CArbon14 Source Term CAST 14 C in TRIGA

CArbon-14 Source Term CAST 14 C in TRIGA Irradiated Graphite and its Release under Alkaline Conditions Name: C. Bucur, C. Ichim, I. Florea, L. Bujoreanu, D. Diaconu Organisation: RATEN ICN Pitesti Date: 17. 01. 2018 The project has received funding from the European Union’s European Atomic Energy Community’s (Euratom) Seventh Framework Programme FP 7/2007 -2013 under grant agreement no. 604779, the CAST project.

Outlines RATEN ICN involvement in CAST WP 5 i-graphite samples available for experimental activities total 14 C measurement leaching test o 14 C release rate o Inorganic/organic 14 C released in alkaline conditions 2

RATEN ICN involvement in WP 5 Task 5. 1 - RATEN ICN reviewed: o the outcome of CARBOWASTE project in the Romanian context o the 14 C content and speciation and their correlation with impurity content and irradiation history in the MTR i-graphite. Task 5. 3 - RATEN ICN carried out leaching tests under aerobe and anaerobe conditions to assess the 14 C release under alkaline conditions Results included in D 5. 5 “Review of current understanding of inventory and release of 14 C from irradiated graphite” Results are reported in D 5. 10 “Final report on C-14 release and inorganic/ organic ratio in leachates from TRIGA irradiated graphite” 3

i-graphite samples (1/2) i-graphite from TRIGA thermal column o Block of 96 rectangular bricks of graphite encased in Aluminum o Each brick is composed by graphite pieces with different geometries o The graphite was imported in the ‘ 50 s from a UK producer - documents of its origin were lost o Irradiation power: ~ 10 MW o Strong neutron energy and flux variations CARBOWASTE project: experimental and modeling activities regarding the radionuclide inventory in TRIGA i-graphite No leaching tests were performed before CAST on i-graphite from TRIGA research reactor, ICN Geometry of a graphite brick 4
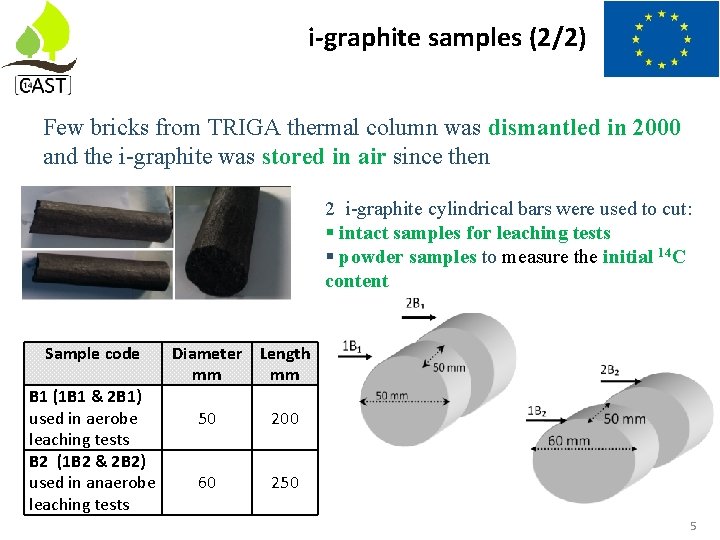
i-graphite samples (2/2) Few bricks from TRIGA thermal column was dismantled in 2000 and the i-graphite was stored in air since then 2 i-graphite cylindrical bars were used to cut: § intact samples for leaching tests § powder samples to measure the initial 14 C content Sample code B 1 (1 B 1 & 2 B 1) used in aerobe leaching tests B 2 (1 B 2 & 2 B 2) used in anaerobe leaching tests Diameter Length mm mm 50 200 60 250 5

Total 14 C measurement 6

Methodology for total 14 C measurement (1/2) Non-catalytic combustion by flame oxidation method i-graphite samples are combusted in an oxygen-enriched atmosphere with a continuous flow of oxygen using Sample Oxidizer, Model 307 Perkin. Elmer® Any hydrogen present is oxidized to H 2 O Any carbon present is oxidized to CO 2 7

Methodology for total 14 C measurement (2/2) - condensed in a cooled coil, washed into a counting vial where it is mixed with LSC cocktail (Monophase®S) 14 CO - trapped by vapour-phase reaction with an amine 2 (Carbo-Sorb® E ) and the resulting product (carbamate) is mixed with the LSC cocktail (Perma-fluor® E+) directly in the counting vial. At the end of the combustion cycle, two separate samples - a 3 H sample and a 14 C sample - are trapped at ambient temperature, minimizing the cross contamination 14 C and 3 H activity is measured by LSC using Tri-Carb® 3110 TR allow in ULL counting mode a count rate between 1‑ 20 CPM above the Bkg 3 H 2 O 8

Tests on labeled graphite Tests carried out using virgin graphite labeled with 14 C as well as other gamma emitters to optimize the mineralization process with total recovery of 14 C to assess the presence of potential interfering radionuclides in the 14 C vials The protocol for complete graphite oxidation was selected: § § ~ 0. 15 g of graphite powder 10 ml Carbosorb. E 10 ml Permafluor. E+ ~ 2 min combustion time recovery - 97% MEM < 0. 04% no gamma emitters identified in the 14 C vials 14 C 9

Total 14 C measurement in TRIGA i-graphite 4 powder samples from B 1 i-graphite bar 12 powder samples from B 2 i-graphite bar 14 C uniform distributed in the two cylindrical bars 14 C content: B 1: 450. 75 Bq/g B 2: 96. 11 Bq/g 10

Leaching experiments 11

Leaching tests conditions Aerobe conditions 2 samples from B 1 bar surface area: 117. 75 cm 2 volume: 98. 13 cm 3 mass: 172. 72 / 168. 24 g leachate volume: 1118 cm 3 (Vleachant/Sspecimen = 0. 1 m) room temperature: 25± 50 C 0. 1 M Na. OH (p. H=13) semi-dynamic: 30 ml of leachate removed for 14 C measurement and 30 ml of fresh Na. OH solution added at each sampling time 14 C measurement in leachate solution by LSC using Tri-Carb® 3110 TR 12

Leaching tests conditions Anaerobe conditions 2 samples from B 2 bar surface area: 150. 72 cm 2 volume: 141. 30 cm 3 mass: 250. 10 / 249. 70 g leachate volume: 1500 cm 3 (Vleachant/Sspecimen = 0. 1 m) room temperature: 25± 50 C 0. 1 M Na. OH (p. H=13) stirring before each sampling step N 2 flushing at the beginning of test and after each leachate renewal step; semi-dynamic: 50 ml of leachate removed for 14 C measurement and 50 ml of fresh Na. OH solution added at each sampling time 14 C measurement in leachate solution by LSC using Tri-Carb® 3110 TR 13

Evaluation of results cumulative leaching fraction in the n-step of the leaching rate in the n-step of the assay, cm/day, ao - the initial amount of 14 C in the i-graphite specimen, Bq an - amount of a 14 C that has been released from the i-graphite specimen in the n-step of the leaching test, Bq tn - duration of the n-step of the assay, days V - volume of the i-graphite specimen, cm 3 S - apparent or geometrical surface of the i-graphite specimen, cm 2 K - characteristic constant of the leaching test, cm/Bq. 14

Leaching tests results 15
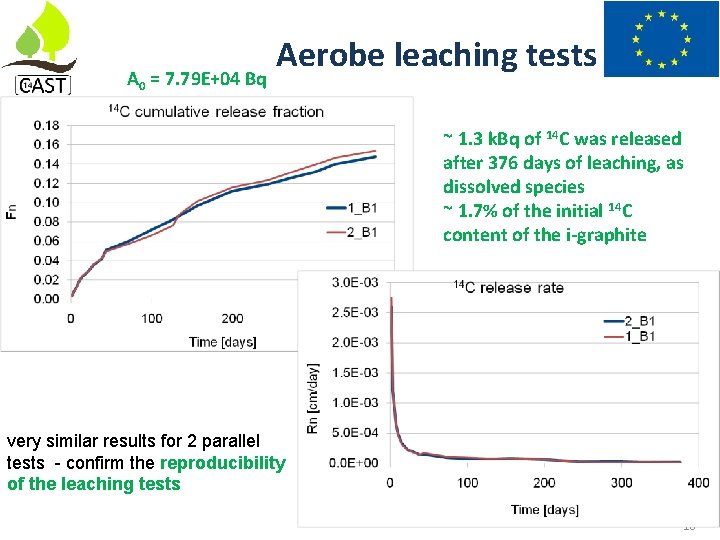
A 0 = 7. 79 E+04 Bq Aerobe leaching tests ~ 1. 3 k. Bq of 14 C was released after 376 days of leaching, as dissolved species ~ 1. 7% of the initial 14 C content of the i-graphite very similar results for 2 parallel tests - confirm the reproducibility of the leaching tests 16
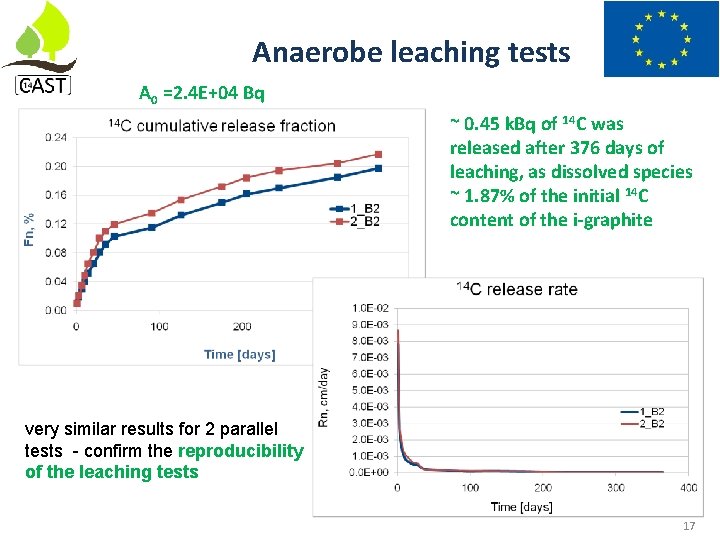
Anaerobe leaching tests A 0 =2. 4 E+04 Bq ~ 0. 45 k. Bq of 14 C was released after 376 days of leaching, as dissolved species ~ 1. 87% of the initial 14 C content of the i-graphite very similar results for 2 parallel tests - confirm the reproducibility of the leaching tests 17

Inorganic and organic 14 C measurement

Inorganic/organic 14 C Sequential extraction (combination of acid stripping and wet oxidation) from the same SIERs (A. Magnusson et all. , 2008) 1 st step: Acid stripping – inorganic fraction of 14 CO 2 is absorbed in the first set of alkaline washing bottles 2 nd step: Wet oxidation – organic fraction of 14 C - K 2 S 2 O 8 and Ag. NO 3 combined with heating (~95 C) organic 14 C are decomposed and it is absorbed in a second set of alkaline washing bottles placed after the catalytic furnace - catalytic furnace (holding Pt + Cu. O catalyst inside a quartz tube) – for complete oxidation of any reduced 14 C compounds vacuum of - 0. 2 bar: to avoid any gas release from the system N 2 carrier gas : flow rate 60 - 80 ml/hour 19
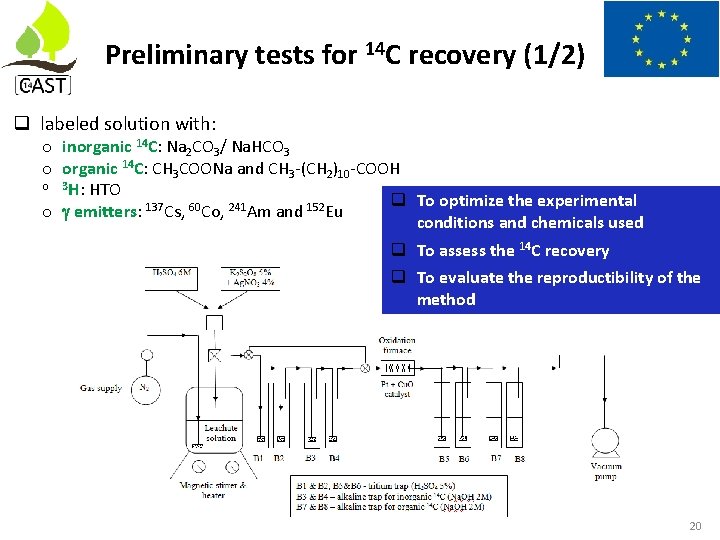
Preliminary tests for 14 C recovery (1/2) labeled solution with: o inorganic 14 C: Na 2 CO 3/ Na. HCO 3 o organic 14 C: CH 3 COONa and CH 3 -(CH 2)10 -COOH o 3 H: HTO To optimize the experimental o emitters: 137 Cs, 60 Co, 241 Am and 152 Eu conditions and chemicals used To assess the 14 C recovery To evaluate the reproductibility of the method 20

Preliminary tests for 14 C recovery (2/2) Acid dissolution: 10 ml HNO 3 Wet oxidation: 10 K 2 S 2 O 8 5% + 4 Ag. NO 3 (3 cycles) inorganic 14 C recovery: 98% (96 – 100%) organic 14 C recovery: 96% MEM: < 1% reproductibility: the standard deviation of the results of six identical tests were below 15% good efficiency for 14 C purification o no gamma emitters identified in solution sampled from alkaline gas washing bottles 21

Inorganic & organic 14 C released in alkaline conditions The ratio between inorganic and organic 14 C release during the leaching test is almost constant during the test Both organic and inorganic 14 C species are released in alkaline conditions Aerobe conditions: more inorganic than organic 14 C was released inorganic 14 C: 66% - 70% from the total 14 C released as dissolved species, with an average of 67. 89% organic 14 C: 30% - 34% from the total 14 C released as dissolved species, with an average of 32. 11%. Anaerobe conditions: more organic than inorganic 14 C was released inorganic 14 C: 32% - 38% from the total 14 C released as dissolved species, with an average of 35. 24% organic 14 C: 61% and 66% from the total 14 C released as dissolved species, , with an average of 64. 57% 22

Conclusions In alkaline conditions, very low amount of 14 C is available for release from the i-graphite aerobic conditions: ~ 1. 70 % of the initial 14 C content of the i-graphite subject of the leaching test was released as dissolved species anaerobic conditions: ~ 1. 85 % of the initial 14 C content of the i-graphite subject of the leaching test was released as dissolved species the leaching rates are high in the first days of immersion and decrease after that, indicating a two stage process: an initial quick release less than 0. 09 % of inventory/day for the first 40 days followed by a slower release rate (around 0. 004 % of inventory/day). more inorganic 14 C was released under aerobe conditions ~ 68% inorganic 14 C and 32% organic 14 C more organic 14 C was released under anaerobe conditions ~ 35% inorganic 14 C and 65% organic 14 C 23

THANK YOU FOR YOUR ATTENTION! 24
- Slides: 24