Carbon carbides carbonates and carbonatitic melts in the

Carbon, carbides, carbonates and carbonatitic melts in the Earth's interior by Vincenzo Stagno Journal of the Geological Society Volume (): jgs 2018 -095 February 5, 2019 © 2019 The Author(s). Published by The Geological Society of London
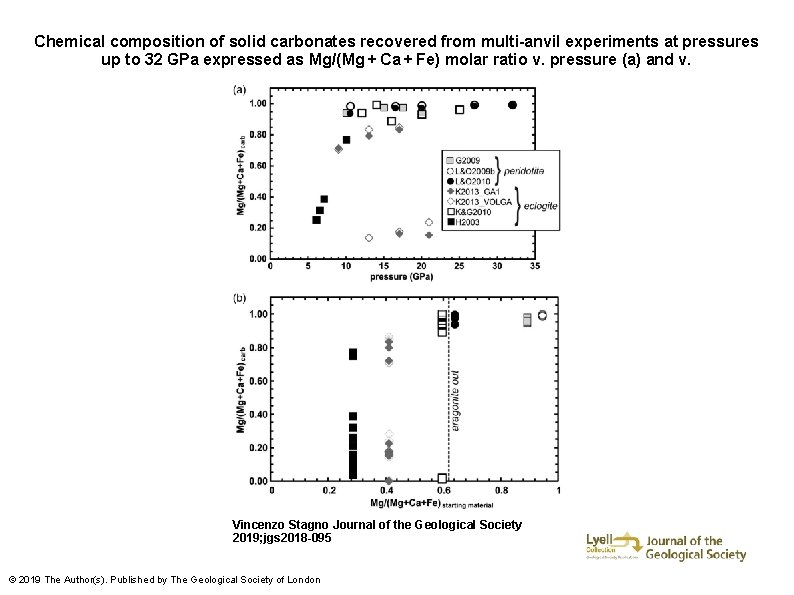
Chemical composition of solid carbonates recovered from multi-anvil experiments at pressures up to 32 GPa expressed as Mg/(Mg + Ca + Fe) molar ratio v. pressure (a) and v. Vincenzo Stagno Journal of the Geological Society 2019; jgs 2018 -095 © 2019 The Author(s). Published by The Geological Society of London
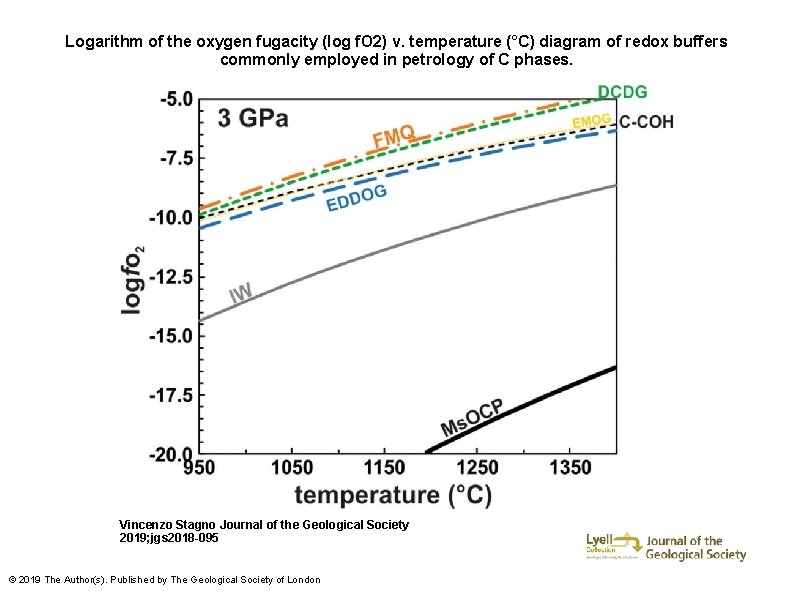
Logarithm of the oxygen fugacity (log f. O 2) v. temperature (°C) diagram of redox buffers commonly employed in petrology of C phases. Vincenzo Stagno Journal of the Geological Society 2019; jgs 2018 -095 © 2019 The Author(s). Published by The Geological Society of London
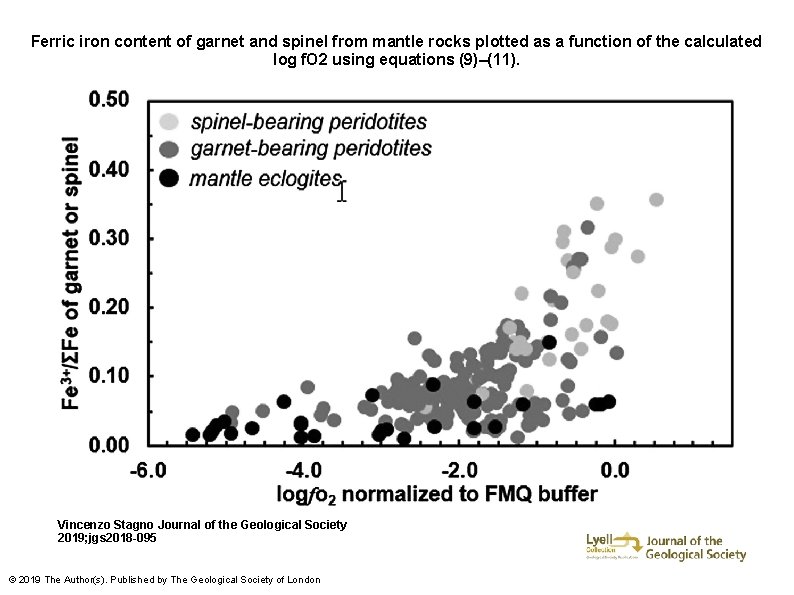
Ferric iron content of garnet and spinel from mantle rocks plotted as a function of the calculated log f. O 2 using equations (9)–(11). Vincenzo Stagno Journal of the Geological Society 2019; jgs 2018 -095 © 2019 The Author(s). Published by The Geological Society of London
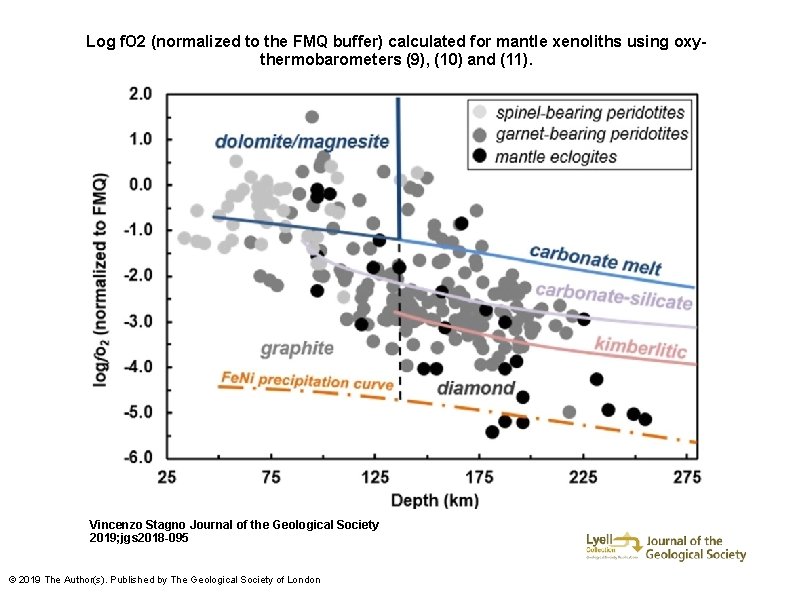
Log f. O 2 (normalized to the FMQ buffer) calculated for mantle xenoliths using oxythermobarometers (9), (10) and (11). Vincenzo Stagno Journal of the Geological Society 2019; jgs 2018 -095 © 2019 The Author(s). Published by The Geological Society of London
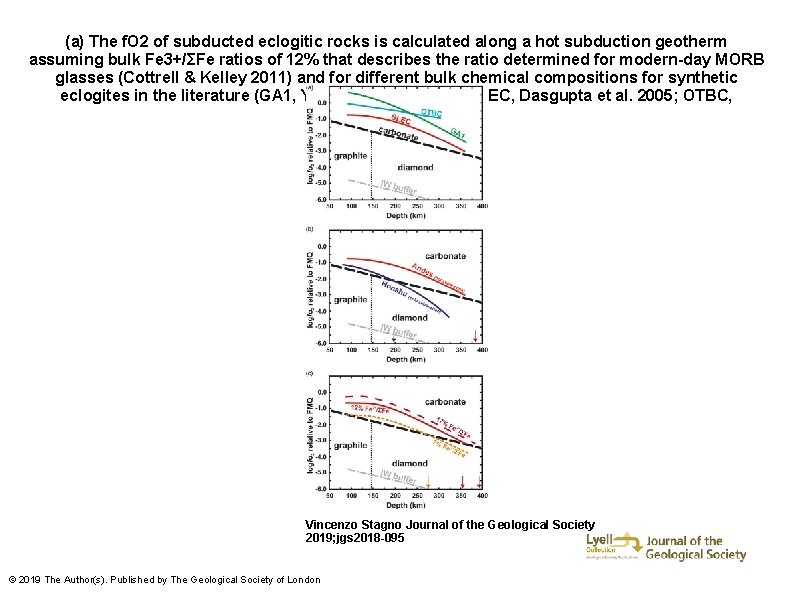
(a) The f. O 2 of subducted eclogitic rocks is calculated along a hot subduction geotherm assuming bulk Fe 3+/ΣFe ratios of 12% that describes the ratio determined for modern-day MORB glasses (Cottrell & Kelley 2011) and for different bulk chemical compositions for synthetic eclogites in the literature (GA 1, Yaxley & Green 1994; SLEC, Dasgupta et al. 2005; OTBC, Hammouda 2003). Vincenzo Stagno Journal of the Geological Society 2019; jgs 2018 -095 © 2019 The Author(s). Published by The Geological Society of London
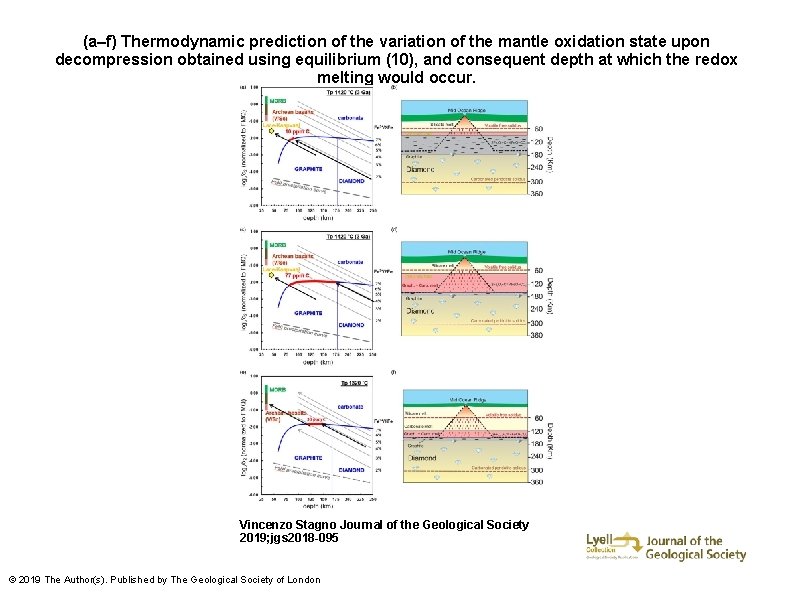
(a–f) Thermodynamic prediction of the variation of the mantle oxidation state upon decompression obtained using equilibrium (10), and consequent depth at which the redox melting would occur. Vincenzo Stagno Journal of the Geological Society 2019; jgs 2018 -095 © 2019 The Author(s). Published by The Geological Society of London
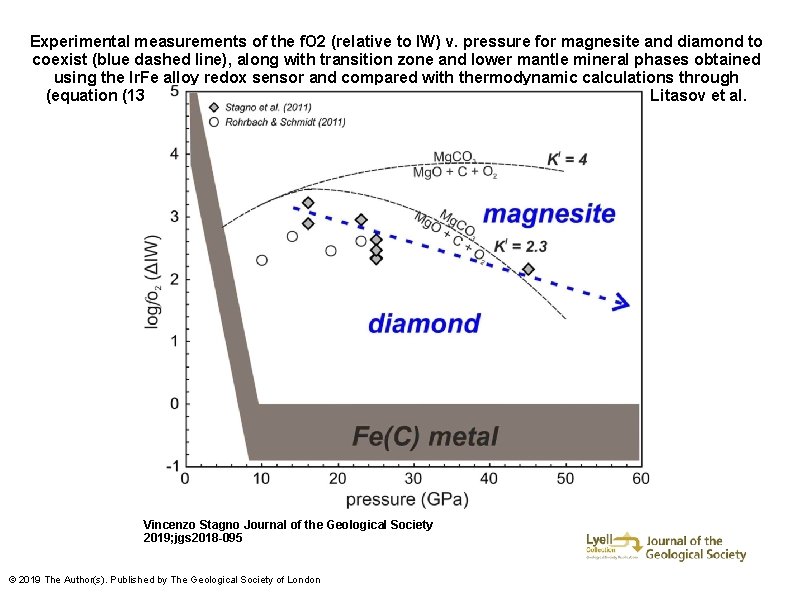
Experimental measurements of the f. O 2 (relative to IW) v. pressure for magnesite and diamond to coexist (blue dashed line), along with transition zone and lower mantle mineral phases obtained using the Ir. Fe alloy redox sensor and compared with thermodynamic calculations through (equation (13)) changing Eo. S data for magnesite (Fiquet et al. 1994; Ross 1997; Litasov et al. 2008). Vincenzo Stagno Journal of the Geological Society 2019; jgs 2018 -095 © 2019 The Author(s). Published by The Geological Society of London
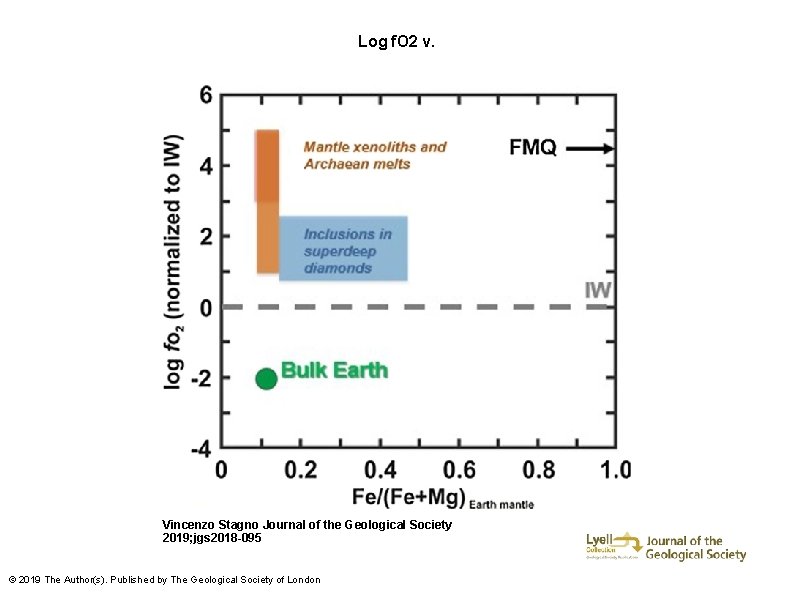
Log f. O 2 v. Vincenzo Stagno Journal of the Geological Society 2019; jgs 2018 -095 © 2019 The Author(s). Published by The Geological Society of London
- Slides: 9