Carbon dioxide capture from automobile flue gas Materials

Carbon dioxide capture from automobile flue gas: Materials and technology potentials Asmaa Khuwaireh Aseel Mousa Muna Shubaiteh Supervisor: Prof. Hamdallah Bearat

Table of content: Introduction Reactors Experiment and Result System I: Compressed Air system System II: Pure CO 2 system Conclusions and Recommendation


What is the problem?

How we can solve it?
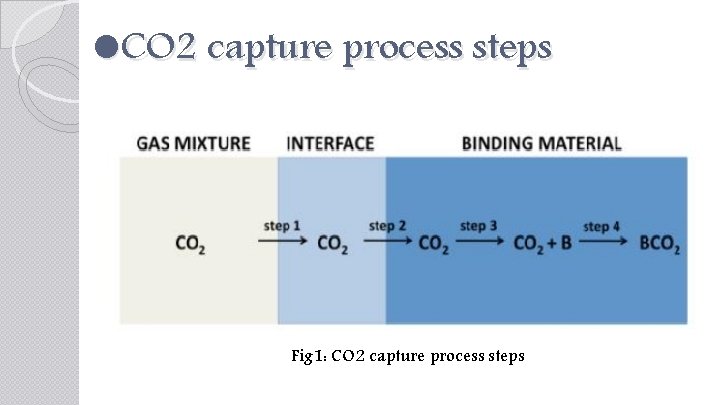
CO 2 capture process steps Fig 1: CO 2 capture process steps

We used Sodium hydroxide as binding material to capture CO 2 � Nao. H is a white solid with a melting point of 318°C. � Sodium hydroxide crystals absorb water very easily. � Solid sodium hydroxide burns the skin. � Solid sodium hydroxide reacts easily with CO 2.

Fig 2 : Sodium Hydroxide Na. OH

Na. OH & CO 2 reactions 2 Na. OH + CO 2 → Na 2 CO 3 + H 2 O ∆H = -169. 8 k. J/mol CO 2 Na. OH+ CO 2� Na. HCO 3 ∆H = -127. 44 k. J/mol CO 2 These reactions are very exothermic.
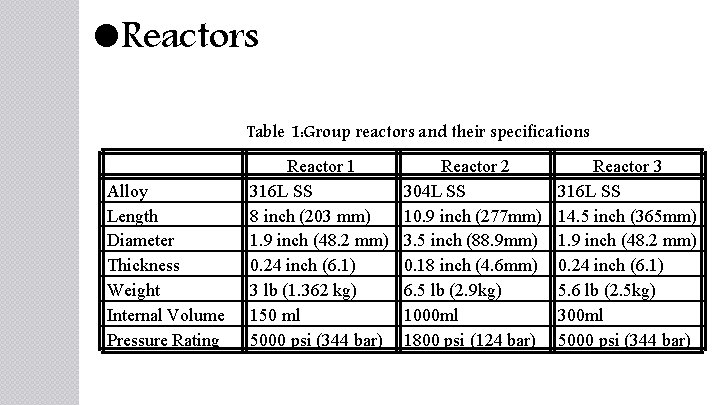
Reactors Table 1: Group reactors and their specifications Alloy Length Diameter Thickness Weight Internal Volume Pressure Rating Reactor 1 316 L SS 8 inch (203 mm) 1. 9 inch (48. 2 mm) 0. 24 inch (6. 1) 3 lb (1. 362 kg) 150 ml 5000 psi (344 bar) Reactor 2 304 L SS 10. 9 inch (277 mm) 3. 5 inch (88. 9 mm) 0. 18 inch (4. 6 mm) 6. 5 lb (2. 9 kg) 1000 ml 1800 psi (124 bar) Reactor 3 316 L SS 14. 5 inch (365 mm) 1. 9 inch (48. 2 mm) 0. 24 inch (6. 1) 5. 6 lb (2. 5 kg) 300 ml 5000 psi (344 bar)

Fig 3 : Bearat’s Group reactors

Fig 4: The reactor used in GP 2 Fig 5: The pipe wrench

Fig 6 : Cylinder used.

Experiment and Result
In our experiments we used two systems : System I: For reaction between Na. OH with Air. System II: For reaction between Na. OH with pure CO 2.

System I: Compressed Air system Compressed air source. Plastic container.

Fig 7: Compressed Air Source Fig 8 : Sample Container

Fig 9 : Na. OH before reaction with air. Fig: 10 Na. OH after reaction with air.
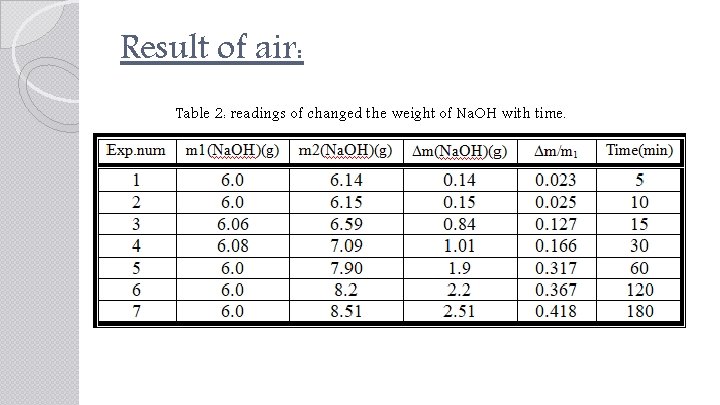
Result of air: Table 2: readings of changed the weight of Na. OH with time.
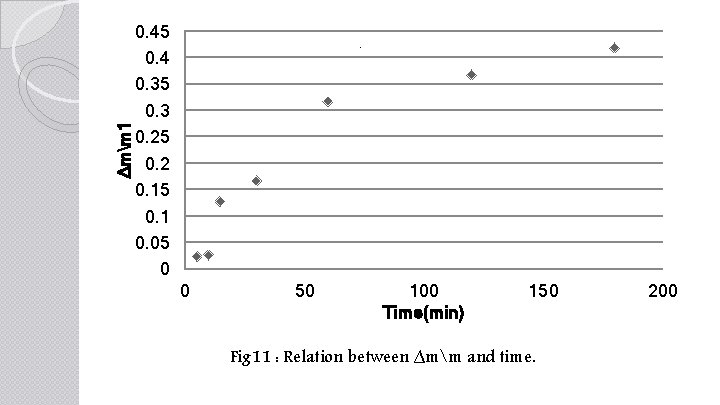
∆mm 1 0. 45 0. 4 0. 35 0. 3 0. 25 0. 2 0. 15 0. 1 0. 05 0 . 0 50 100 Time(min) 150 Fig 11 : Relation between ∆mm and time. 200
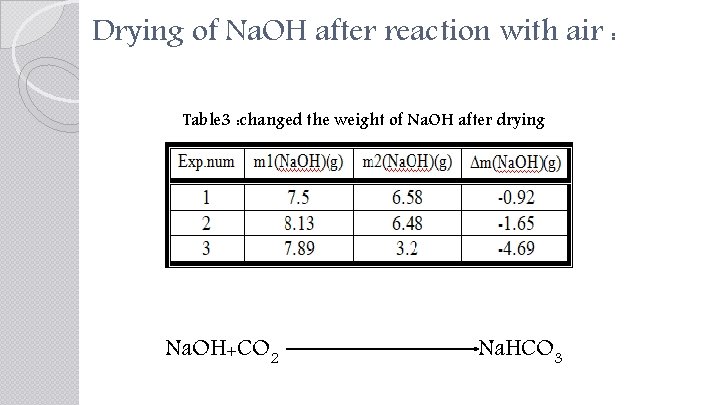
Drying of Na. OH after reaction with air : Table 3 : changed the weight of Na. OH after drying Na. OH+CO 2 Na. HCO 3
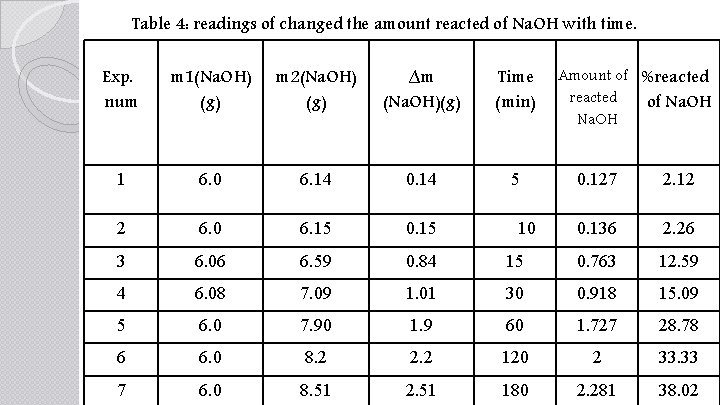
Table 4: readings of changed the amount reacted of Na. OH with time. Exp. num m 1(Na. OH) (g) m 2(Na. OH) (g) ∆m (Na. OH)(g) Time (min) 1 6. 0 6. 14 0. 14 5 2 6. 0 6. 15 0. 15 3 6. 06 6. 59 0. 84 4 6. 08 7. 09 5 6. 0 6 7 Amount of %reacted of Na. OH 0. 127 2. 12 0. 136 2. 26 15 0. 763 12. 59 1. 01 30 0. 918 15. 09 7. 90 1. 9 60 1. 727 28. 78 6. 0 8. 2 2. 2 120 2 33. 33 6. 0 8. 51 2. 51 180 2. 281 38. 02 10
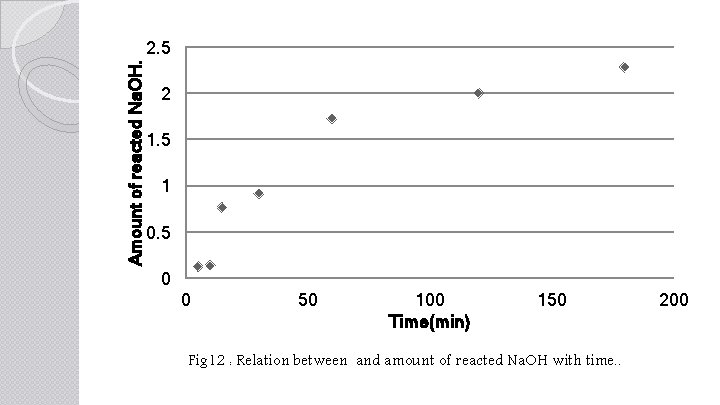
Amount of reacted Na. OH. 2. 5 2 1. 5 1 0. 5 0 0 50 100 Time(min) 150 Fig 12 : Relation between and amount of reacted Na. OH with time. . 200

Amount of CO 2 in 1 m 3 air : By assume this data In our planet the percentage CO 2 of air is 0. 0409% The total pressure is 1. 013 atm Assume the room temp is 22 C In volume = 1 m 3 = 1000 L by some calculations get In 1 m 3 of air there is 0. 77 g of CO 2
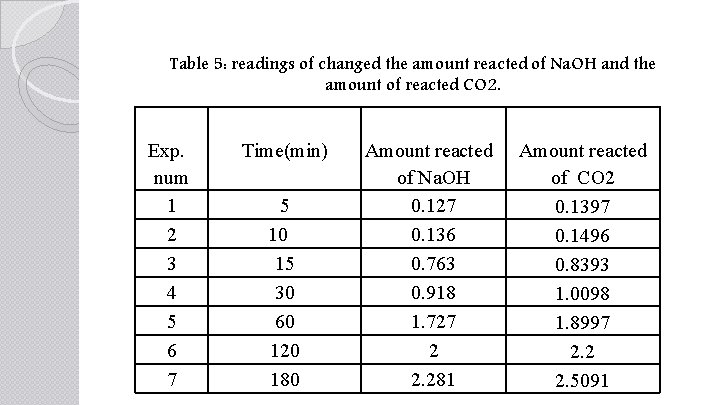
Table 5: readings of changed the amount reacted of Na. OH and the amount of reacted CO 2. Exp. num 1 2 3 4 5 6 7 Time(min) 5 10 15 30 60 120 180 Amount reacted of Na. OH 0. 127 0. 136 0. 763 0. 918 1. 727 2 2. 281 Amount reacted of CO 2 0. 1397 0. 1496 0. 8393 1. 0098 1. 8997 2. 2 2. 5091

System II: Pure CO 2 system

Fig 13 : System II

Fig 14 : Na. OH before reaction Fig 15 : Na. OH after reaction
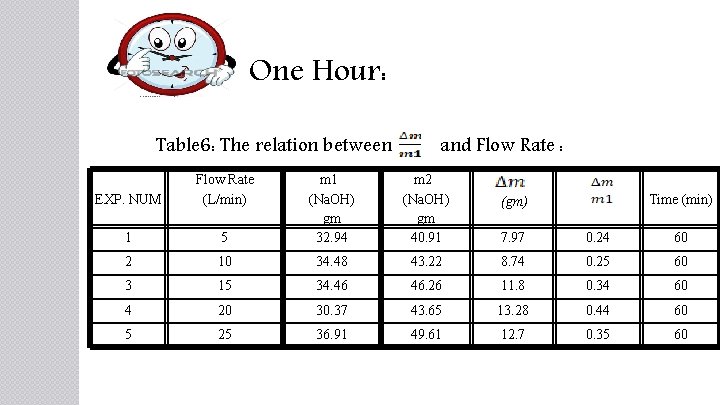
One Hour: Table 6: The relation between and Flow Rate : EXP. NUM Flow Rate (L/min) 1 5 m 1 (Na. OH) gm 32. 94 m 2 (Na. OH) gm 40. 91 7. 97 0. 24 60 2 10 34. 48 43. 22 8. 74 0. 25 60 3 15 34. 46 46. 26 11. 8 0. 34 60 4 20 30. 37 43. 65 13. 28 0. 44 60 5 25 36. 91 49. 61 12. 7 0. 35 60 Time (min) (gm)
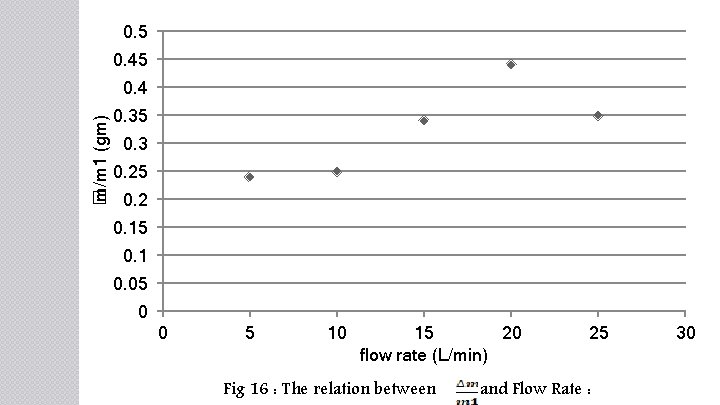
0. 5 0. 45 � m/m 1 (gm) 0. 4 0. 35 0. 3 0. 25 0. 2 0. 15 0. 1 0. 05 0 0 5 10 15 20 flow rate (L/min) Fig 16 : The relation between 25 and Flow Rate : 30

Temperature of the reaction: Fig 17: Thermocouple.
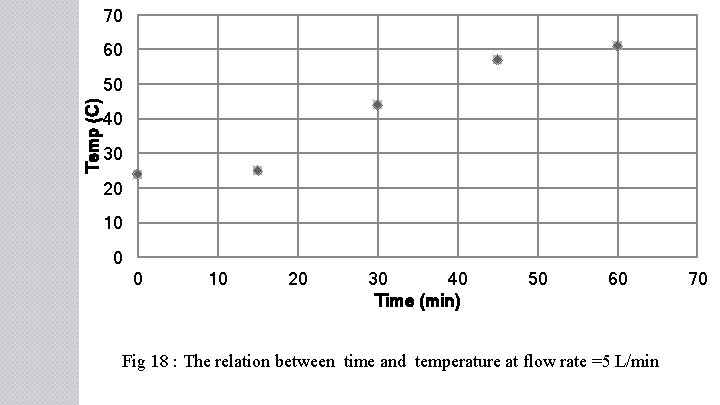
70 60 Temp (C) 50 40 30 20 10 0 0 10 20 30 40 Time (min) 50 60 Fig 18 : The relation between time and temperature at flow rate =5 L/min 70
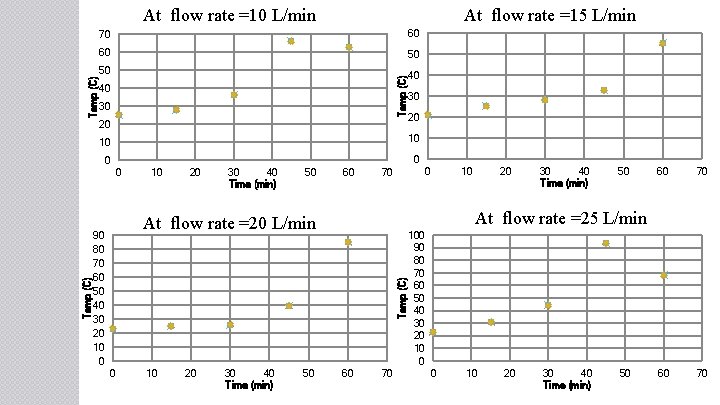
At flow rate =15 L/min 70 60 60 50 50 40 Temp (C) At flow rate =10 L/min 40 30 20 0 0 10 20 30 40 Time (min) 50 60 0 70 10 20 30 40 Time (min) 50 60 70 At flow rate =25 L/min At flow rate =20 L/min Temp (C) 0 0 20 10 10 90 80 70 60 50 40 30 20 10 0 30 60 70 100 90 80 70 60 50 40 30 20 10 0 0 10 20 30 40 Time (min) 50
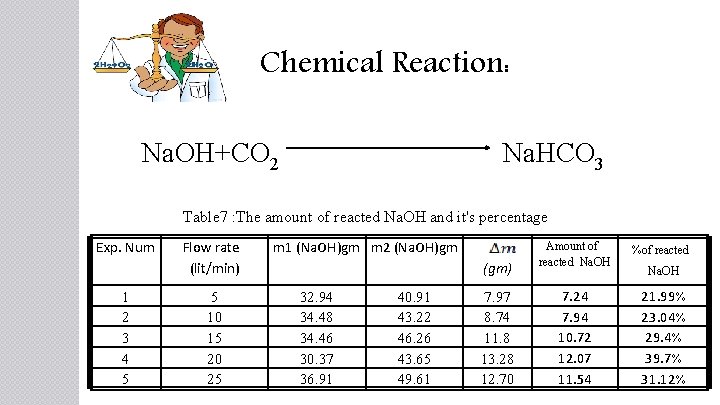
Chemical Reaction: Na. OH+CO 2 Na. HCO 3 Table 7 : The amount of reacted Na. OH and it's percentage Exp. Num 1 2 3 4 5 Flow rate (lit/min) 5 10 15 20 25 m 1 (Na. OH)gm m 2 (Na. OH)gm (gm) 32. 94 34. 48 34. 46 30. 37 36. 91 40. 91 43. 22 46. 26 43. 65 49. 61 7. 97 8. 74 11. 8 13. 28 12. 70 Amount of reacted Na. OH %of reacted 7. 24 7. 94 10. 72 12. 07 11. 54 21. 99% 23. 04% 29. 4% 39. 7% 31. 12% Na. OH
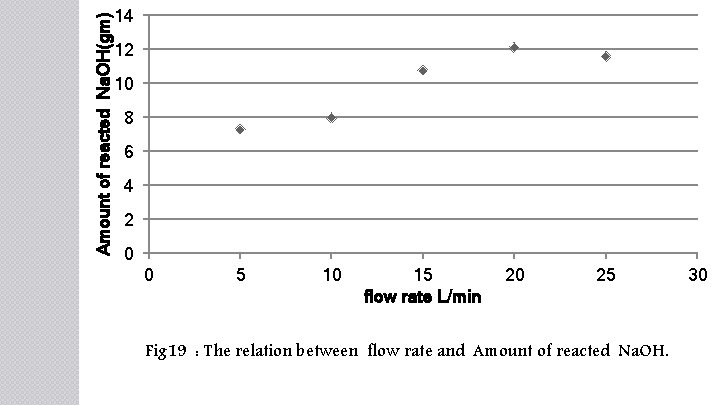
Amount of reacted Na. OH(gm) 14 12 10 8 6 4 2 0 0 5 10 15 flow rate L/min 20 25 Fig 19 : The relation between flow rate and Amount of reacted Na. OH. 30
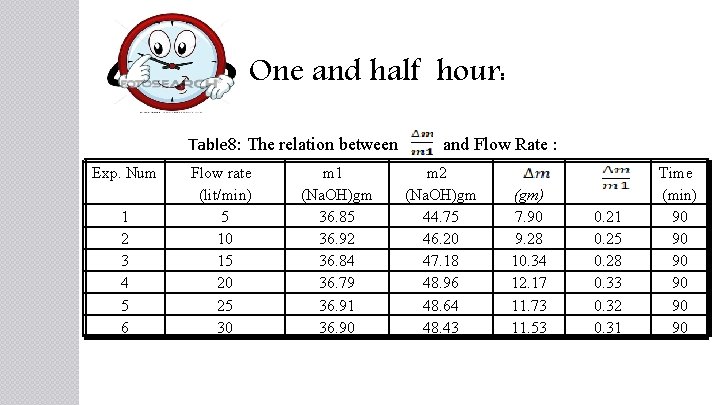
One and half hour: Table 8: The relation between and Flow Rate : Exp. Num 1 2 3 4 5 6 Flow rate (lit/min) 5 10 15 20 25 30 m 1 (Na. OH)gm 36. 85 36. 92 36. 84 36. 79 36. 91 36. 90 m 2 (Na. OH)gm 44. 75 46. 20 47. 18 48. 96 48. 64 48. 43 (gm) 7. 90 9. 28 10. 34 12. 17 11. 73 11. 53 0. 21 0. 25 0. 28 0. 33 0. 32 0. 31 Time (min) 90 90 90
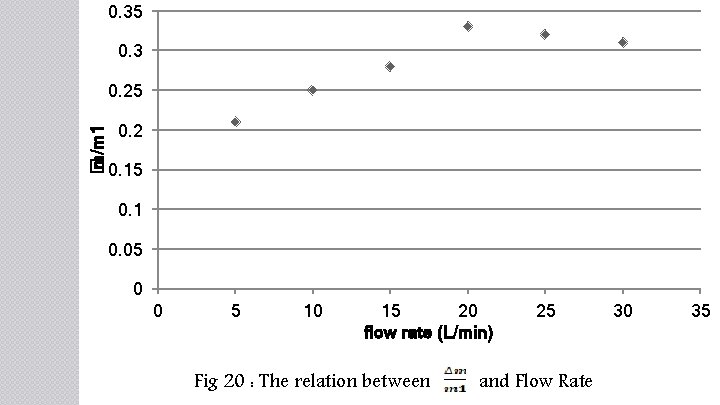
0. 35 0. 3 � m/m 1 0. 25 0. 2 0. 15 0. 1 0. 05 0 0 5 10 15 20 flow rate (L/min) Fig 20 : The relation between 25 and Flow Rate 30 35

Temperature of the reaction: Fig 21: Thermocouple.
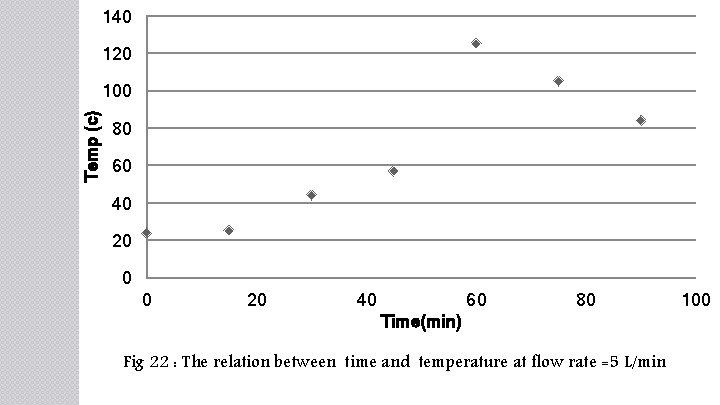
140 120 Temp (c) 100 80 60 40 20 0 0 20 40 60 80 Time(min) Fig 22 : The relation between time and temperature at flow rate =5 L/min 100
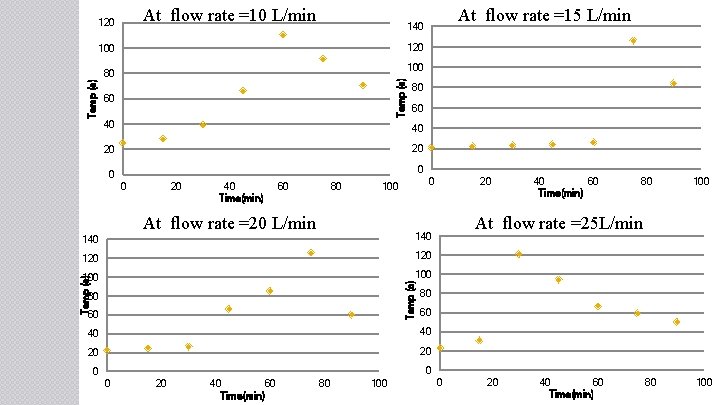
At flow rate =10 L/min 120 100 80 Temp (c) At flow rate =15 L/min 140 60 80 60 40 40 20 20 40 Time(min) 60 80 0 100 At flow rate =20 L/min 140 120 100 Temp (c) 140 80 60 20 40 60 Time(min) 80 100 At flow rate =25 L/min 80 60 40 40 20 20 40 60 Time(min) 80 100 0 20 40 60 Time(min) 80 100
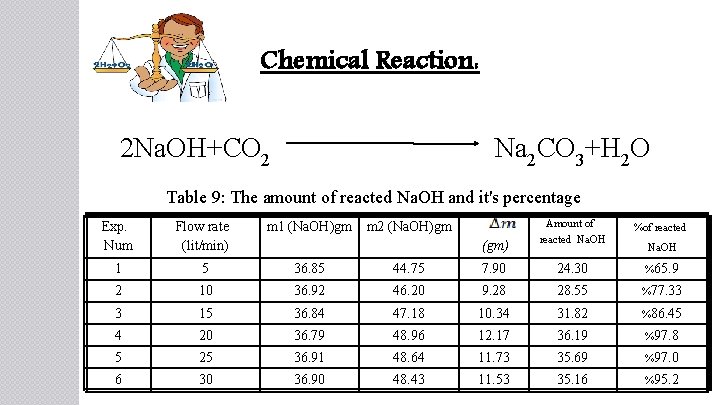
Chemical Reaction: 2 Na. OH+CO 2 Na 2 CO 3+H 2 O Table 9: The amount of reacted Na. OH and it's percentage Amount of reacted Na. OH %of reacted 7. 90 24. 30 %65. 9 46. 20 9. 28 28. 55 %77. 33 36. 84 47. 18 10. 34 31. 82 %86. 45 20 36. 79 48. 96 12. 17 36. 19 %97. 8 5 25 36. 91 48. 64 11. 73 35. 69 %97. 0 6 30 36. 90 48. 43 11. 53 35. 16 %95. 2 Exp. Num Flow rate (lit/min) m 1 (Na. OH)gm m 2 (Na. OH)gm 1 5 36. 85 44. 75 2 10 36. 92 3 15 4 (gm) Na. OH
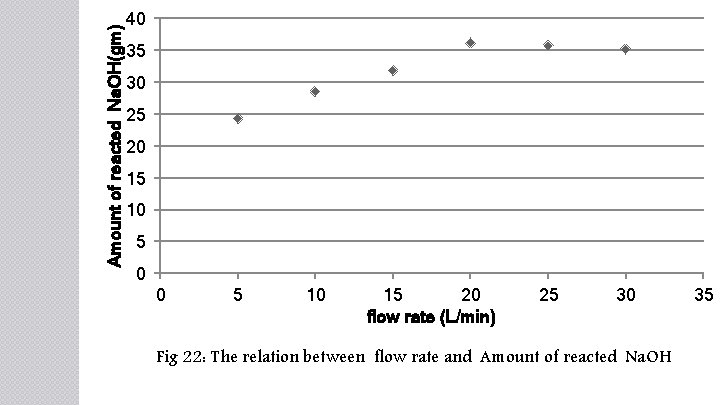
Amount of reacted Na. OH(gm) 40 35 30 25 20 15 10 5 0 0 5 10 15 20 flow rate (L/min) 25 30 Fig 22: The relation between flow rate and Amount of reacted Na. OH 35
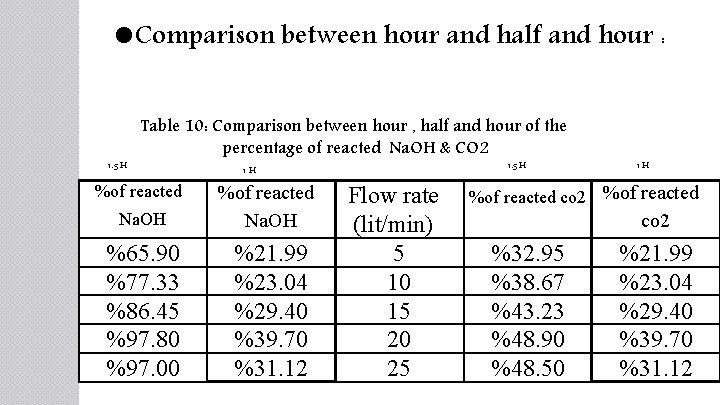
Comparison between hour and half and hour : 1. 5 H Table 10: Comparison between hour , half and hour of the percentage of reacted Na. OH & CO 2 %of reacted 1 H Na. OH %of reacted Na. OH %65. 90 %77. 33 %86. 45 %97. 80 %97. 00 %21. 99 %23. 04 %29. 40 %39. 70 %31. 12 1. 5 H Flow rate (lit/min) 5 10 15 20 25 1 H %of reacted co 2 %of reacted co 2 %32. 95 %38. 67 %43. 23 %48. 90 %48. 50 %21. 99 %23. 04 %29. 40 %39. 70 %31. 12
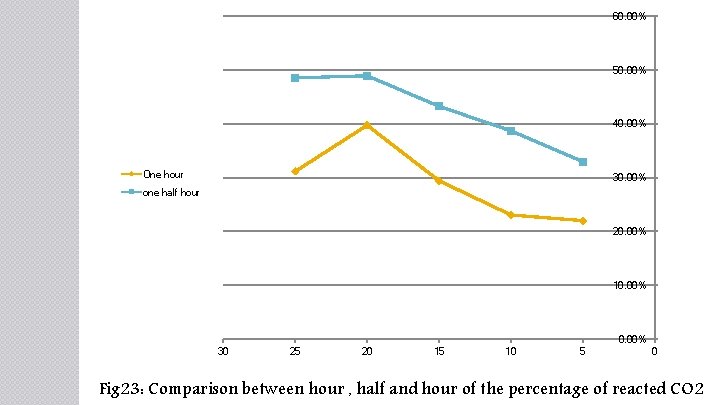
60. 00% 50. 00% 40. 00% One hour 30. 00% one half hour 20. 00% 10. 00% 30 25 20 15 10 5 0 Fig 23: Comparison between hour , half and hour of the percentage of reacted CO 2
Conclusions and Recommendation Co 2 capture process Co 2 flow rate Time

Problems A nitrogen (N 2) cylinder was installed in the lab instead of CO 2. This caused us a lot of delay because of wasted experiments. Problems in the balance: readings were sometimes very wrong. Knowing the flow rate of the air from the lineof laboratory and possibility to vary it. The lack of vise to assemble and disassemble the reactor in the laboratory.

-To help improve the research we started, some points must be recommended: ● Making a tool suitable for cars exhaust that contains Na. OH to capture CO 2 gas into stable and environmentally friendly products. ● Searching for materials that are cheaper than sodium hydroxide and with the same efficiency. ● Studying the temperature and the pressure in the reaction of sodium hydroxide and carbon dioxide and compare them with the conditions of the car. ● Studying more about the resulting material from the reaction of sodium hydroxide and carbon dioxide and how will the recovered CO 2 be used in other applications.
- Slides: 47