CARBONS Start by Counting the number of Carbons


CARBONS Start by Counting the number of Carbons in the longest chain. Be careful of bent molecules. � 2. Find the appropriate prefix (Table P ). � 3. Look for any DOUBLE bonds, TRIPLE bonds, or functional groups. If there are some, so to next slide (step 5). � 4. If there are no extras on the molecule add the suffix “ ANE ”. � e. g. methane, propane � 1.

� 5. If there is a functional group, go to the next slide (step 9). � 6. If there is a DOUBLE bond, use the prefix from step 2 and add the suffix “ ENE ” � 7. If there is a TRIPLE bond, use the prefix from step 2 and add “ YNE”. � 8. Regents trick question: If there is more than one multi-bond, but the appropriate prefix in front of the “ene” or the “yne”. � e. g. butadiene, petatriene

Number the longest CONTINUOUS chain of carbons starting with 1 so that the branch or CARBONS functional group is bonded to the lowest CARBON possible numbered carb on. � 10. If it is just a hydrocarbon branch, go to the next slide, step 11. If it is a functional group on table R, skip the next slide and go to step 15 � 9.

CARBONS count the number of hydrocarbons on the branch. � 12. Use the prefix from table P , add “ YL” � 13. Write the number of the carbon the branch is on, then a dash (-) � 14. list the branch name, then the name of the base HYDROCARBONS hydrocarbon. � e. g. 2 -methyl butane � 11.

Use Table R to determine which functional group you have. � Jot down brief notes for each of the functional groups: � 15.

� Use the number of the carbon where the halogen is bonded � Add the prefix for that halogen from table R to the name of the hydrocarbon. � e. g. 2 -fluorobutane

� Use the number of the carbon where the –OH is bonded � Change the suffix of the hydrocarbon to “ol” � e. g. ethanol, 2 -propanol

� Count � Name the carbons on each side of the O those with the prefixes from Table P � Add “yl” to those. � If they are both the same use “di” � Put them in front of the word “ether” � e. g. methyl butyl ether � Dimethyl ether
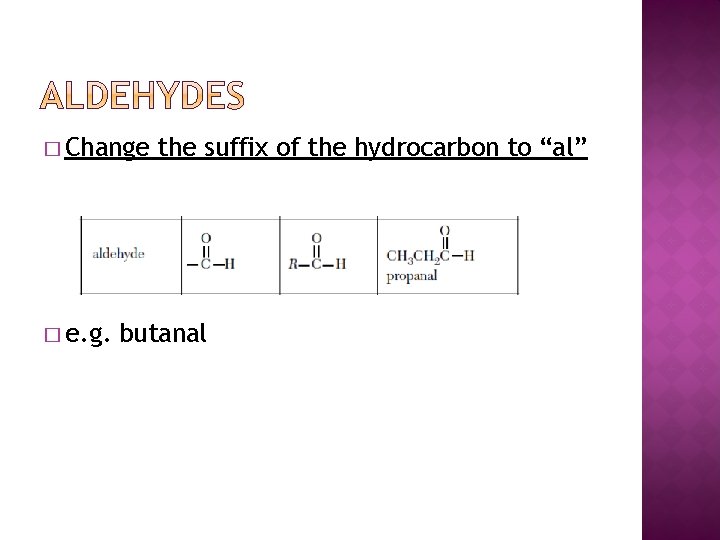
� Change � e. g. the suffix of the hydrocarbon to “al” butanal

� Use � Add the number which carbon the =O is the suffix “one” to the name of the hydrocarbon � e. g. 2 -butanone

� Also called carboxilic acids � Change the suffix from the hydrocarbon to “ic” or “oic” � Add the word “acid” � e. g. acetic acid � Butyric acid

� Count � Add the carbon chains on either side of the –O- the suffix “yl” to the shorter chain. � Name the longer chain with the suffix “oate” � e. g. methyl butanoate

� Make sure there is a nitrogen and no oxygen � Count the carbons � Use the prefixes from Table P � Use the suffix “amine” � Don’t worry about the carbon numbers, they never ask that
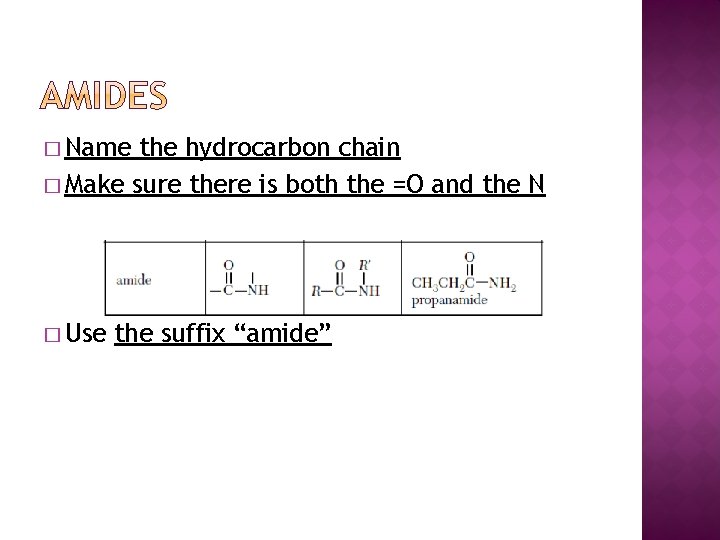
� Name the hydrocarbon chain � Make sure there is both the =O and the N � Use the suffix “amide”
- Slides: 15