Carbon Compounds The Chemistry of Carbon Organic compounds

Carbon Compounds
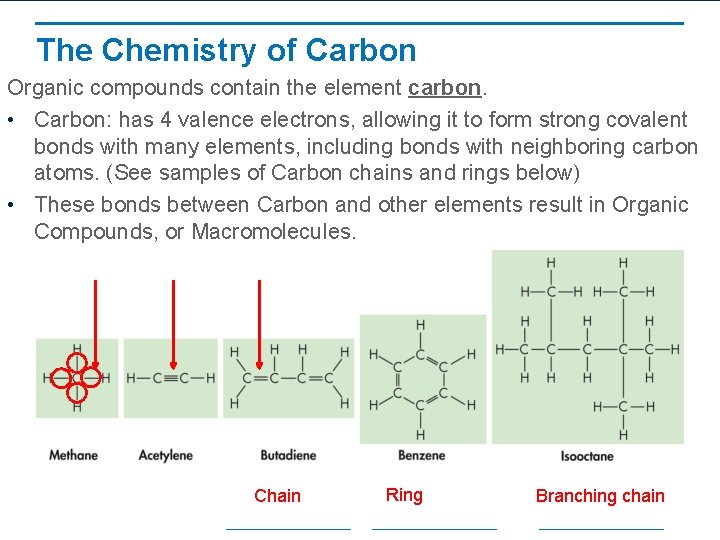
The Chemistry of Carbon Organic compounds contain the element carbon. • Carbon: has 4 valence electrons, allowing it to form strong covalent bonds with many elements, including bonds with neighboring carbon atoms. (See samples of Carbon chains and rings below) • These bonds between Carbon and other elements result in Organic Compounds, or Macromolecules. Chain Ring Branching chain
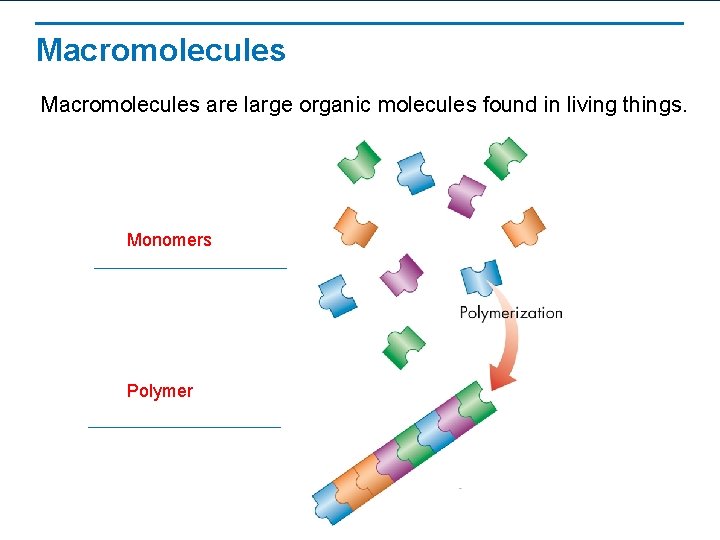
Macromolecules are large organic molecules found in living things. Monomers Polymer

Macromolecules 4 classes of Organic Compounds (Polymers) or Macromolecules: 1. Carbohydrates 2. Lipids 3. Proteins 4. Nucleic Acids
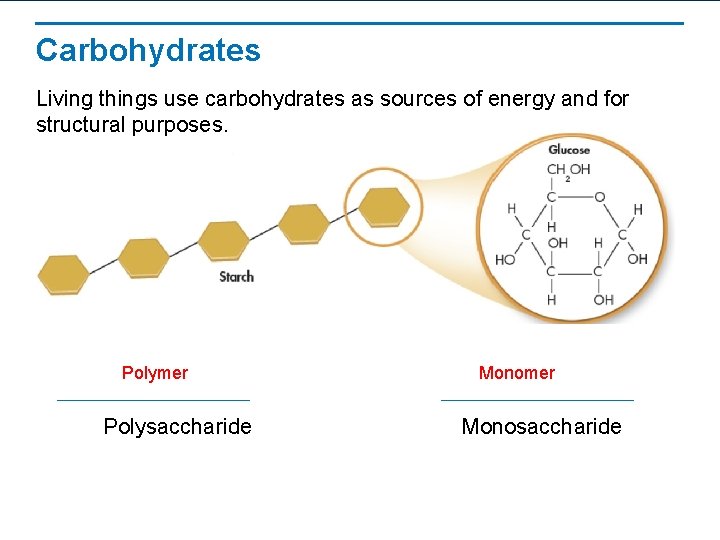
Carbohydrates Living things use carbohydrates as sources of energy and for structural purposes. Polymer Polysaccharide Monomer Monosaccharide

Carbohydrates • • Carbohydrates are known as “sugars” Made up of Carbon, Hydrogen, & Oxygen atoms Key source of energy for living things 3 kinds: 1. Monosaccharides • single sugars • ex. Glucose, fructose 2. Disaccharides • double sugars • ex. Sucrose 3. Polysaccharides • 3 or more monosaccharides put together • ex. starch
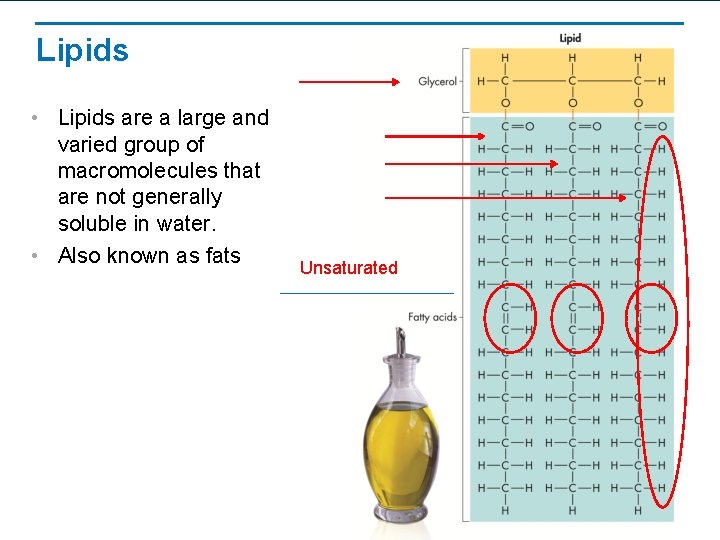
Lipids • Lipids are a large and varied group of macromolecules that are not generally soluble in water. • Also known as fats Unsaturated

Lipids— 2 types Saturated fats • Molecules saturated with hydrogens • Animal fats – butter, lard • Solid at room temperature Unsaturated fats • Molecules not saturated with hydrogens • Oils – olive oil, fish oil • Liquid at room temperature
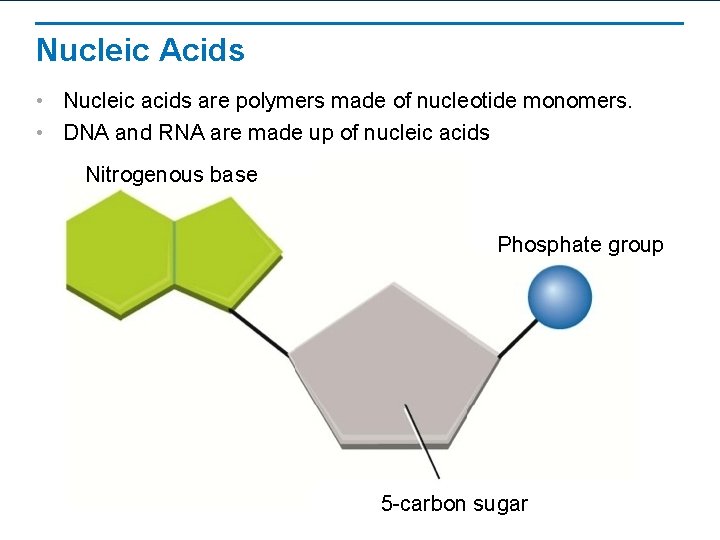
Nucleic Acids • Nucleic acids are polymers made of nucleotide monomers. • DNA and RNA are made up of nucleic acids Nitrogenous base Phosphate group 5 -carbon sugar
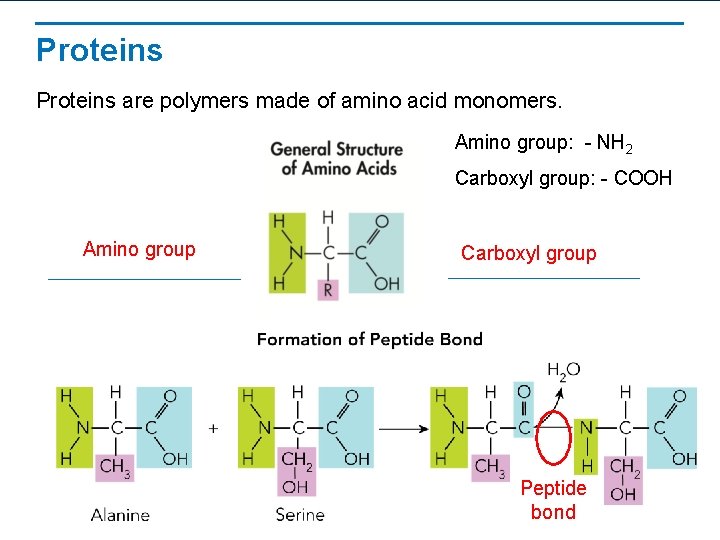
Proteins are polymers made of amino acid monomers. Amino group: - NH 2 Carboxyl group: - COOH Amino group Carboxyl group Peptide bond
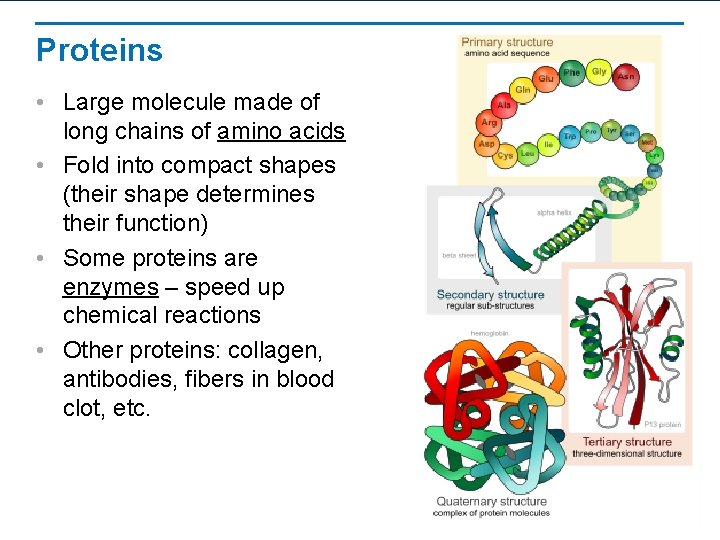
Proteins • Large molecule made of long chains of amino acids • Fold into compact shapes (their shape determines their function) • Some proteins are enzymes – speed up chemical reactions • Other proteins: collagen, antibodies, fibers in blood clot, etc.
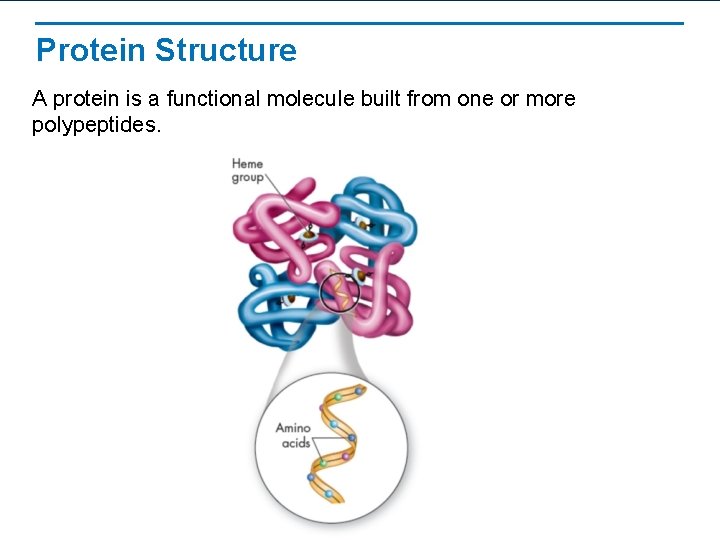
Protein Structure A protein is a functional molecule built from one or more polypeptides.

Chemical Reactions A chemical reaction changes one set of chemicals into another. The elements or compounds that engage in a chemical reaction are known as reactants. The elements or compounds produced by a chemical reaction are known as products.
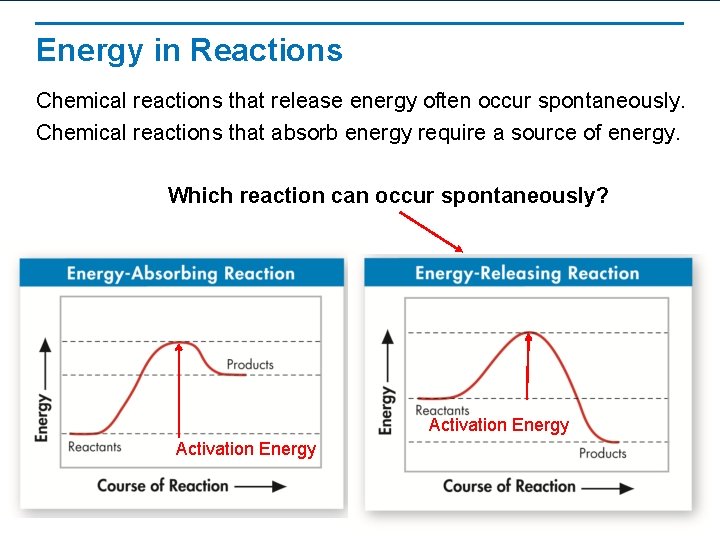
Energy in Reactions Chemical reactions that release energy often occur spontaneously. Chemical reactions that absorb energy require a source of energy. Which reaction can occur spontaneously? Activation Energy
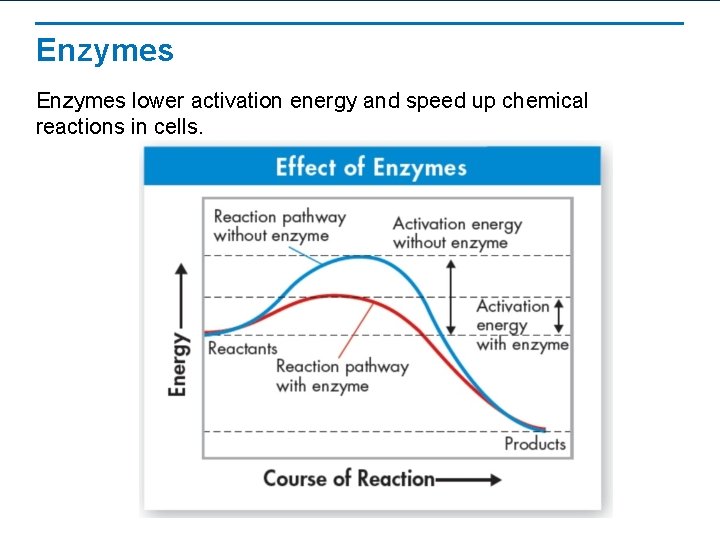
Enzymes lower activation energy and speed up chemical reactions in cells.

The Enzyme-Substrate Complex Substrate – substance that an enzyme acts upon Active Sites – deep folds/pockets on the enzyme • Substrate attaches to an enzyme’s active site (fit like lock & key) • Enzyme reduces activation energy • When reaction is complete, the enzyme is free to work again (REUSABLE!!)
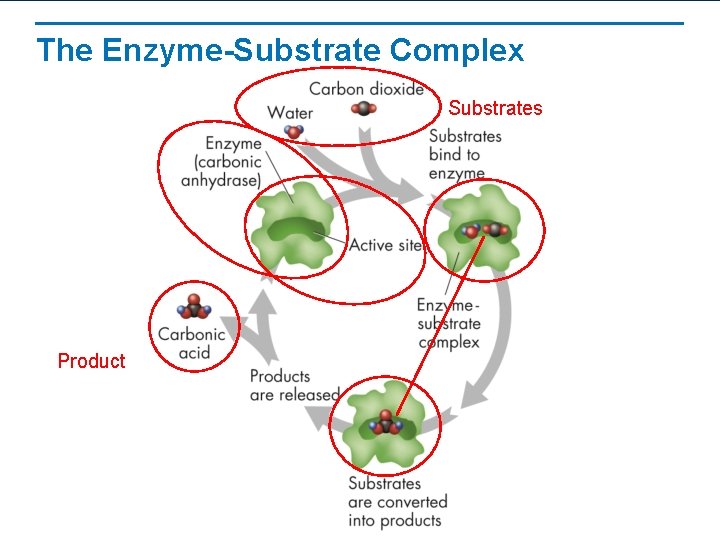
The Enzyme-Substrate Complex Substrates Product
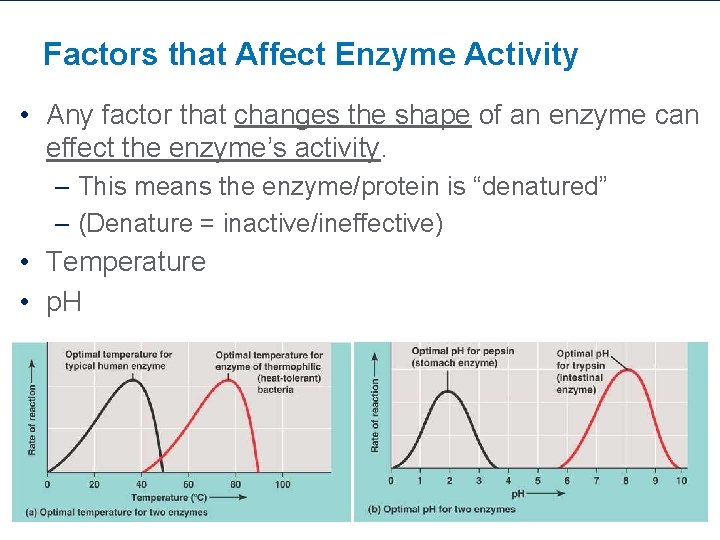
Factors that Affect Enzyme Activity • Any factor that changes the shape of an enzyme can effect the enzyme’s activity. – This means the enzyme/protein is “denatured” – (Denature = inactive/ineffective) • Temperature • p. H
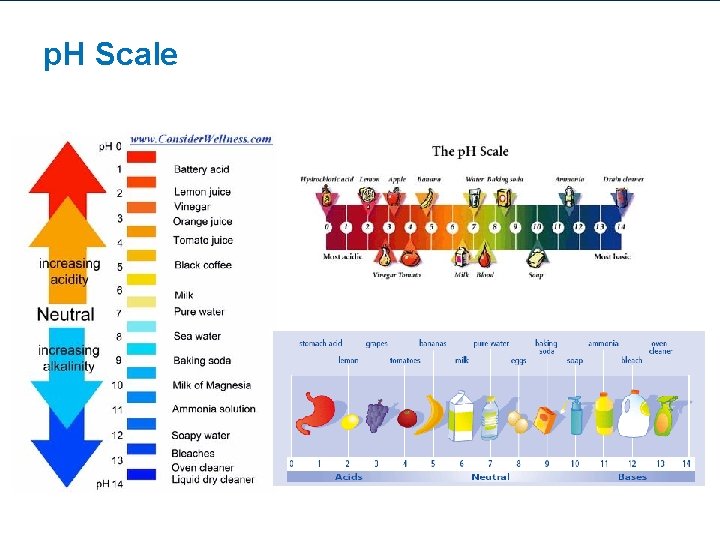
p. H Scale
- Slides: 19