Carbon capture at moderate pressures and temperatures Moderate

Carbon capture at moderate pressures and temperatures • Moderate pressures around 15 bar • Moderate temperatures – Hydrate formation at 2 -6 o. C • Not far below cooling water temperatures in Nordic countries – CO 2 release 20 – 40 o. C • Typical waste heat temperatures
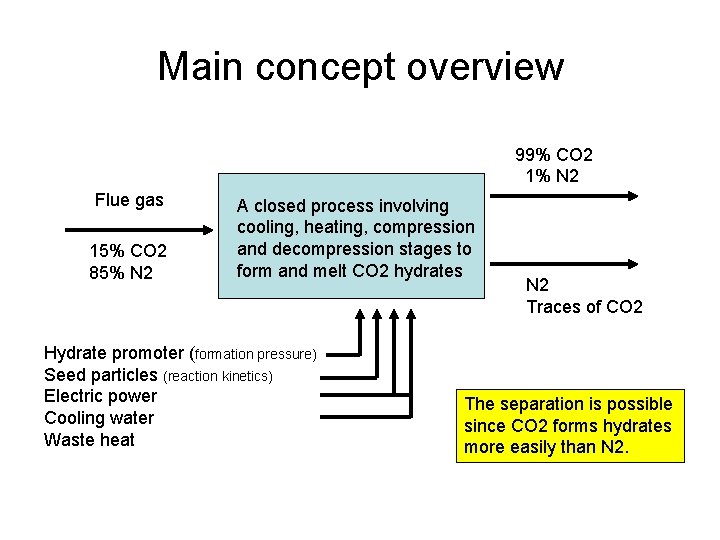
Main concept overview 99% CO 2 1% N 2 Flue gas 15% CO 2 85% N 2 A closed process involving cooling, heating, compression and decompression stages to form and melt CO 2 hydrates Hydrate promoter (formation pressure) Seed particles (reaction kinetics) Electric power Cooling water Waste heat N 2 Traces of CO 2 The separation is possible since CO 2 forms hydrates more easily than N 2.

The problem with hydrate processes • There is usually a long induction time before hydrate production start – Seed must form and grow to a certain size before detectable gas absorption is observed – This takes a long time – hours and days in pure systems • The hydrate forms first where the gas concentration is high – Thus a droplet gets a hydrate crust around a wet inside • This hinders the transport of gas into the water phase • And heat away from the reaction centre • Speeding up the process – the IFE contribution and a possible breakthrough – Using heterogeneous seed particle to speed up hydrate formation – induction time reduced by a factor of 200

Comparing to chilled ammonia process • From literature chilled ammonia process consumes energy in the range: – 470 – 550 k. Wh/ton CO 2 • A first rough estimate for a hydrate process: – 220 – 330 k. W/ton CO 2 (0. 8 – 1. 2 GJ)
- Slides: 4