Carbon Saturated Hydrocabons Carbon makes four bonds Alkanes

Carbon Saturated Hydrocabons

Carbon makes four bonds.
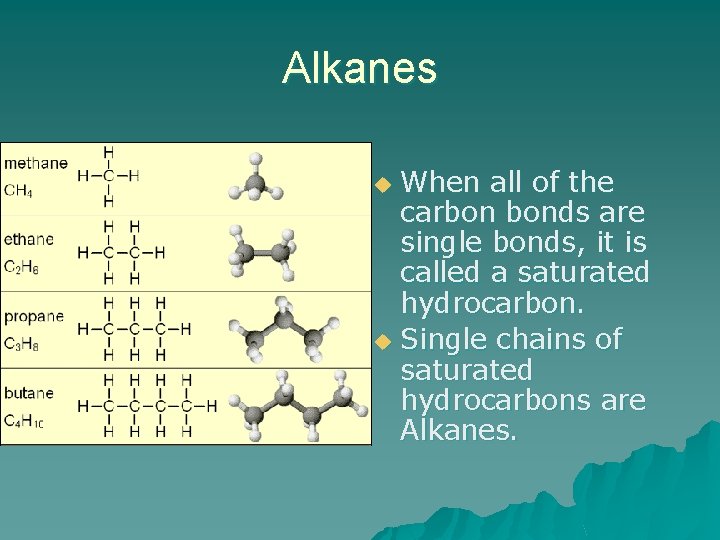
Alkanes When all of the carbon bonds are single bonds, it is called a saturated hydrocarbon. u Single chains of saturated hydrocarbons are Alkanes. u

IUPAC nomenclature u IUPAC stands for the INTERNATIONAL UNION OF PURE AND APPLIED CHEMISTRY u It is the accepted form for naming chemicals. u Many compounds have more than one name. u We will accept only IUPAC in this class
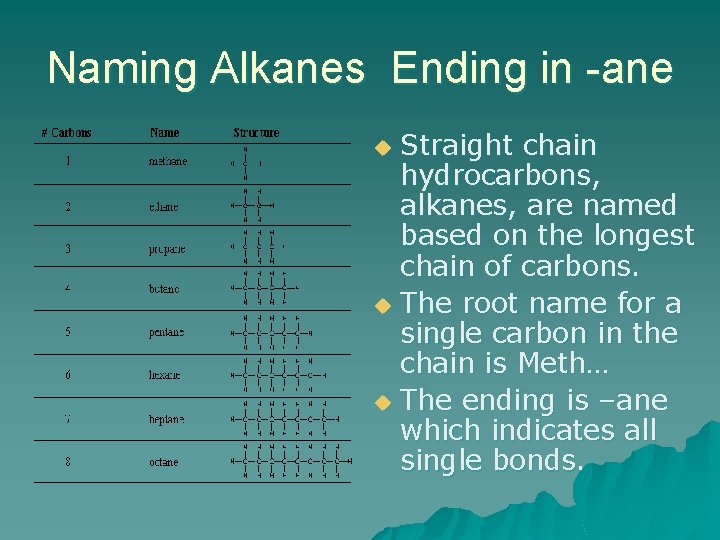
Naming Alkanes Ending in -ane Straight chain hydrocarbons, alkanes, are named based on the longest chain of carbons. u The root name for a single carbon in the chain is Meth… u The ending is –ane which indicates all single bonds. u
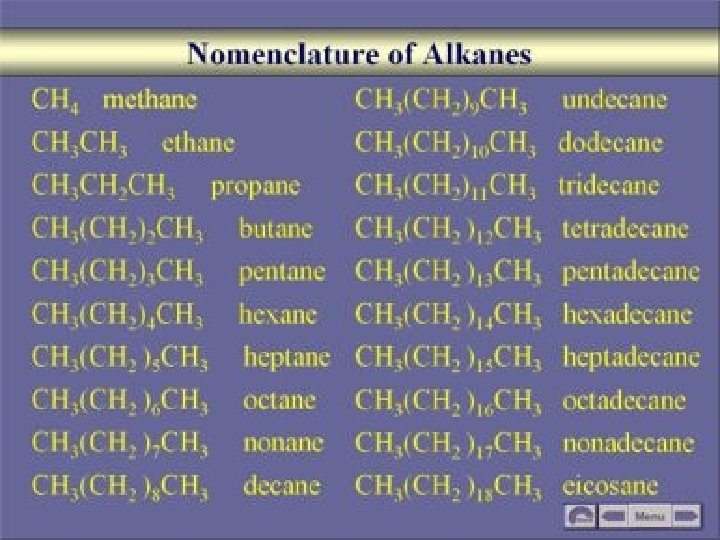

Determining the longest chain: Counting carbons of the longest continuous unbroken chain of carbons. u Bends in the chain do not count as a break. u
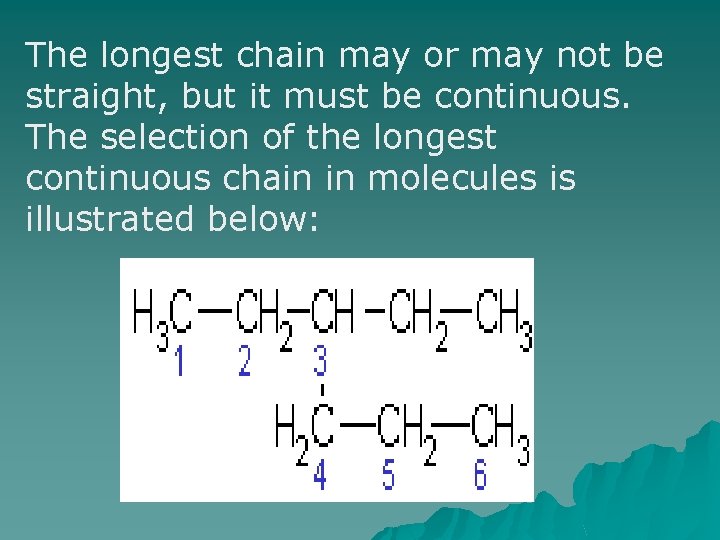
The longest chain may or may not be straight, but it must be continuous. The selection of the longest continuous chain in molecules is illustrated below:

How many in the chain and Name that chain! A. octane u B. octane u C. octane u

Traits of Alkanes u The more carbons in the chain, the heavier the molecule. u What do you think happens to the boiling point of alkanes as the chain grows larger?
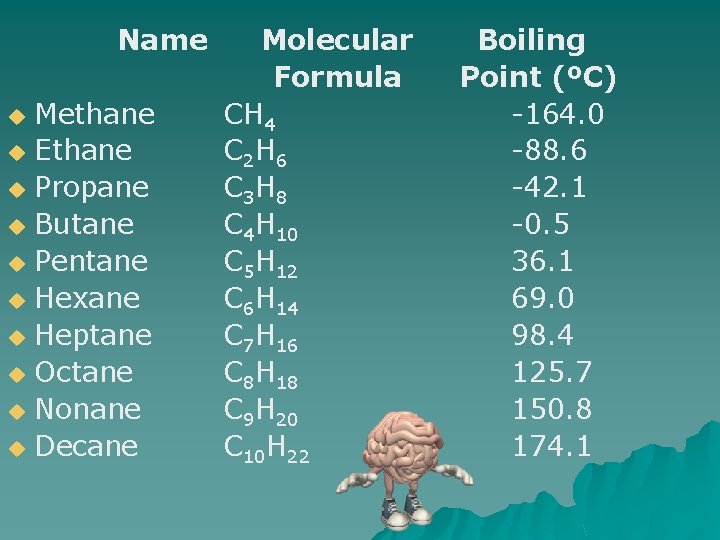
Name Methane u Ethane u Propane u Butane u Pentane u Hexane u Heptane u Octane u Nonane u Decane u Molecular Formula CH 4 C 2 H 6 C 3 H 8 C 4 H 10 C 5 H 12 C 6 H 14 C 7 H 16 C 8 H 18 C 9 H 20 C 10 H 22 Boiling Point (ºC) -164. 0 -88. 6 -42. 1 -0. 5 36. 1 69. 0 98. 4 125. 7 150. 8 174. 1

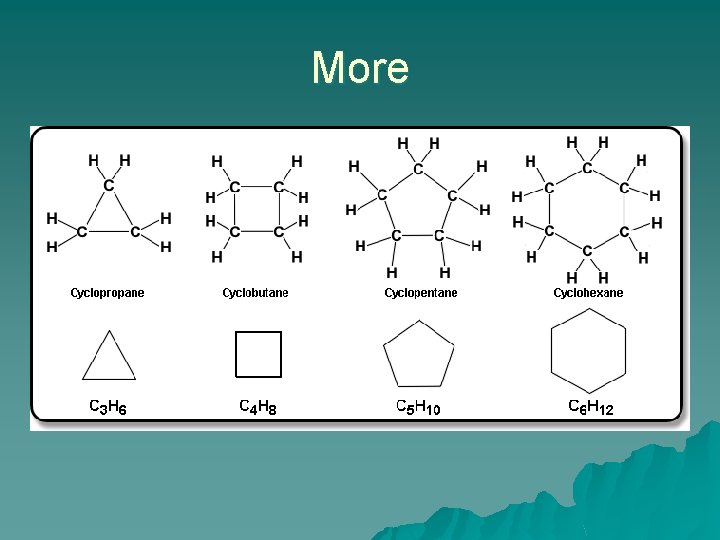
Cyclic Alkanes Rings formed by the hydrocarbon chain. u The name is based on the number of carbons in the ring. u A is cyclopropane u B is cyclobutane u C is cyclohexane u
More
- Slides: 13