Carbon Dioxide Transport The solubility of CO 2

Carbon Dioxide Transport The solubility of CO 2 in blood is about 20 times that of O 2 at equal partial pressures. Fate of CO 2 in blood In plasma 1. Dissolved 2. Formation of carbamino compounds with plasma protein 3. Hydration, H+ buffered, HCO 3– in plasma.

Carbon dioxide transport
104

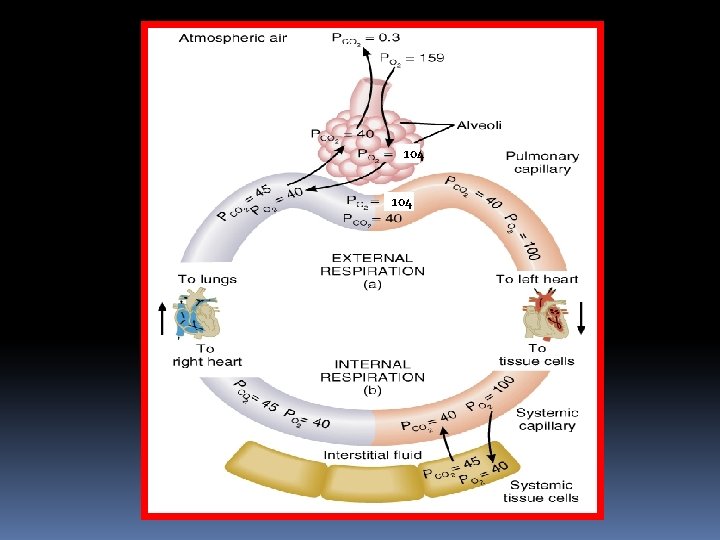
DIFFUSION OF Carbon dioxide From the BLOOD into the ALVEOLI From the ALVEOLI into the ATMOSPHERE

CO 2 Transport Arterial blood – CO 2 content (concentration) – 48 ml/dl CO 2 tension -- PCO 2 of 40 mm Hg Venous blood– blood CO 2 content (concentration) – 52 ml/dl CO 2 tension -- PCO 2 of 45 mm Hg A-V CO 2 difference = 4 ml/dl

CO 2 is transported from the tissues to the lungs depends on Blood O 2 content ii) number of RBC present in the blood iii) the amount of reduced Hb Transport of Co 2 takes place in three steps Uptake by Co 2 by blood from the tissue Transport in the blood Delivery to the lung. CO 2 present in the blood in 3 forms 1. Simple solution -- 7% 2. Bicarbonate form (HCO 3) – 70% 3. Carbamino-compound -- 23% (Carbamino-Hemoglobin)

Uptake by lung due to pressure gradient intracellular 46 mmhg Interstitial fluid 45 mm Hg Arterial blood 40 mm. Hg From the tissue to the blood it diffuses due to pressure this is also facilitated by release of O 2 to the tissue. When Co 2 enters the blood it either enter to the plasma or RBC

Out of 4 ml of co 2 In dissolved form (0. 3 ml/dl) As carbamino compounds(0. 7 ml/dl) As bicarbonate (3 m. L/dl) In dissolved form In plasma Co 2 ₊ H 20 H 2 co 3 In RBC Co 2 ₊ H 20 CA H 2 co 3

Simple solution (Dissolved form in plasma) Carbon dioxide diffuses into blood and dissolves in the fluid of plasma forming a simple solution (3 ml) Accounts for about 7% of CO 2 transport Solubility of CO 2 is 20 times of O 2

Carbamino Hemoglobin

In plasma Directly combines with plasma proteins and forms carbamino protein In RBC It forms carbaminohemoglobin

Formation of Carbamino hemoglobin Some of the CO 2 enter into the RBC binds with the amino groups of the Hb to form carbamino hemoglobin. CO 2 + Hb NH 2 Hb NH COOH = Hb NH COO + H+ CO 2 + R-NH 2 R-N-H-COOH (any protein) This rapid reaction does not require any catalyst. This type accounts of 15 to 30% (about 24%) of total CO 2 transport in the blood.

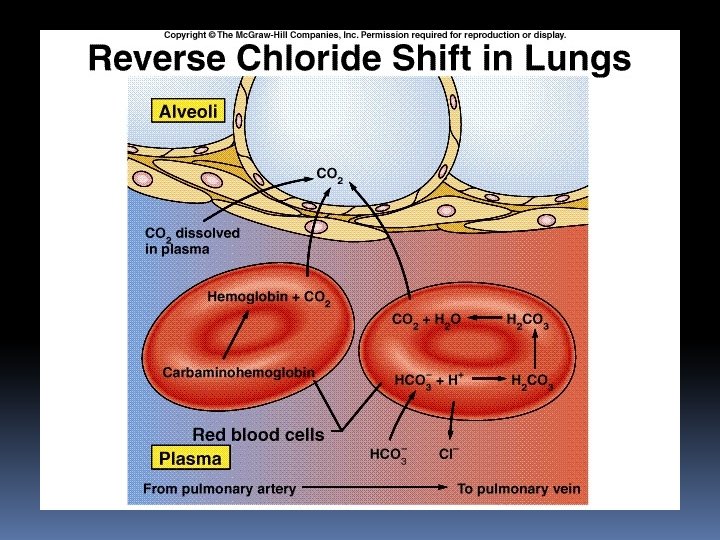
Bicarbonate form PLASMA RBC Co 2 H 20 + Co 2 Carbonic anhydrase H 20 INTERSTITIAL FLUID Co 2 H 2 Co 3 Cl- Hco 3 H+ Hb Co 2 Na+ HHb CELL Na. Cl Na+ Hco 3

Account for 70% of CO 2 transport Formation of HCO 3 is very slow in plasma & Rapid in RBC due to presence of Carbonic anhydrase (CA) 2/3 rd of HCO 3 formed in RBC enter into plasma in exchange of Cl- through ionic channel formed by band-3 membrane protein [chloride shift] occurs rapidly & completed within one second

CO 2 -Dissociation curve

Two separate Co 2 dissociation curves One curve Po 2 100 mm in the lung Curve Po 2 40 mmhg tissue capillaries PCO 2 of 45 mm in the tissues causes 52 volume % co 2 to combine with Hb In the lung PCo 2 falls to 40 mm. Hg and Po 2 increases If the CO 2 dissociation did not shift because of Halden’s effect the Co 2 content of blood would have been fall only 50 voulme % which would be a loss of only 2 volume%

Mechanism of Haldane effect Combination of oxygen with hemoglobin in the lungs cause the hemoglobin to becomes a stronger acid. Therefore: The increased acidity of the hemoglobin also causes it to release an excess of hydrogen ions Oxygenation of Hb shifts the carbon dioxide curve to shift to right. Loading of oxygen causes unloading of carbon dioxide at lung level. 17

Haldane’s effect Binding of O 2 with Hb tends to displace the CO 2 from the blood Takes place at lung level (promotes carbon dioxide transport)

1. The combination of oxygen with Hb causes the Hb to become a stronger acid. ØThe more highly acidic Hb has less tendency to combine with carbon dioxide to form carbaminohemo-globin ØThus displacing carbon dioxide

2. The increased acidity of Hb causes it to release an excess of hydrogen ions Ø hydrogen ions bind with bicarbonate ions to form carbonic acid Ø carbonic acid dissociates into water and carbon dioxide Ø carbon dioxide is released from Øthe blood into the alveoli

Therefore, in the tissue capillaries the haldane’s effect causes increased pick up of Co 2 because of O 2 removal from the Hb In the lungs it causes increased release of Co 2 because of O 2 pickup by the Hb.

However increase PO 2 in the lung shift the Co 2 dissociation curve from dash curve to solid curve so that Co 2 content falls to 48 volume % This accounts an additional 2 volume% loss of Co 2 Thus halden’s effect doubles amount of Co 2 released from the blood in the lungs Approx. doubles pickup in the tissues.

DIFFUSION OF CARBON DIOXIDE FROM BLOOD INTO THE TISSUES TISSUE PCO 2 = 46 mm Hg Arterial end PCO 2 = 40 mm Hg CO 2 Content = 48 ml% CO 2 Capillary Venous end PCO 2 = 46 mm Hg CO 2 Content = 52 ml%


Chloride shift mechanism 70% of carbon dioxide is transported in this form. Hamburger shift RBC swells up and Hct of venous blood is more. This facilitates release of oxygen from Hb into tissue.


Delivery of carbon di oxide to the lungs Pressure gradient Haldane effect. Pressure gradient of pco 2 Venous blood 45 mm Hg Alveoli 40 mm Hg Oxygenation of Hb favours unloading of carbondioxide.
- Slides: 28