CarbonCarbon Bond Formation and Synthesis 24 1 Heck
Carbon-Carbon Bond Formation and Synthesis 24 -1
Heck Reaction u u u The palladium-catalyzed C-C coupling between aryl halides or vinyl halides and activated alkenes in the presence of a base is referred as the "Heck Reaction". Recent developments in the catalysts and reaction conditions have resulted in a much broader range of donors and acceptors being amenable to the Heck Reaction. One of the benefits of the Heck Reaction is its outstanding trans selectivity. 24 -2
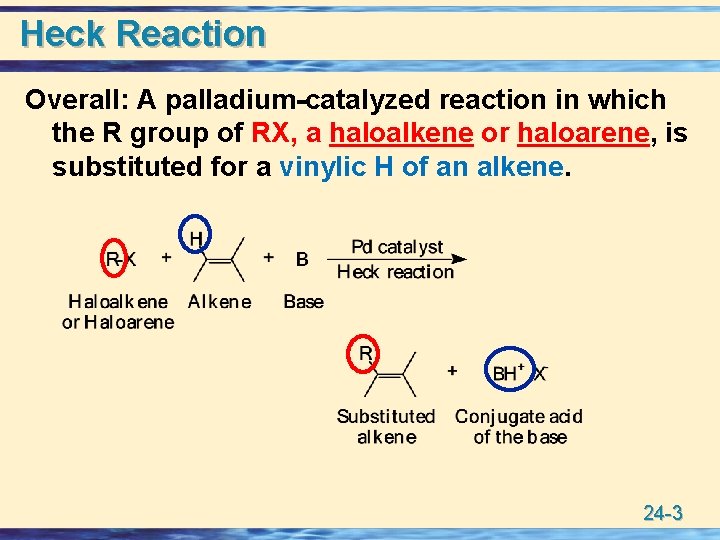
Heck Reaction Overall: A palladium-catalyzed reaction in which the R group of RX, a haloalkene or haloarene, is substituted for a vinylic H of an alkene. 24 -3
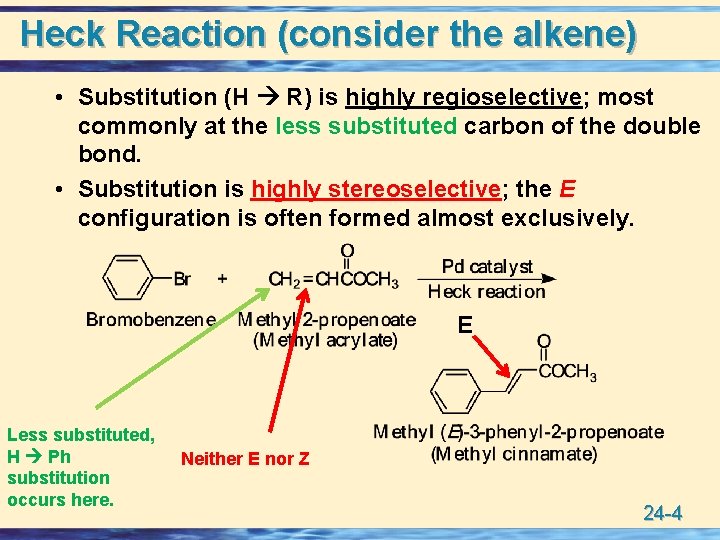
Heck Reaction (consider the alkene) • Substitution (H R) is highly regioselective; most commonly at the less substituted carbon of the double bond. • Substitution is highly stereoselective; the E configuration is often formed almost exclusively. E Less substituted, H Ph substitution occurs here. Neither E nor Z 24 -4
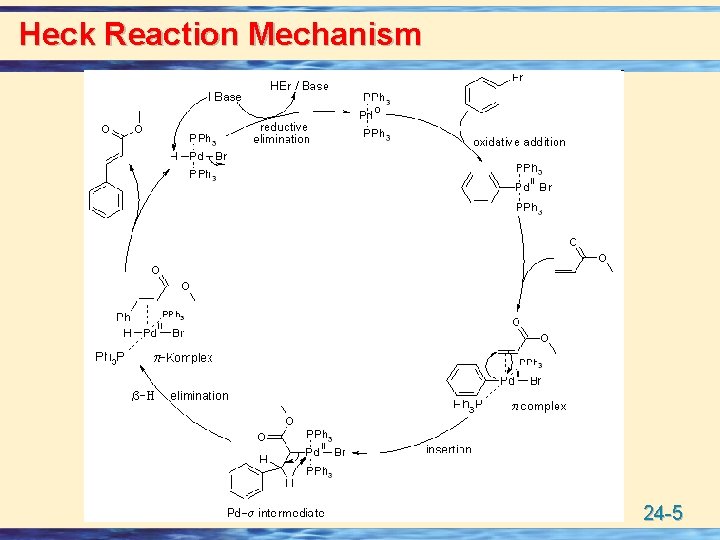
Heck Reaction Mechanism 24 -5

Heck Reaction. Conti…. . u The alkene • The less the crowding on the alkene, the more reactive it is. u The base • Triethylamine, Na+/K+ acetate, and sodium hydrogen carbonate are most common. u The solvent. • Polar aprotic solvents such as DMF, acetonitrile, and DMSO. • aqueous methanol may also be used. u The ligand • Triphenylphosphine, PPh 3, is one of the most common. 24 -6
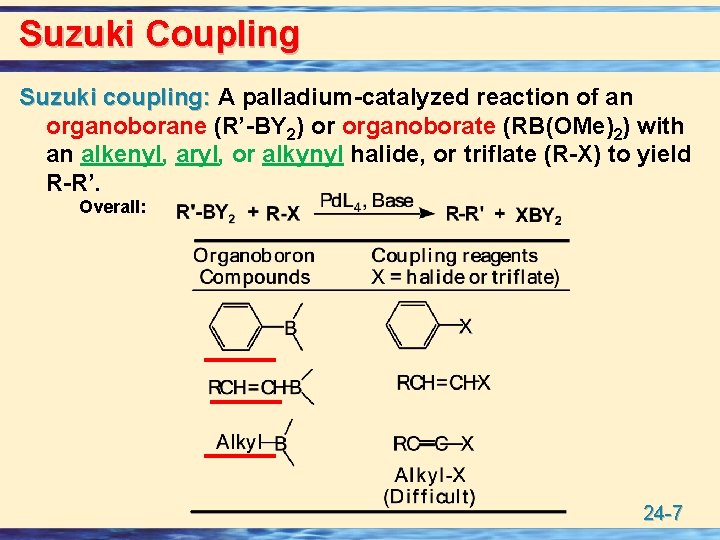
Suzuki Coupling Suzuki coupling: A palladium-catalyzed reaction of an organoborane (R’-BY 2) or organoborate (RB(OMe)2) with an alkenyl, aryl, or alkynyl halide, or triflate (R-X) to yield R-R’. Overall: 24 -7
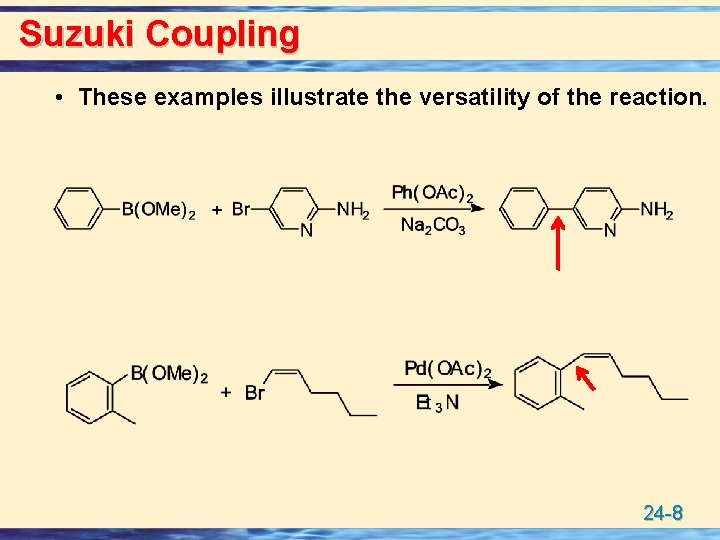
Suzuki Coupling • These examples illustrate the versatility of the reaction. 24 -8
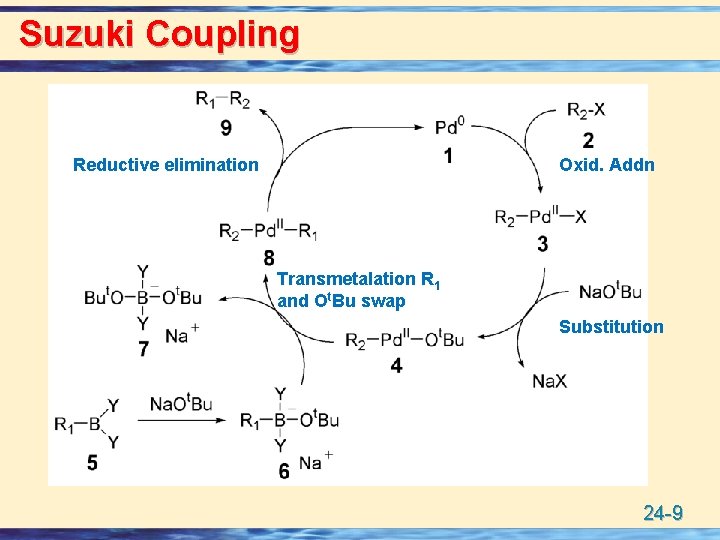
Suzuki Coupling Reductive elimination Oxid. Addn Transmetalation R 1 and Ot. Bu swap Substitution 24 -9
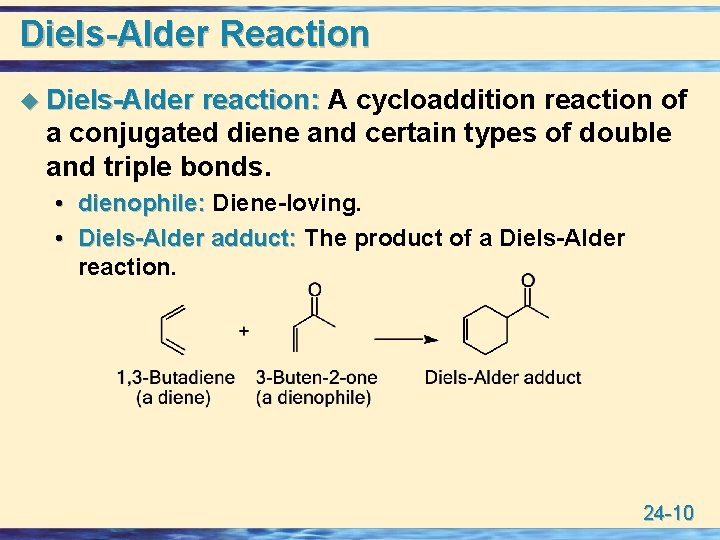
Diels-Alder Reaction u Diels-Alder reaction: A cycloaddition reaction of a conjugated diene and certain types of double and triple bonds. • dienophile: Diene-loving. • Diels-Alder adduct: The product of a Diels-Alder reaction. 24 -10
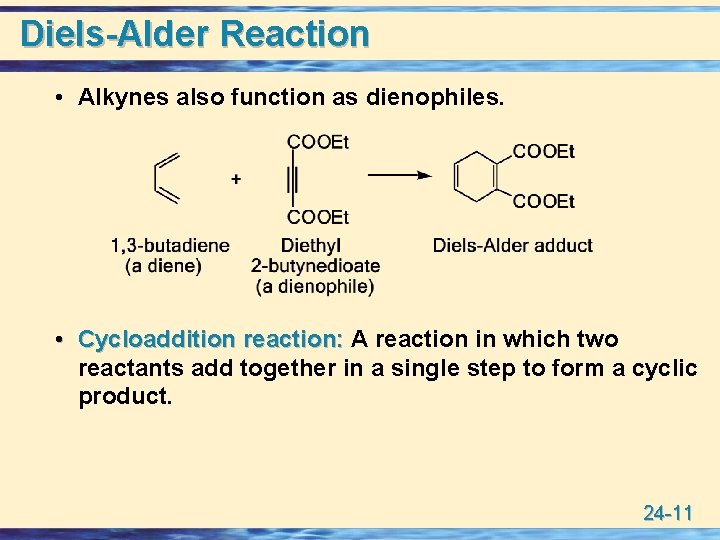
Diels-Alder Reaction • Alkynes also function as dienophiles. • Cycloaddition reaction: A reaction in which two reactants add together in a single step to form a cyclic product. 24 -11

Diels-Alder Reaction • We write a Diels-Alder reaction in the following way: • The special value of D-A reactions are that they: 1. form six-membered rings. 2. form two new C-C bonds at the same time. 3. are stereospecific and regioselective. Note the reaction of butadiene and ethylene gives only traces of cyclohexene. 24 -12
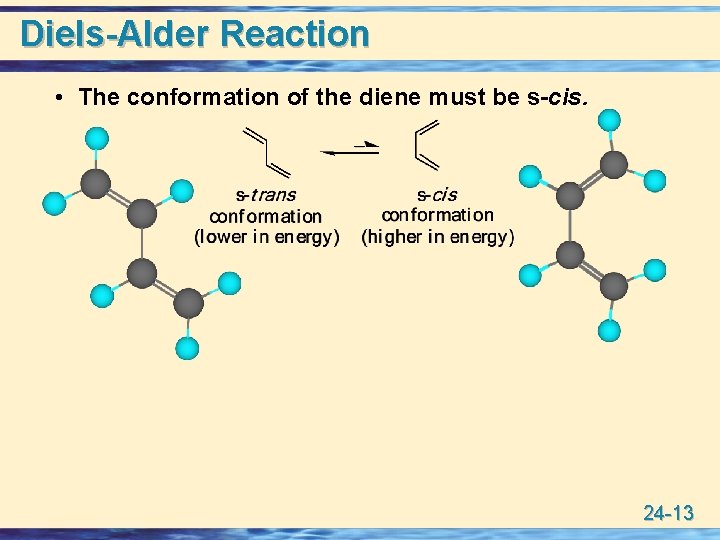
Diels-Alder Reaction • The conformation of the diene must be s-cis. 24 -13
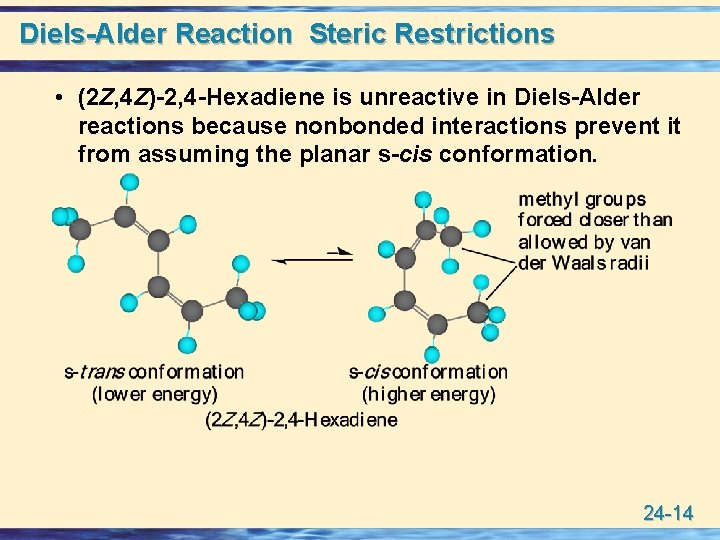
Diels-Alder Reaction Steric Restrictions • (2 Z, 4 Z)-2, 4 -Hexadiene is unreactive in Diels-Alder reactions because nonbonded interactions prevent it from assuming the planar s-cis conformation. 24 -14
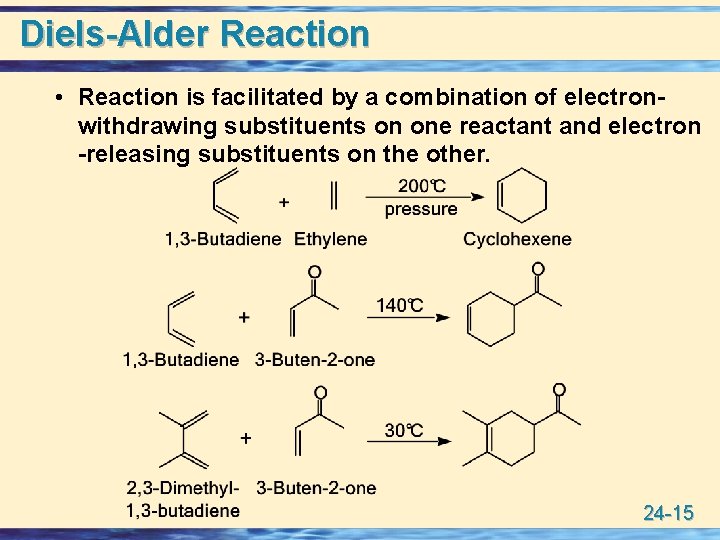
Diels-Alder Reaction • Reaction is facilitated by a combination of electronwithdrawing substituents on one reactant and electron -releasing substituents on the other. 24 -15

Diels-Alder Reaction 24 -16
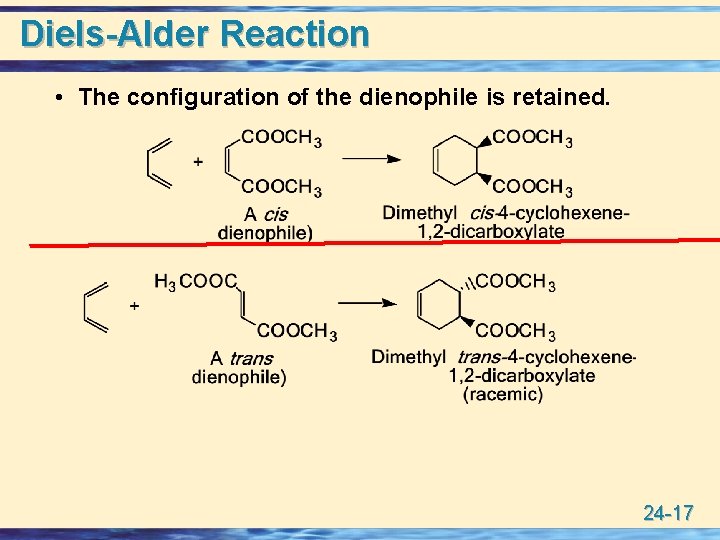
Diels-Alder Reaction • The configuration of the dienophile is retained. 24 -17

Diels-Alder Reaction u Mechanism • No evidence for the participation of either radical of ionic intermediates. • Chemists propose that the Diels-Alder reaction is a concerted pericyclic reaction. u Pericyclic reaction: reaction A reaction that takes place in a single step, without intermediates, and involves a cyclic redistribution of bonding electrons. u Concerted reaction: All bond making and bond breaking occurs simultaneously. 24 -18
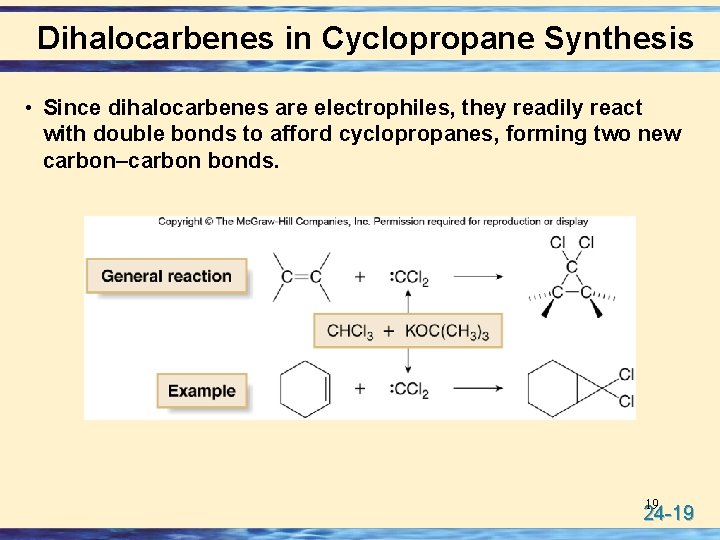
Dihalocarbenes in Cyclopropane Synthesis • Since dihalocarbenes are electrophiles, they readily react with double bonds to afford cyclopropanes, forming two new carbon–carbon bonds. 19 24 -19

Carbenes • A carbene, R 2 C: , is a neutral reactive intermediate that contains a divalent carbon surrounded by six electrons—the lone pair, and two each from the two R groups. • These three groups make the carbene carbon sp 2 hybridized, with a vacant p orbital extending above and below the plane containing the C and the two R groups. • The lone pair occupies an sp 2 hybrid orbital. Singlet carbene 20 24 -20
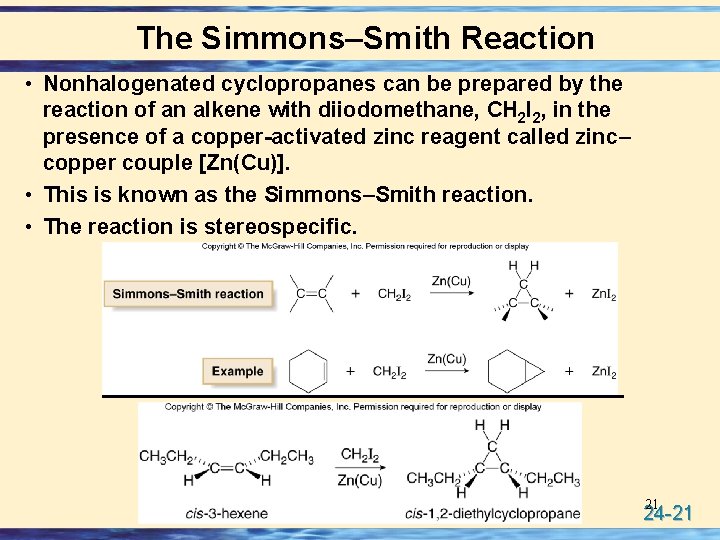
The Simmons–Smith Reaction • Nonhalogenated cyclopropanes can be prepared by the reaction of an alkene with diiodomethane, CH 2 I 2, in the presence of a copper-activated zinc reagent called zinc– copper couple [Zn(Cu)]. • This is known as the Simmons–Smith reaction. • The reaction is stereospecific. 21 24 -21
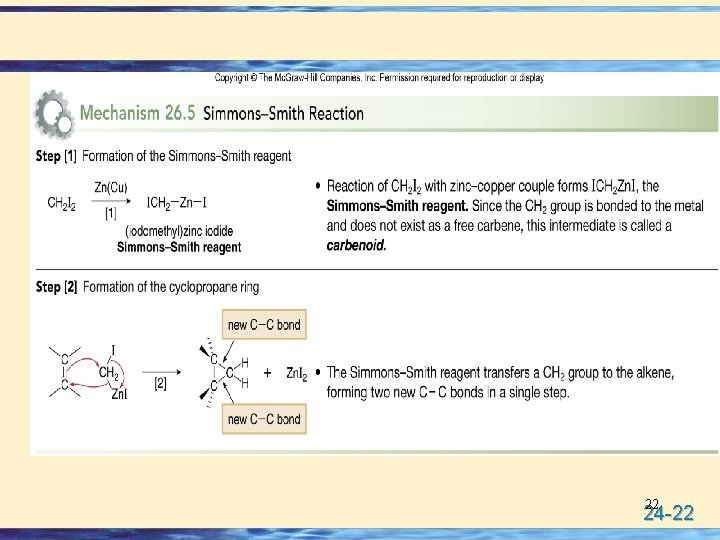
22 24 -22
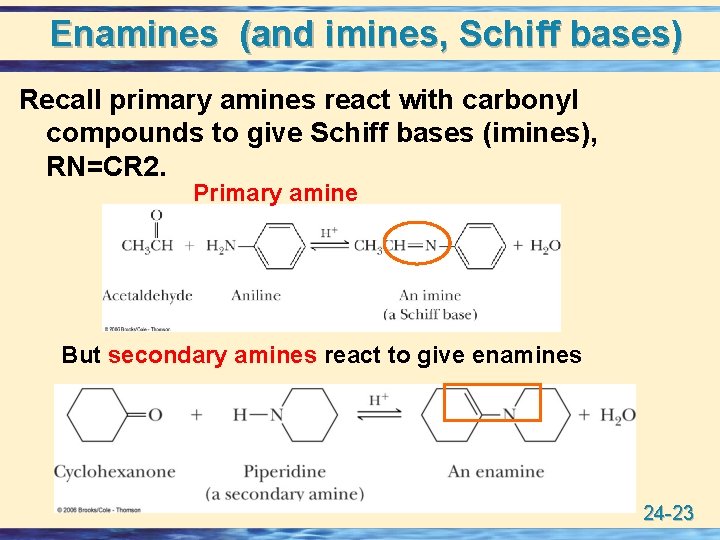
Enamines (and imines, Schiff bases) Recall primary amines react with carbonyl compounds to give Schiff bases (imines), RN=CR 2. Primary amine But secondary amines react to give enamines 24 -23

Formation of Enamines u Enamines are formed by the reaction of a 2° amine with the carbonyl group of an aldehyde or ketone. • The 2° amines most commonly used to prepare enamines are pyrrolidine and morpholine. 24 -24
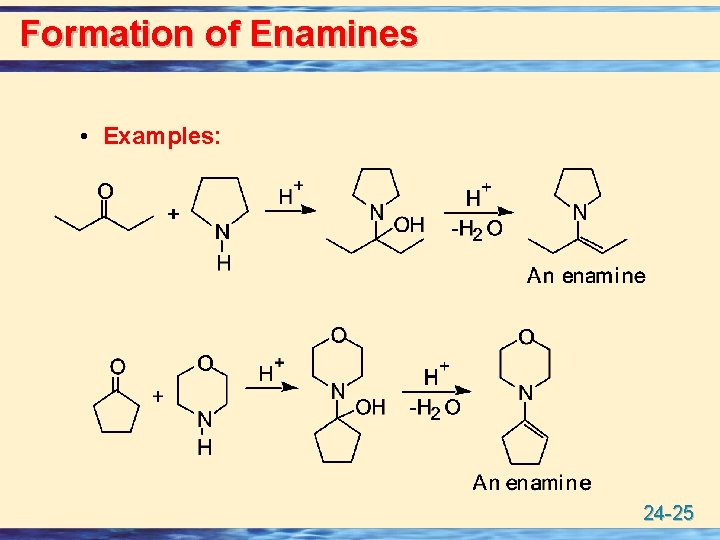
Formation of Enamines • Examples: 24 -25
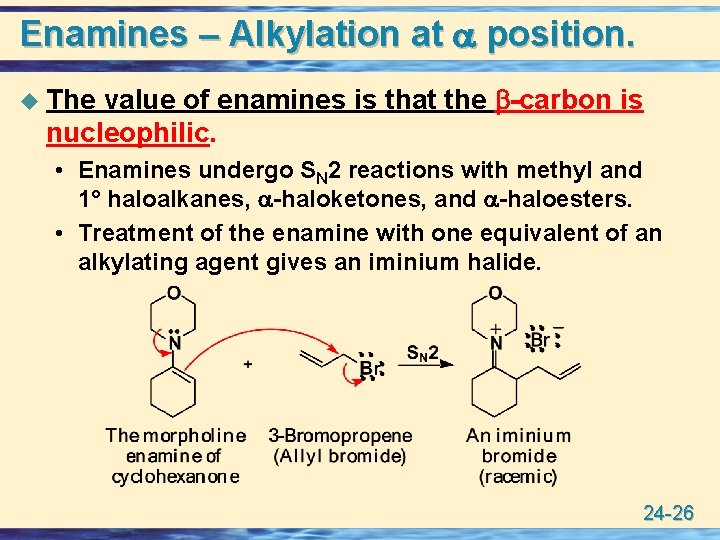
Enamines – Alkylation at position. value of enamines is that the -carbon is nucleophilic. u The • Enamines undergo SN 2 reactions with methyl and 1° haloalkanes, -haloketones, and -haloesters. • Treatment of the enamine with one equivalent of an alkylating agent gives an iminium halide. 24 -26
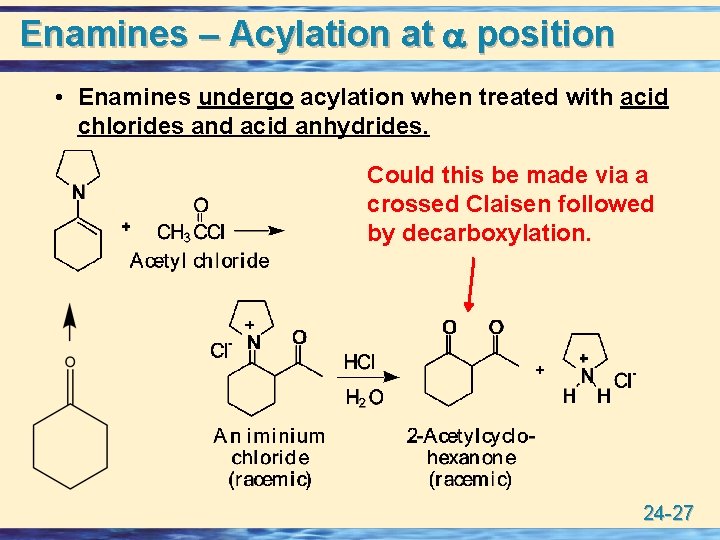
Enamines – Acylation at position • Enamines undergo acylation when treated with acid chlorides and acid anhydrides. Could this be made via a crossed Claisen followed by decarboxylation. 24 -27

THE END 24 -28
- Slides: 28