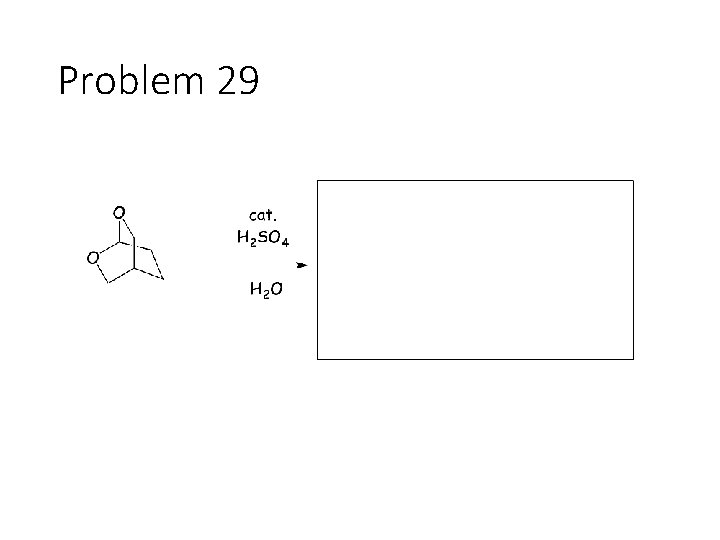
Problem 29
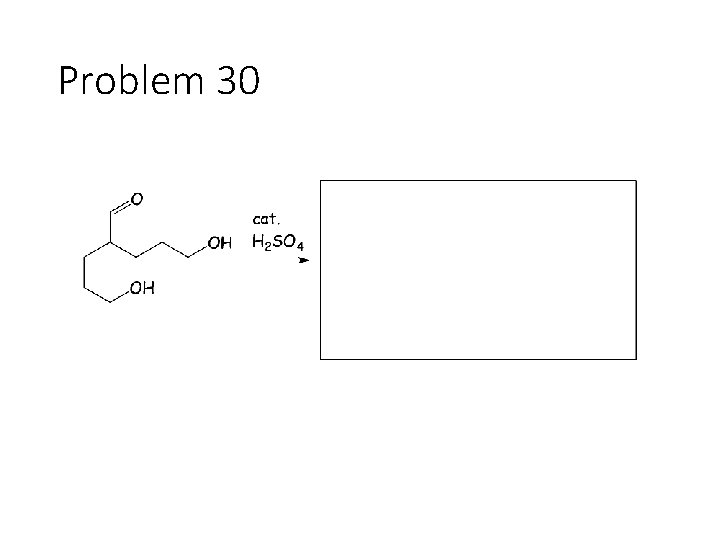
Problem 30
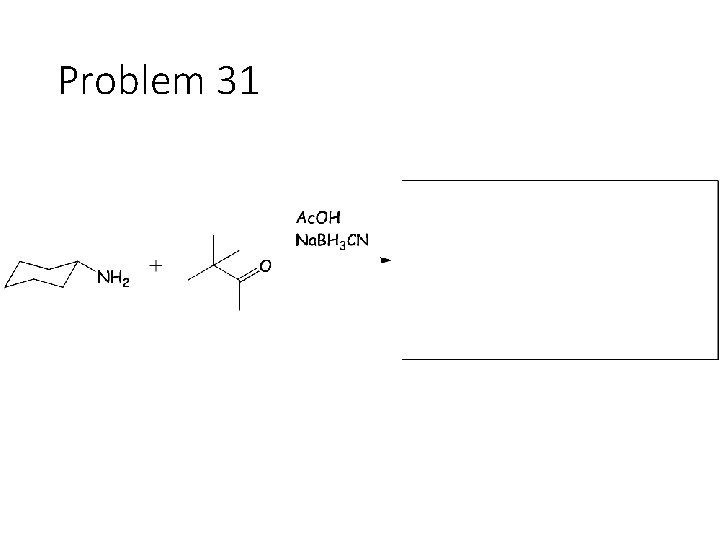
Problem 31
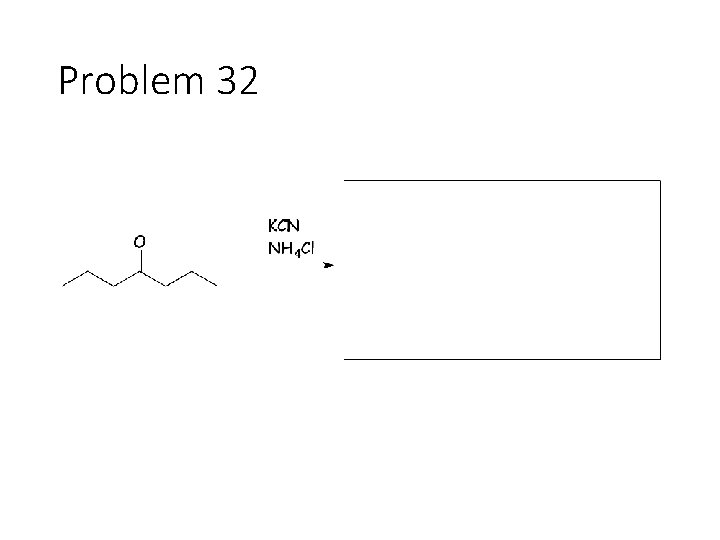
Problem 32
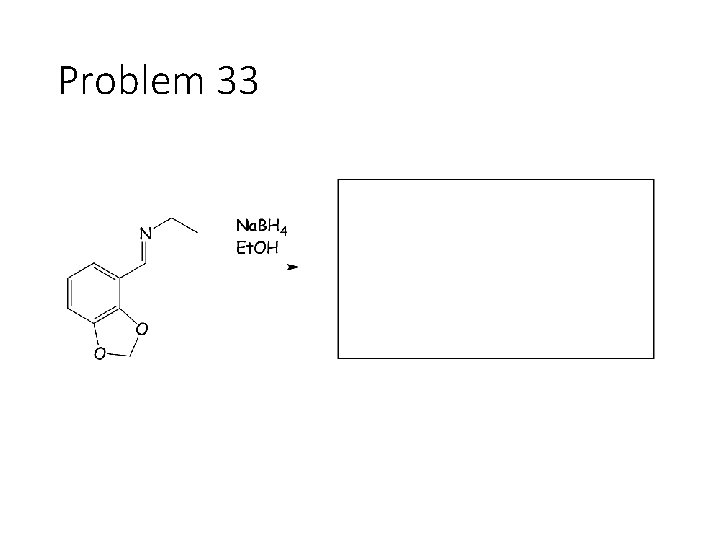
Problem 33
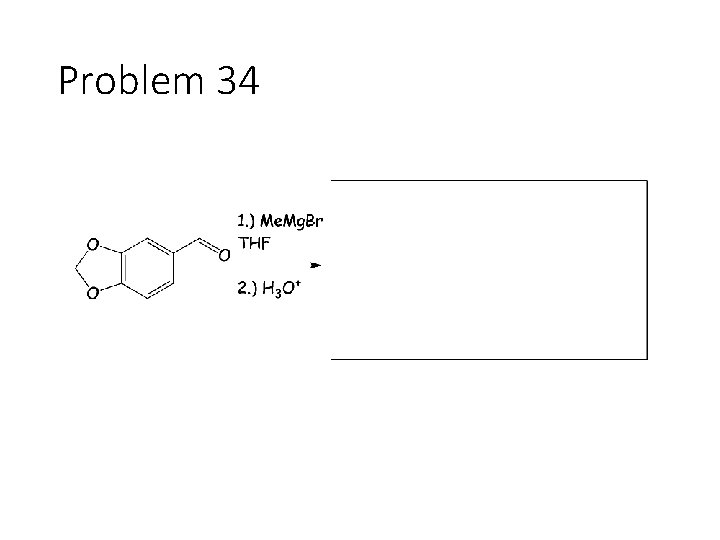
Problem 34
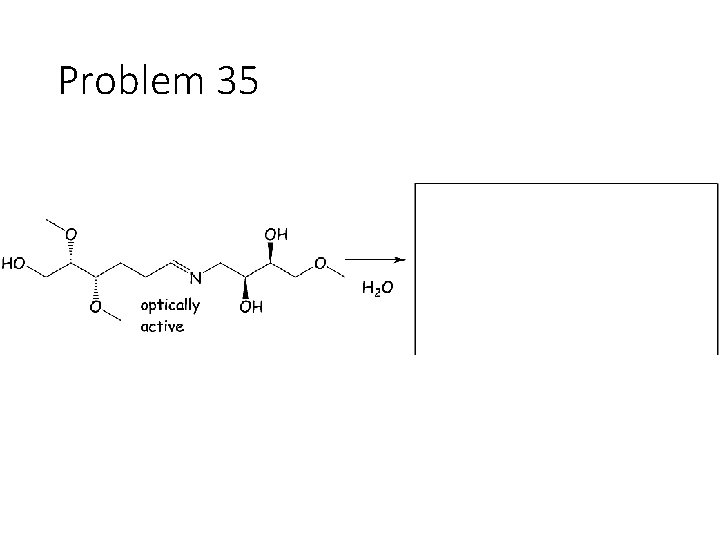
Problem 35
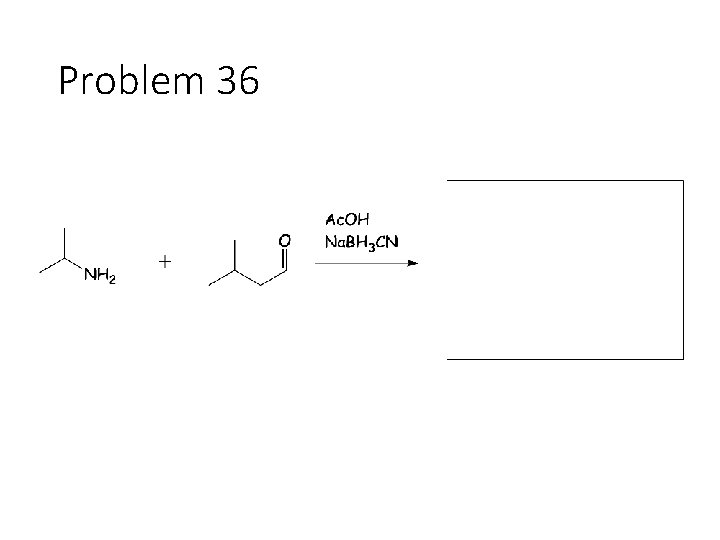
Problem 36
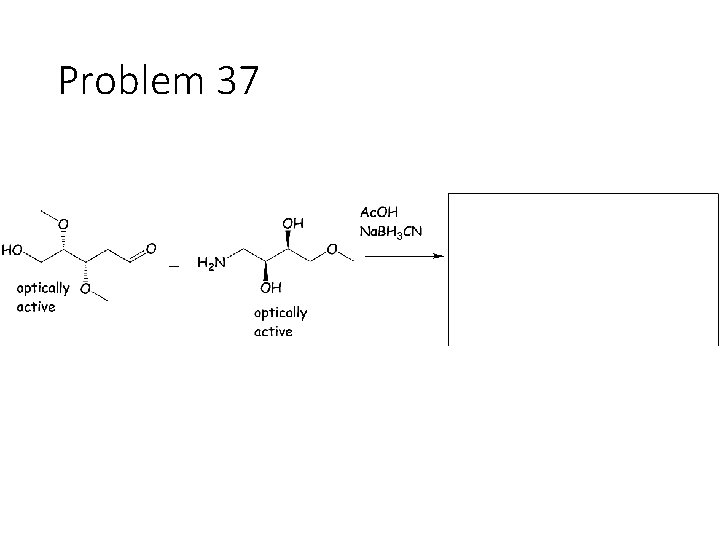
Problem 37
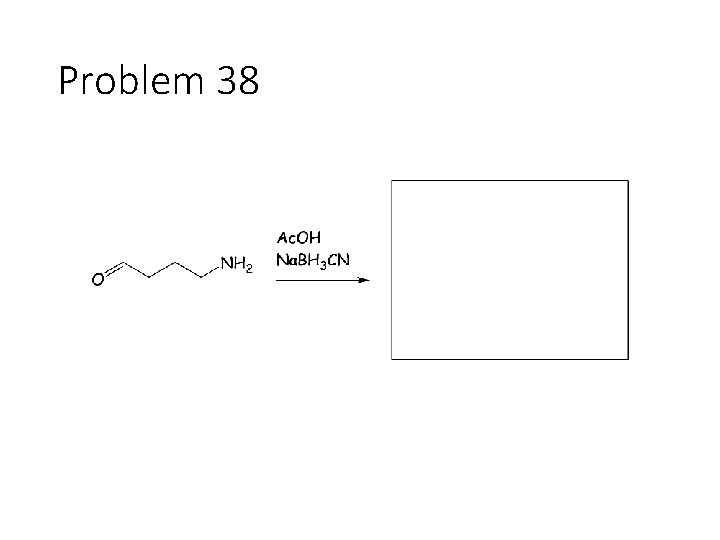
Problem 38
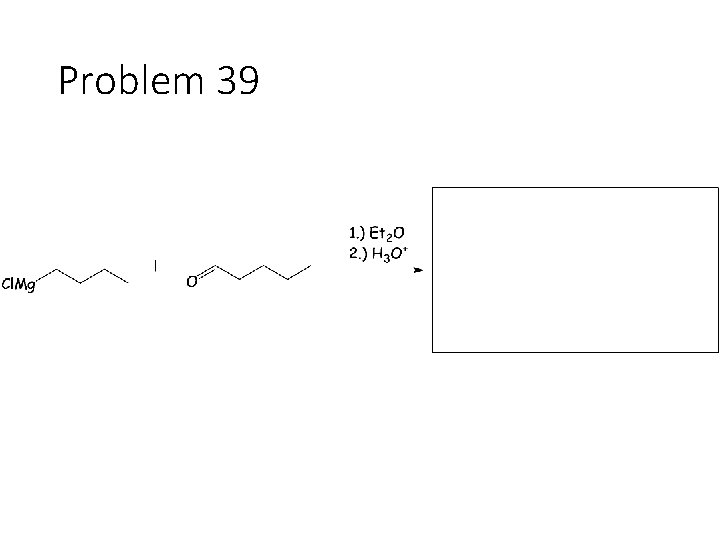
Problem 39
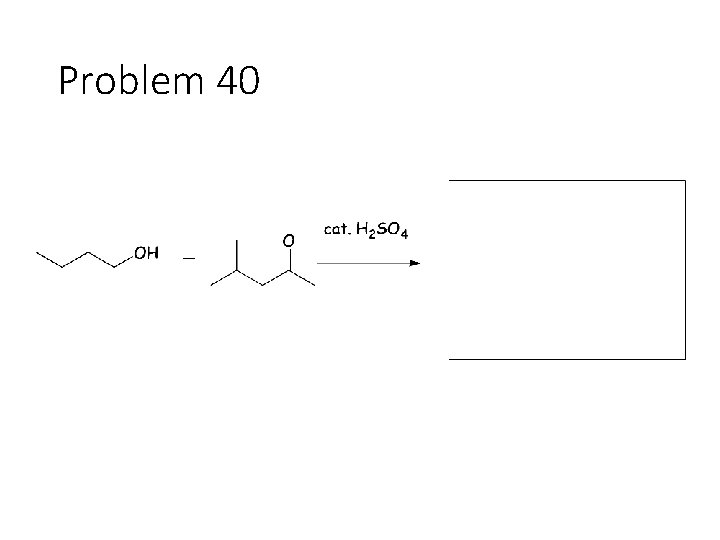
Problem 40
- Slides: 45