Carbon nanomaterials Module 3 Carbon 3 DDiamond Hybrid

Carbon nanomaterials Module 3
Carbon 3 D-Diamond
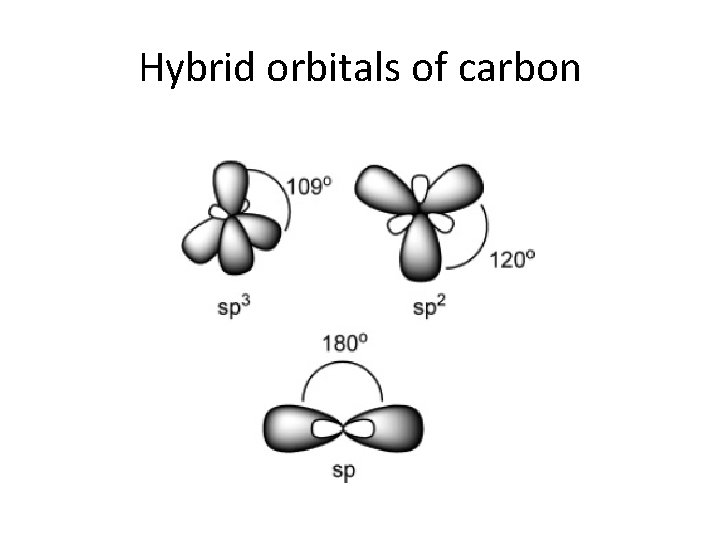
Hybrid orbitals of carbon
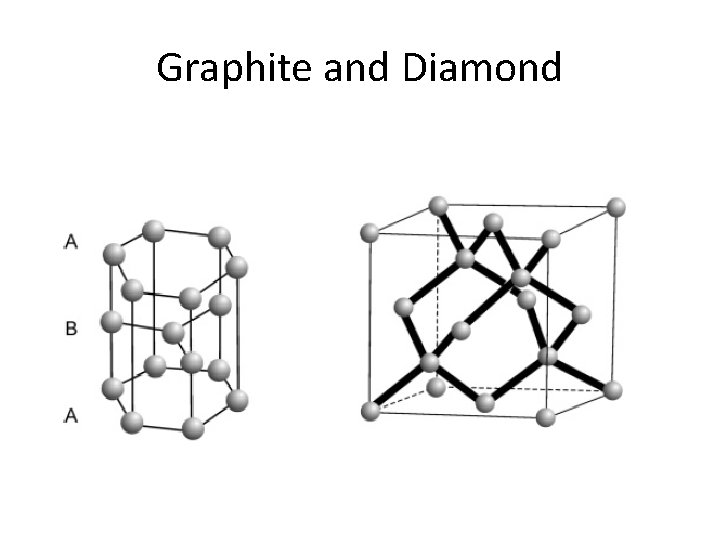
Graphite and Diamond
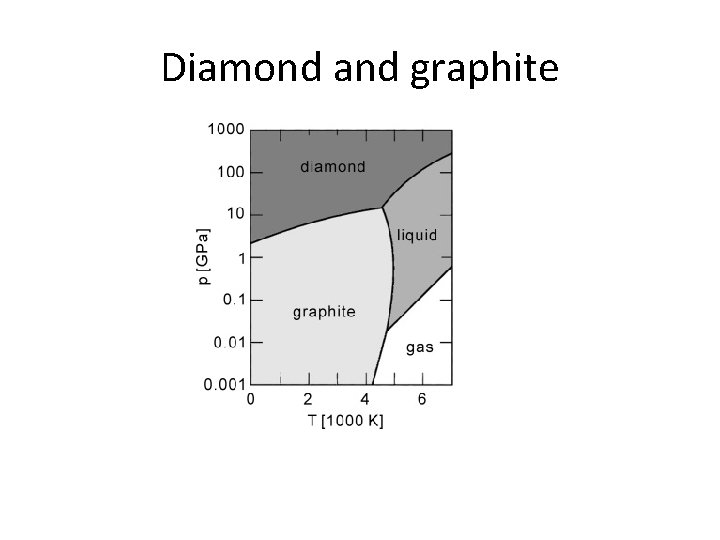
Diamond and graphite
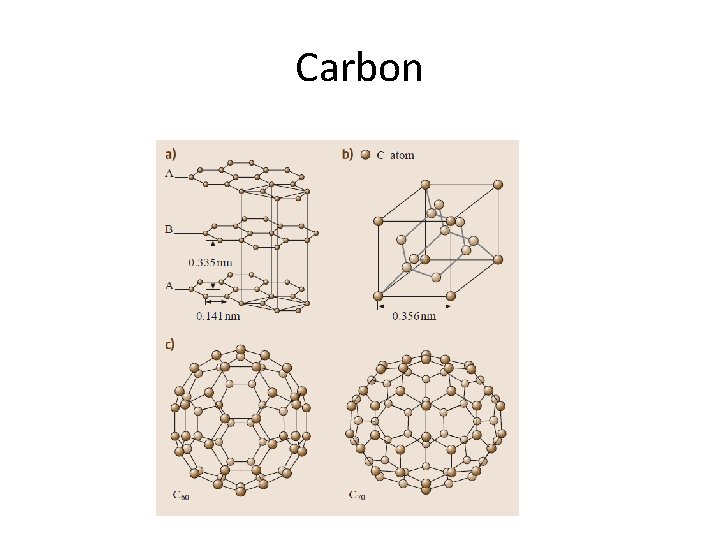
Carbon

Fullerenes

Fullerenes • Fullerenes are a modification of carbon with cage like structure • They are spherical and symmetrical new molecular allotrope of carbon • Symmetrical Icosahedral buckminsterfullerene (C 60 – Ih) • Cage diameter are in nm range ( 0. 7 nm in C 60) • Interesting chemical and physical properties • Functionalisation - exohedral, endohedral, hetero, open structures • Various applications due to their interesting structural, electronic properties

Fullerenes • Fullerenes are not just C 60 , there are higher members. • C 70 is next higher stable fullerene – it has extra 6 membered rings and same 5 membered rings • Higher members are C 76, C 78, C 84, C 90 • Hetero, Endohedral, Exohedral fullerenes
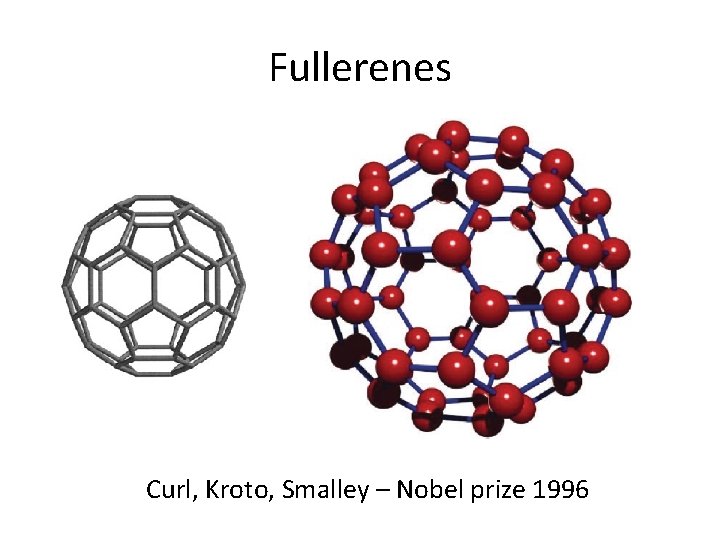
Fullerenes Curl, Kroto, Smalley – Nobel prize 1996

Stephanopyxis turris Biological structure similar to fullerenes
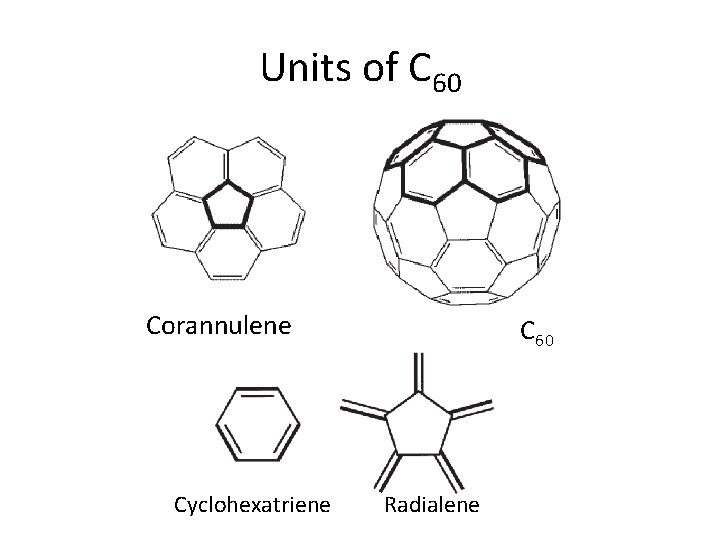
Units of C 60 Corannulene Cyclohexatriene C 60 Radialene
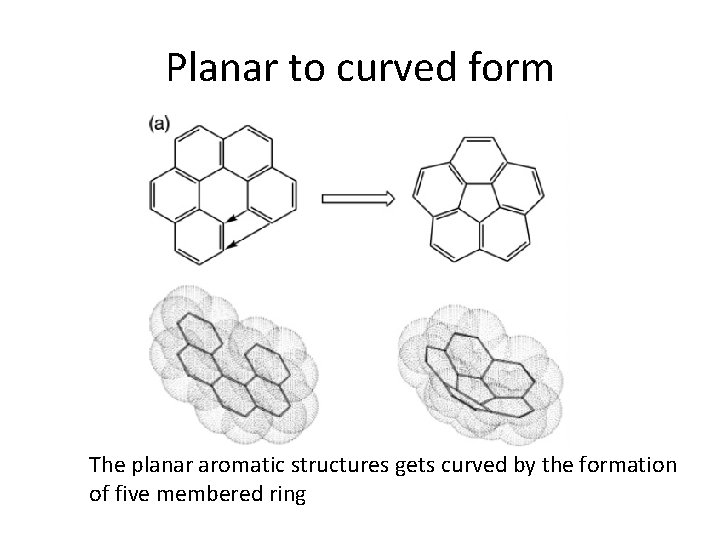
Planar to curved form The planar aromatic structures gets curved by the formation of five membered ring
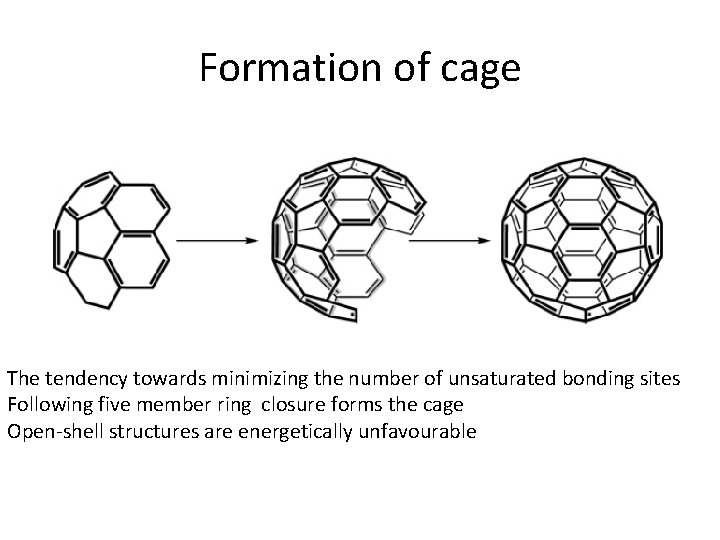
Formation of cage The tendency towards minimizing the number of unsaturated bonding sites Following five member ring closure forms the cage Open-shell structures are energetically unfavourable
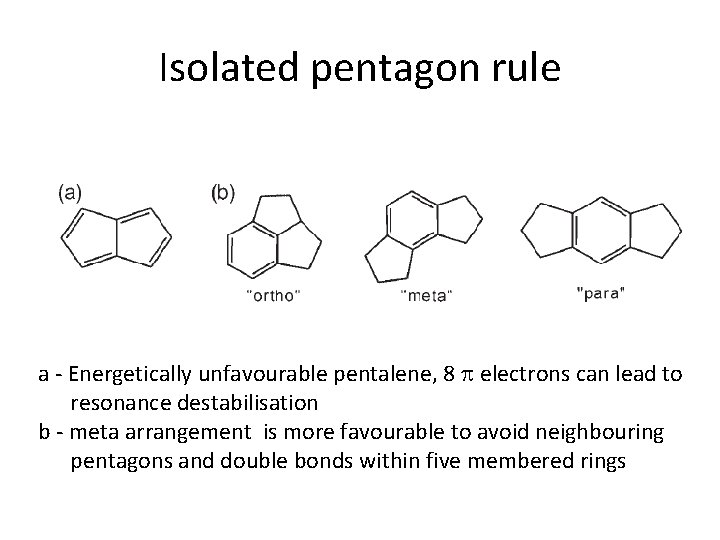
Isolated pentagon rule a - Energetically unfavourable pentalene, 8 electrons can lead to resonance destabilisation b - meta arrangement is more favourable to avoid neighbouring pentagons and double bonds within five membered rings

Structure and bonding • C 60 has 12 five and 20 six membered rings • Not all the C-C bonds of C 60 is of same length • The double bonds are localized in 6 membered rings • Five membered rings are evenly distributed on the surface and isolated (Isolated pentagon rule) • Diameter of C 60 molecule is 0. 702 nm • High symmetry molecule – easily identified by spectroscopy

Bonds in C 60 Bonds between two 6 membered rings – 6, 6 bond Bonds between 5 and 6 membered rings – 5, 6 bond Double bonds are preferably located in 6 membered rings and it is energetically unfavourable in 5 membered ring
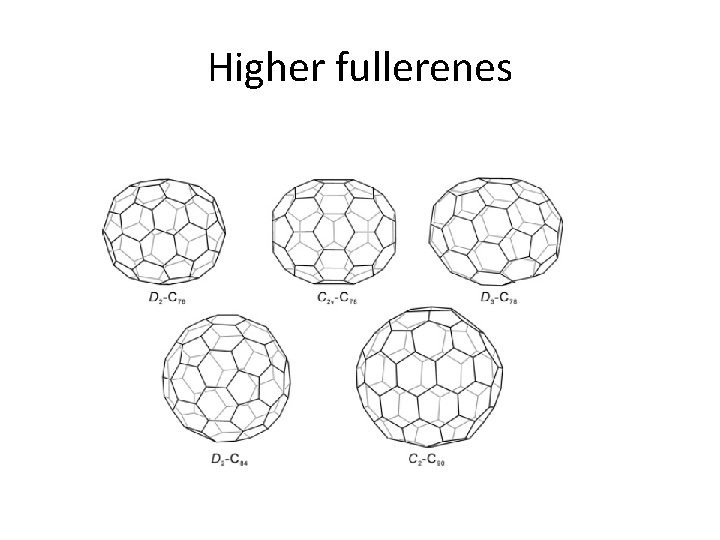
Higher fullerenes
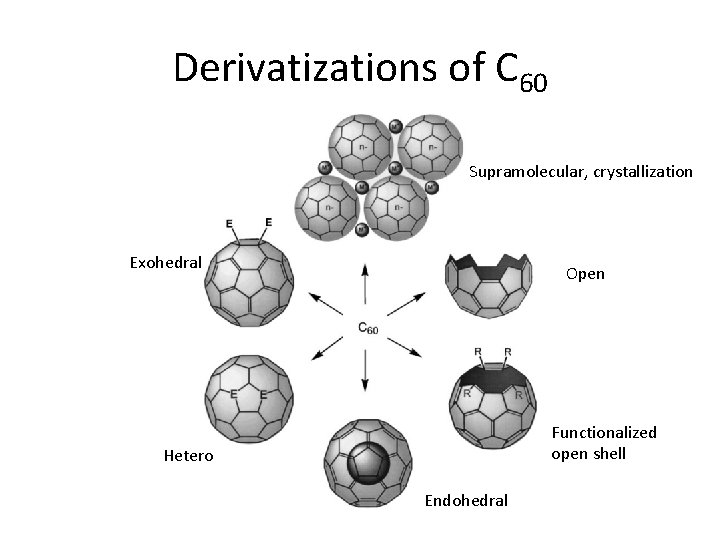
Derivatizations of C 60 Supramolecular, crystallization Exohedral Open Functionalized open shell Hetero Endohedral
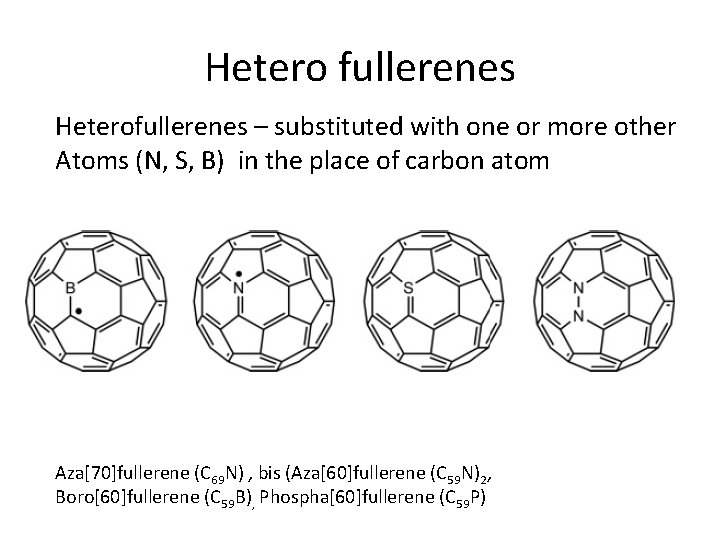
Hetero fullerenes Heterofullerenes – substituted with one or more other Atoms (N, S, B) in the place of carbon atom Aza[70]fullerene (C 69 N) , bis (Aza[60]fullerene (C 59 N)2, Boro[60]fullerene (C 59 B), Phospha[60]fullerene (C 59 P)

Exohedral fullerenes • Hydrogenated, halogenated, other functionalities by addition reactions • Exohedral fullerenes shows higher solubility • Functionalization are useful to form suprmolecular compounds C 60 H 36
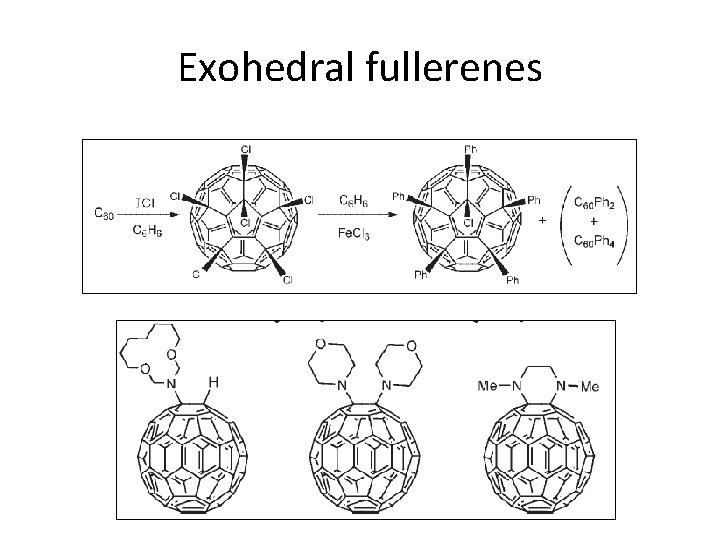
Exohedral fullerenes

Endohedral fullerenes • Fullerenes filled with atoms, molecules inside the cavity are called endohedral fullerenes • Metallofullerene - M@Cn ( M -Li, Ca, Sc, Y, La, Ce, Eu) • Nonmetallic endohedral fullerenes ( N, P) • Noble gas containing fullerenes – X@C 60 ( X = He, Ne, Ar, Kr, Xe)

Preparation • • • Thermal method – pyrolysis of hydrocarbons (HCs) Combustion – partial combustion of HCs Arc discharge method – graphite Resistance heating method Rational synthesis Pulse laser beam method - Graphite electrodes

Thermal method - pyrolysis • Poly aromatic hydrocarbons(PAH) are suitable for synthesis of fullerenes by pyrolysis • Here the structural elements have the structural framework of fullerene cage • Hydrocarbons already consisting of 5 and 6 membered rings • Napthalene, corannulenes, PAH pyrolysed at 1000 o. C in an inert atmosphere.
- Slides: 25