Carbon Compounds Macromolecules Organic Compounds contain carbon atoms

Carbon Compounds Macromolecules

Organic Compounds • contain carbon atoms covalently bonded to other atoms – covalent bond = sharing electrons • makes up most matter in your body that is not water • has an entire branch of chemistry devoted to it called Organic Chemistry

Carbon Bonds • Carbon has 4 valence electrons, allowing it to form 4 bonds – Typically bonds with H, O, P, S, & N • can bond to itself forming single, double, and triple bonds in chains – can be almost unlimited in length – can also close to form rings

Macromolecules • A large molecule made from thousands or hundreds of thousands of smaller molecules.

Macromolecules • Monomers - molecule able to bond in long chains join to form • Polymers - many monomers joined together to form a macromolecule
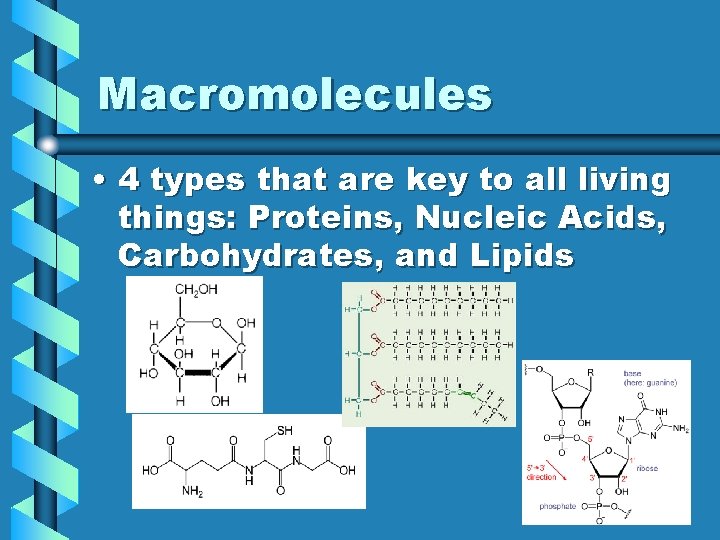
Macromolecules • 4 types that are key to all living things: Proteins, Nucleic Acids, Carbohydrates, and Lipids
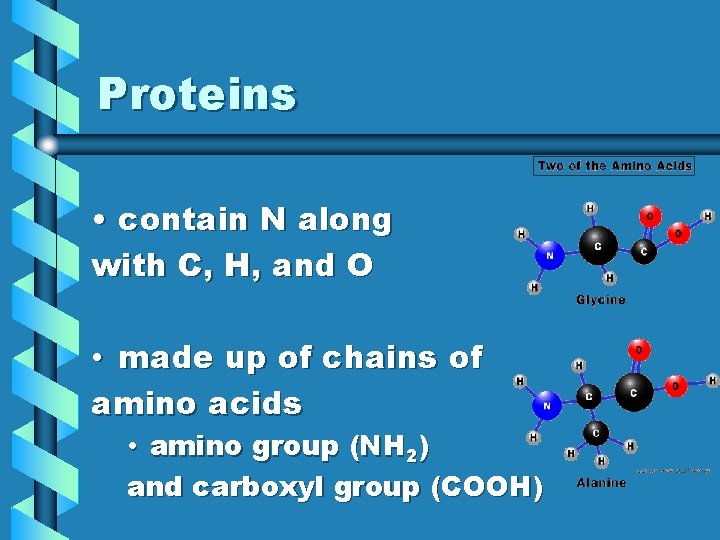
Proteins • contain N along with C, H, and O • made up of chains of amino acids • amino group (NH 2) and carboxyl group (COOH)

Amino Acids • 20 different amino acids found in nature. • Long chains of amino acids then fold into compact shapes

Amino Acids • Nonessential amino acids – the body can make • Essential amino acids – must obtain through our diet

Proteins in our bodies • Next to water, proteins make up the biggest portion of our body weight • Form tissues, muscles, skin, hormones (ex: insulin), & enzymes (which speed up chemical reactions)

Proteins in our bodies • Genes (DNA) code for proteins – that determines what we look like

Nucleic Acids • contain H, O, N, C, and P • made up of nucleotides – a sugar, a nitrogen base, and a phosphate group • Two Types of Nucleic Acids are – DNA (Deoxyribonucleic acid) – RNA (Ribonucleic acid)
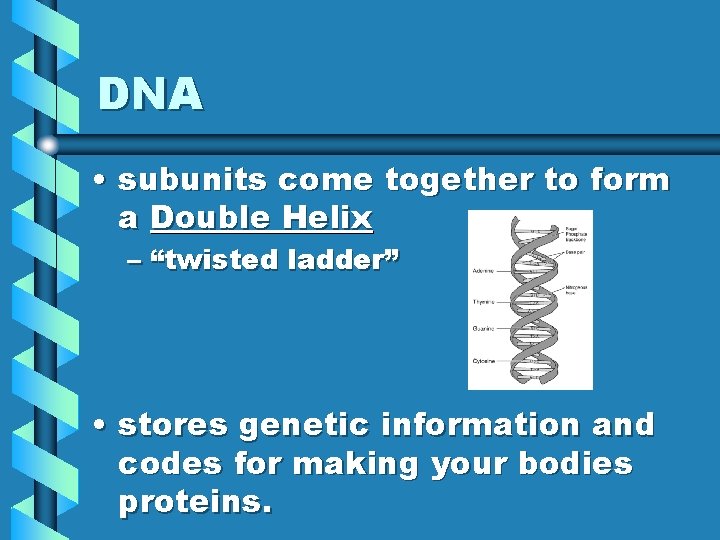
DNA • subunits come together to form a Double Helix – “twisted ladder” • stores genetic information and codes for making your bodies proteins.

RNA • a copy of DNA that carries a message (code) through the cell • The code is then used to build proteins

Carbohydrates • Contain C, H, and O oriented in a carbon ring. • Main source of energy, and used for a structural purpose. • Sugars are used for immediate energy.
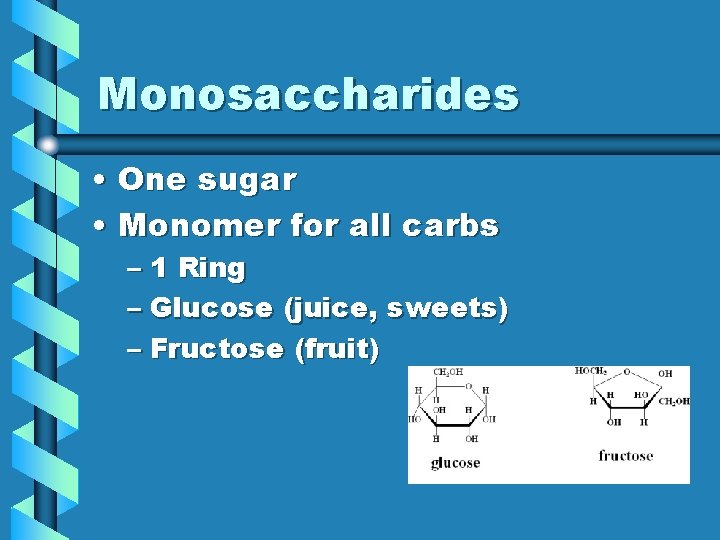
Monosaccharides • One sugar • Monomer for all carbs – 1 Ring – Glucose (juice, sweets) – Fructose (fruit)
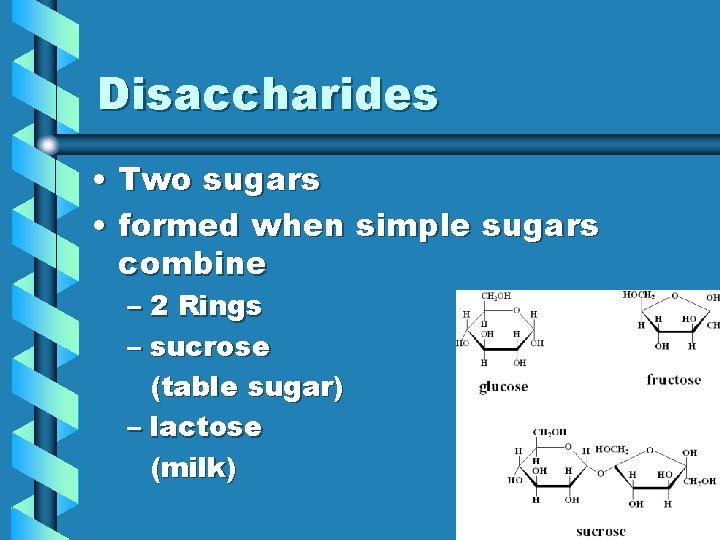
Disaccharides • Two sugars • formed when simple sugars combine – 2 Rings – sucrose (table sugar) – lactose (milk)

Polysaccharides • many sugars • formed when many monosaccharides combine – Starch (made by plants) – Cellulose (cell wall) – Glycogen (made by animals – stored in the liver)

Lipids • Large C-H molecules with some O that are not soluble in water. – Lipids are nonpolar • Made up of Fatty Acids combined with a glycerol molecule.

Lipids • Saturated - carbon chains that have no double bonds – generally solids at room temperature • Unsaturated - carbon chains that have at least one double bond – generally liquid at room temperature – Polyunsaturated - More than 1 double bond

Lipids

Lipids • Trans fats – naturally unsaturated fatty acids that have been saturated artificially by the addition of hydrogen atoms • Used to extend shelf life of food and to enhance food flavor, etc. , but at a dangerous price……….

Common Types of Lipids • Fats • Oils • Steroids • Waxes

Organic Uses of Lipids • Store energy • Biological membranes (waterproof) • Lipid hormones • Plant pigments
- Slides: 24