Carbon Capture and Storag ebook ebook overview 1

Carbon Capture and Storag e-book

e-book overview 1. 2. 3. 4. 5. 6. 7. 8. 9. 10. Climate change and energy consumption CCS as an option for CO 2 emission reduction Geologic storage and trapping mechanisms Storage potentials and capacity estimates Site selection + characterisation CO 2 -EOR Storage risks Monitoring Numerical modeling of CO 2 storage Regulatory and social aspects of CCS technology

lecture 3 Geological storage and trapping by Karen Kirk mechanisms British Geological Survey

lecture overview § § § § § overview of geological storage options geology of reservoir and caprocks properties of CO 2 injectivity, pressure buildup pressure management trapping mechanisms interaction and relative importance of trapping mechanisms summary glossary references

overview of geological storage options To store carbon dioxide geologically a reservoir rock capable of storing fluids (e. g. compressed CO 2) is required. The best options are in: (1) Depleted oil and gas reservoirs (2) Enhanced Oil Recovery (EOR) (3) Deep saline formations There is also further potential in unmineable coal seams (4) Image copyright CO 2 CRC

geological storage options Depleted oil and gas reservoirs Pros § have proven geological traps § ability to store buoyant fluids over geological timescales § well-known/thoroughly investigated § injection/production facilities present if utilised as soon as field is depleted § possible benefits to hydrocarbon industry for Enhanced Oil Recovery/Enhanced Gas Recovery (EOR/EGR) Cons § generally far from CO 2 sources § storage capacity limited § many sites not available for 40 plus years

geological storage options Saline water-saturated reservoir rocks (saline aquifers) Pros § some saline aquifers may exhibit required criteria for CO 2 storage § large porous volume capable of storing large amounts of CO 2 § commonly occur in major accumulations known as sedimentary basins § not being used for other purposes e. g. extraction for drinking water Cons § no injection/production facilities § generally poorly investigated

geological storage options Un-mineable coal fields Pros § CO 2 can be adsorbed onto coal surface § coal contains micropores in which natural gas can occur § CO 2 can be used to extract Coalbed Methane (CBM) gas § CO 2 has a greater affinity to be adsorbed onto coal than methane Cons § coal does not contain large pore spaces § probably will be limited capacity due to low permeability § need to ensure displaced methane is captured as it is a more powerful greenhouse gas than CO 2 § coal may have another economic use such as a source of energy (mining or underground coal gasification)

geology of reservoirs – sandstone The most common types of reservoir rock are sandstones. § sandstones contain individual grains of sand (composed of quartz) up to about 2 mm in size. § the sand grains are cemented together usually by quartz and/or calcite due to precipitation after the sandstone material has been deposited. § due to the variety of sizes and shapes of the sand grains, when cemented together, interconnected spaces between the grains are formed. These can be measured as porosity (pore spaces) and permeability (pathways through the pore spaces).

geology of reservoirs – limestone Another reservoir rock is Limestone. § limestones are composed of more than 50 % calcium carbonate. § many limestones are similar to sandstones in that they contain sand-sized carbonate grains though others are similar to mudstones which are composed of fine-grained muds. § limestones also may contain fossil material e. g. skeletal and shell debris. § reef limestones are dominated by in-situ skeletons. They contain cavities which are infilled with internal sediment and cement. § limestones react with acid (dissolve).

desirable parameters for reservoirs as CO 2 storage sites § minimum depth (top of reservoir formation): > 800 m (to ensure the maximum amount of CO 2 can be stored †) § maximum depth (base of reservoir formation): < 2500 m (below this the porosity and permeability of the storage formation may be too low) § net thickness of reservoir rocks within reservoir formation: > 20 m § porosity of reservoir rocks: > 10 % § permeability of reservoir rocks: > 200 millidarcies (m. D) Parameters derived from the ‘Best Practice for the storage of CO 2 in Saline Aquifers’, based on EU projects ‘SACS’ and ‘CO 2 STORE’ [Chadwick et al. , 2008]. † explained in greater detail in CO 2 properties section

coal seams as potential storage sites There is also the possibility of permanent storage in unmineable coal seams using CO 2 for Enhanced Coal Bed Methane (ECBM) retrieval. § coals are organic-rich deposits containing plant material. § coal seams have natural fractures (known as cleat) along which the coal breaks into fragments. § it is along the cleat surfaces where CO 2 can be preferentially be adsorbed and in some instances be exchanged for methane.

test What is the desirable minimum depth the store CO 2 at? 20 metres 50 metres 800 metres 2500 metres skip test

test What is the desirable minimum depth the store CO 2 at? x 20 metres 50 metres 800 metres 2500 metres submit

test What is the desirable minimum depth the store CO 2 at? 20 metres x 50 metres 800 metres 2500 metres submit

test What is the desirable minimum depth the store CO 2 at? 20 metres 50 metres x 800 metres 2500 metres submit

test What is the desirable minimum depth the store CO 2 at? 20 metres 50 metres 800 metres x 2500 metres submit
test unfortunately, this is not correct try again skip test

test What is the desirable minimum depth the store CO 2 at? 20 metres 50 metres x 800 metres 2500 metres this is correct, keep it up

test Which of the following rock types are suitable as reservoir (storage) rocks? halite sandstone siltstone claystone skip test

test Which of the following rock types are suitable as reservoir (storage) rocks? x halite sandstone siltstone claystone submit

test Which of the following rock types are suitable as reservoir (storage) rocks? halite x sandstone siltstone claystone submit

test Which of the following rock types are suitable as reservoir (storage) rocks? halite sandstone x siltstone claystone submit

test Which of the following rock types are suitable as reservoir (storage) rocks? halite sandstone siltstone x claystone submit
test unfortunately, this is not correct try again skip test

test Which of the following rock types are suitable as reservoir (storage) rocks? halite x sandstone siltstone claystone this is correct, good job

geology of caprocks Caprocks are provided by impermeable or low permeability rocks for example: § halite (salt layers) § mudrocks (shales, mudstones, siltstones, claystones). The rock providing the seal may be porous and contain brine/water but due to low permeability, capillary forces should prevent the CO 2 from entering it. Caprocks should provide a large areal distribution, be continuous, thick and preferably unfaulted. They should also be non-reactive to CO 2 (e. g. limestone is no good as it will dissolve in CO 2 -rich fluids due to the acidity).

geology of caprocks – halites Halite is commonly known as rock salt. § it occurs over large areas and can be hundreds of metres thick § it forms as evaporite minerals where enclosed seas and large lakes dry out § it can self-heal in the event that it is fractured because it has the ability to creep (it is mobile due to the weight of the overlying sediments/rock) § halite is impermeable and provides an excellent seal for reservoirs.

geology of caprocks – mudrocks Mudrocks are fine-grained rocks composed of clay minerals and silt-sized grains. § shales are thinly bedded and fissile (ability to be split – see photo right) § mudstones are usually massive and blocky in structure and are non-fissile § claystones are extremely fine-grained (< 0. 004 mm) with a homogenous appearance § siltstones are mainly composed of quartz grains sized 0. 004 – 0. 062 mm § mudrocks can have fairly high porosity but low permeability

desirable parameters for caprocks § adequate lithology, e. g. claystones, salt (halite) § depth (base of barrier formation): > 800 m § thickness of adequate sealing rocks: > 20 m Parameters derived from the ‘Best Practice for the storage of CO 2 in Saline Aquifers’, based on EU projects ‘SACS’ and ‘CO 2 STORE’ [Chadwick et al. , 2008].

test Which of these statements is correct? effective caprocks are impermeable and react to CO 2 effective caprocks cover a large area and have high permeability effective caprocks cover a large area and may be porous but low permeability prevents CO 2 from entering it limestones are effective caprocks skip test

test Which of these statements is correct? x effective caprocks are impermeable and react to CO 2 effective caprocks cover a large area and have high permeability effective caprocks cover a large area and may be porous but low permeability prevents CO 2 from entering it limestones are effective caprocks submit

test Which of these statements is correct? effective caprocks are impermeable and react to CO 2 x effective caprocks cover a large area and have high permeability effective caprocks cover a large area and may be porous but low permeability prevents CO 2 from entering it limestones are effective caprocks submit

test Which of these statements is correct? effective caprocks are impermeable and react to CO 2 effective caprocks cover a large area and have high permeability x effective caprocks cover a large area and may be porous but low permeability prevents CO 2 from entering it limestones are effective caprocks submit

test Which of these statements is correct? effective caprocks are impermeable and react to CO 2 effective caprocks cover a large area and have high permeability effective caprocks cover a large area and may be porous but low permeability prevents CO 2 from entering it x limestones are effective caprocks submit
test unfortunately, this is not correct try again skip test

test Which of these statements is correct? effective caprocks are impermeable and react to CO 2 effective caprocks cover a large area and have high permeability x effective caprocks cover a large area and may be porous but low permeability prevents CO 2 from entering it limestones are effective caprocks this is correct, keep it up

test Which of these would make a good caprock? a heavily faulted claystone that has low permeability a continuous limestone full of skeletal debris a discontinuous mudstone that contains no faults a halite with faults that have self-healed skip test

test Which of these would make a good caprock? x a heavily faulted claystone that has low permeability a continuous limestone full of skeletal debris a discontinuous mudstone that contains no faults a halite with faults that have self-healed submit

test Which of these would make a good caprock? a heavily faulted claystone that has low permeability x a continuous limestone full of skeletal debris a discontinuous mudstone that contains no faults a halite with faults that have self-healed submit

test Which of these would make a good caprock? a heavily faulted claystone that has low permeability a continuous limestone full of skeletal debris x a discontinuous mudstone that contains no faults a halite with faults that have self-healed submit

test Which of these would make a good caprock? a heavily faulted claystone that has low permeability a continuous limestone full of skeletal debris a discontinuous mudstone that contains no faults x a halite with faults that have self-healed submit
test unfortunately, this is not correct try again skip test

test Which of these would make a good caprock? a heavily faulted claystone that has low permeability a continuous limestone full of skeletal debris a discontinuous mudstone that contains no faults x a halite with faults that have self-healed this is correct, good job

trap types There are many different trap geometries which rely upon the presence of a closure. A closure is where structural contours terminate against a caprock (sealing unit). Defining the geometry of a trap can be achieved by a thorough investigation of the: § caprock § migration routes § rock properties Most traps require a combination of multiple seals (top, lateral, bottom) Trapping mechanisms related to the geology of a reservoir e. g. physical trapping are: § structural trapping – formed by tectonic processes that take place after the deposition of sediments or localised mobilisation of salts. § stratigraphic trapping – formed by variations in lithology due to environment of deposition, base level changes or postdepositional diagenetic alteration over time.
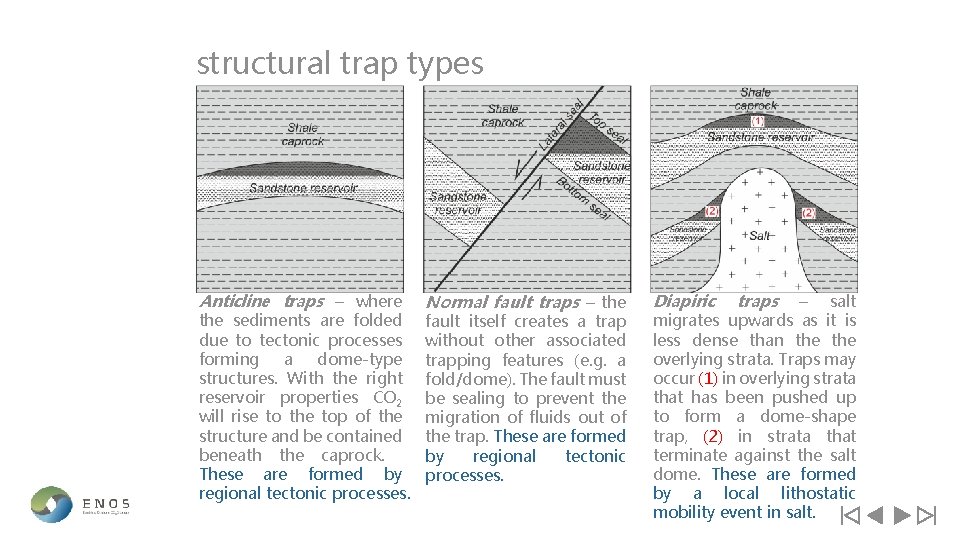
structural trap types Anticline traps – where the sediments are folded due to tectonic processes forming a dome-type structures. With the right reservoir properties CO 2 will rise to the top of the structure and be contained beneath the caprock. These are formed by regional tectonic processes. Normal fault traps – the fault itself creates a trap without other associated trapping features (e. g. a fold/dome). The fault must be sealing to prevent the migration of fluids out of the trap. These are formed by regional tectonic processes. Diapiric traps – salt migrates upwards as it is less dense than the overlying strata. Traps may occur (1) in overlying strata that has been pushed up to form a dome-shape trap, (2) in strata that terminate against the salt dome. These are formed by a local lithostatic mobility event in salt.
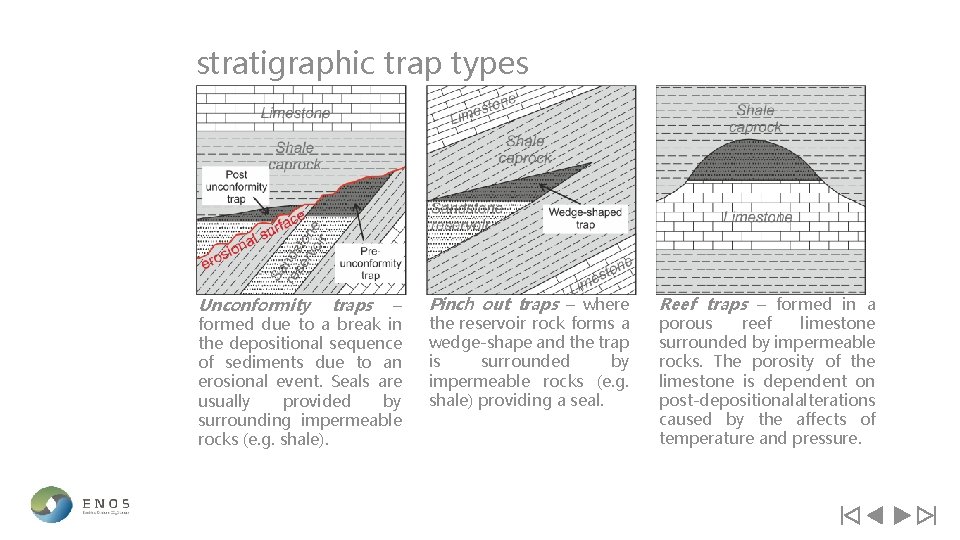
stratigraphic trap types Unconformity traps – formed due to a break in the depositional sequence of sediments due to an erosional event. Seals are usually provided by surrounding impermeable rocks (e. g. shale). Pinch out traps – where the reservoir rock forms a wedge-shape and the trap is surrounded by impermeable rocks (e. g. shale) providing a seal. Reef traps – formed in a porous reef limestone surrounded by impermeable rocks. The porosity of the limestone is dependent on post-depositionalalterations caused by the affects of temperature and pressure.

stratigraphic trap types (cont. ) Diagentic trap – locally modified porosity and peremeability due to cementation where chemical reactions have taken place. These processes can both enhance and reduce the porosity and permeability. Shoestring sandstone traps – long narrow lenses of sandstone surrounded by shales. They are ususally deposited as beaches, barrier islands or spits.

test Which of the following are structural trapping mechanisms? normal fault traps pinch-out traps unconformity traps diapiric traps skip test

test Which of the following are structural trapping mechanisms? x normal fault traps pinch-out traps unconformity traps diapiric traps submit

test Which of the following are structural trapping mechanisms? normal fault traps x pinch-out traps unconformity traps diapiric traps submit

test Which of the following are structural trapping mechanisms? normal fault traps pinch-out traps x unconformity traps diapiric traps submit

test Which of the following are structural trapping mechanisms? normal fault traps pinch-out traps unconformity traps x diapiric traps submit

test Which of the following are structural trapping mechanisms? x normal fault traps x pinch-out traps unconformity traps diapiric traps submit

test Which of the following are structural trapping mechanisms? x normal fault traps pinch-out traps x unconformity traps diapiric traps submit

test Which of the following are structural trapping mechanisms? x normal fault traps pinch-out traps unconformity traps x diapiric traps submit

test Which of the following are structural trapping mechanisms? normal fault traps x pinch-out traps x unconformity traps diapiric traps submit

test Which of the following are structural trapping mechanisms? normal fault traps x pinch-out traps unconformity traps x diapiric traps submit

test Which of the following are structural trapping mechanisms? normal fault traps pinch-out traps x unconformity traps x diapiric traps submit

test Which of the following are structural trapping mechanisms? x normal fault traps x pinch-out traps x unconformity traps diapiric traps submit

test Which of the following are structural trapping mechanisms? x normal fault traps x pinch-out traps unconformity traps x diapiric traps submit

test Which of the following are structural trapping mechanisms? x normal fault traps pinch-out traps x unconformity traps x diapiric traps submit

test Which of the following are structural trapping mechanisms? normal fault traps x pinch-out traps x unconformity traps x diapiric traps submit

test Which of the following are structural trapping mechanisms? x normal fault traps x pinch-out traps x unconformity traps x diapiric traps submit
test unfortunately, this is not correct try again skip test

test Which of the following are structural trapping mechanisms? x normal fault traps pinch-out traps unconformity traps x diapiric traps this is correct, good job
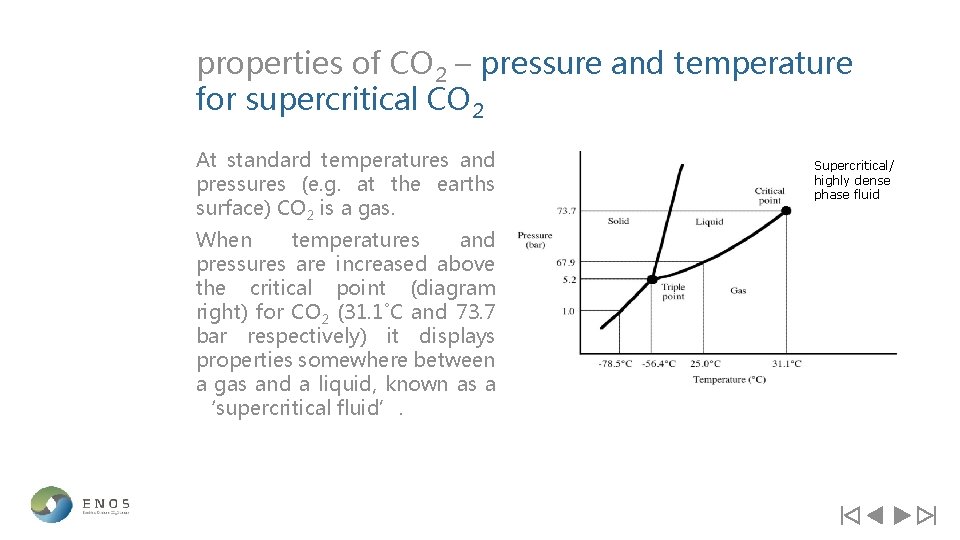
properties of CO 2 – pressure and temperature for supercritical CO 2 At standard temperatures and pressures (e. g. at the earths surface) CO 2 is a gas. When temperatures and pressures are increased above the critical point (diagram right) for CO 2 (31. 1˚C and 73. 7 bar respectively) it displays properties somewhere between a gas and a liquid, known as a ‘supercritical fluid’. Supercritical/ highly dense phase fluid
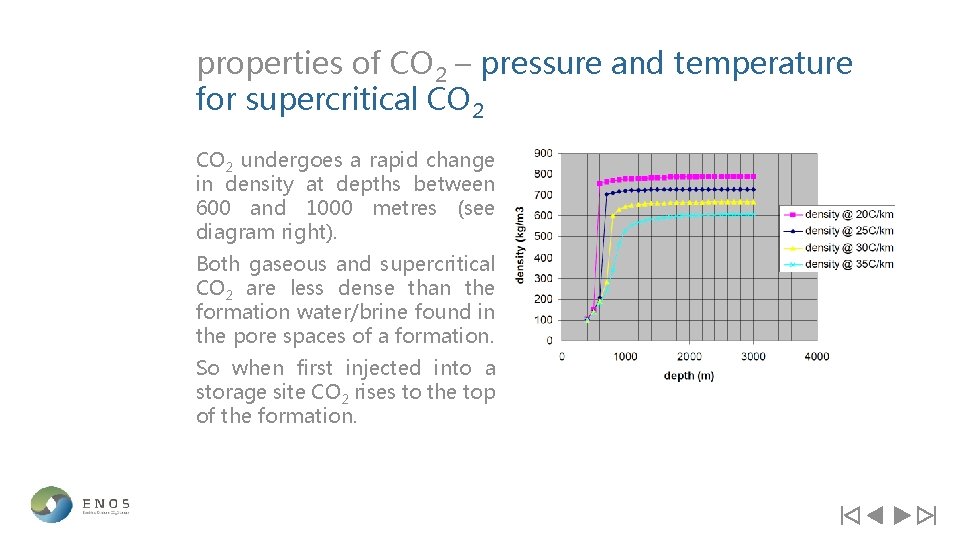
properties of CO 2 – pressure and temperature for supercritical CO 2 undergoes a rapid change in density at depths between 600 and 1000 metres (see diagram right). Both gaseous and supercritical CO 2 are less dense than the formation water/brine found in the pore spaces of a formation. So when first injected into a storage site CO 2 rises to the top of the formation.

properties of CO 2 – how big is one tonne of CO 2 and why do we store CO 2 at depth? § To add perspective: § An adult polar bear weighs approximately one tonne § A medium car weighs up to 1. 2 tonnes § One tonne of CO 2 at the earths surface would occupy the equivalent of a box that is around 8 metres high by 8 metres wide by 8 metres deep (537 cubic metres). § One tonne of CO 2 at a depth of 800 metres or greater occupies much less space - equivalent of a box of about 1. 15 metres high by 1. 15 metres wide by 1. 15 metres deep (1. 54 cubic metres) – therefore more CO 2 can be stored at depth.

properties of CO 2 – density and viscosity CO 2 injected at depths greater than 800 metres will be a supercritical dense phase fluid. Initially the CO 2 will buoyantly rise up through the reservoir because as a supercritical fluid it is less dense than the native fluid (brine). It will continue to rise until it is trapped by a barrier, usually an impermeable caprock. Over time, with the right conditions (temperature and water salinity etc. ) injected CO 2 may start to dissolve into the native brine, a process known as solubility trapping (described in more detail later on). The CO 2 -rich brine will start to sink toward the bottom of a reservoir formation as it becomes more dense than the non-saturated brine at a rate that is influenced by viscosity as well as other factors. Viscosity is the property that is a fluid’s resistance to flow and relates to its thickness e. g. honey has a higher viscosity than water. Injected supercritical (fluid) CO 2 has a much lower viscosity than the native brine in a geological formation [Eke et al. , 2011].

properties of CO 2 – density and viscosity (cont. ) The downward motion of the plumes along with the difference in viscosity of the CO 2 -rich brine and the native brine creates circulation of fluids in the reservoir and allows undissolved CO 2 to come into contact with fresh brine [Ennis-King & Paterson, 2003]. This process helps to increase the rate of dissolution for solubility trapping [Mc. Bride-Wight, 2015]. Knowledge of the CO 2 density and viscosity therefore is essential for reservoir flow simulations to model injection and long-term fate in a storage project [Mc. Bride-Wight, 2015]. Darcy’s law formulated by Henry Darcy describes the flow of fluid through a porous medium e. g. a reservoir rock. This was based on the results of experiments that he carried out on the flow of water through sands. Morris Muskat later refined Darcy‘s equation (shown in more detail next) for single phase flow (e. g. a fluid) by including the parameter viscosity.

properties of CO 2 – density and viscosity (cont. ) Darcy’s Law Fluid flow in the subsurface as represented by Darcy’s Law, an equation that defines a fluids ability to flow through a porous media e. g. a reservoir rock. Reliant upon the amount of flow between two points Darcy’s Law is directly related to: § the pressure difference between the two points § the distance between those two points § the interconnectivity of flow pathways in the rock between the points. Equation as refined by Morris Muskat: Where: Q – the total discharge (volume per time) K – intrinsic permeability of the medium (m 2) A – area of flow (m 2) pb−pa – total pressure drop between points a and b (pascals) L – length over which the pressure drops (m) µ – viscosity The negative sign is needed because fluid flows from high pressure to low pressure.

test What happens to CO 2 at depths greater than 800 metres? It becomes a gas and occupies more space than it would at the surface It becomes a supercritical fluid and occupies more space than it would at the surface It becomes a gas and occupies less space than it would at the surface It becomes a supercritical fluid and occupies less space than it would at the surface skip test

test What happens to CO 2 at depths greater than 800 metres? x It becomes a gas and occupies more space than it would at the surface It becomes a supercritical fluid and occupies more space than it would at the surface It becomes a gas and occupies less space than it would at the surface It becomes a supercritical fluid and occupies less space than it would at the surface submit

test What happens to CO 2 at depths greater than 800 metres? It becomes a gas and occupies more space than it would at the surface x It becomes a supercritical fluid and occupies more space than it would at the surface It becomes a gas and occupies less space than it would at the surface It becomes a supercritical fluid and occupies less space than it would at the surface submit

test What happens to CO 2 at depths greater than 800 metres? It becomes a gas and occupies more space than it would at the surface It becomes a supercritical fluid and occupies more space than it would at the surface x It becomes a gas and occupies less space than it would at the surface It becomes a supercritical fluid and occupies less space than it would at the surface submit

test What happens to CO 2 at depths greater than 800 metres? It becomes a gas and occupies more space than it would at the surface It becomes a supercritical fluid and occupies more space than it would at the surface It becomes a gas and occupies less space than it would at the surface x It becomes a supercritical fluid and occupies less space than it would at the surface submit
test unfortunately, this is not correct try again skip test

test What happens to CO 2 at depths greater than 800 metres? It becomes a gas and occupies more space than it would at the surface It becomes a supercritical fluid and occupies more space than it would at the surface It becomes a gas and occupies less space than it would at the surface x It becomes a supercritical fluid and occupies less space than it would at the surface this is correct, keep it up

test What contributes to the mixing of CO 2 in the native brine in a geological formation? introduction of water to the system dissolution density and viscosity difference between the CO 2 and brine upward movement of CO 2 in the geological formation skip test

test What contributes to the mixing of CO 2 in the native brine in a geological formation? x introduction of water to the system dissolution density and viscosity difference between the CO 2 and brine upward movement of CO 2 in the geological formation submit

test What contributes to the mixing of CO 2 in the native brine in a geological formation? introduction of water to the system x dissolution density and viscosity difference between the CO 2 and brine upward movement of CO 2 in the geological formation submit

test What contributes to the mixing of CO 2 in the native brine in a geological formation? introduction of water to the system dissolution x density and viscosity difference between the CO 2 and brine upward movement of CO 2 in the geological formation submit

test What contributes to the mixing of CO 2 in the native brine in a geological formation? introduction of water to the system dissolution density and viscosity difference between the CO 2 and brine x upward movement of CO 2 in the geological formation submit

test What contributes to the mixing of CO 2 in the native brine in a geological formation? x introduction of water to the system x dissolution density and viscosity difference between the CO 2 and brine upward movement of CO 2 in the geological formation submit

test What contributes to the mixing of CO 2 in the native brine in a geological formation? x introduction of water to the system dissolution x density and viscosity difference between the CO 2 and brine upward movement of CO 2 in the geological formation submit

test What contributes to the mixing of CO 2 in the native brine in a geological formation? x introduction of water to the system dissolution density and viscosity difference between the CO 2 and brine x upward movement of CO 2 in the geological formation submit

test What contributes to the mixing of CO 2 in the native brine in a geological formation? introduction of water to the system x dissolution x density and viscosity difference between the CO 2 and brine upward movement of CO 2 in the geological formation submit

test What contributes to the mixing of CO 2 in the native brine in a geological formation? introduction of water to the system x dissolution density and viscosity difference between the CO 2 and brine x upward movement of CO 2 in the geological formation submit

test What contributes to the mixing of CO 2 in the native brine in a geological formation? introduction of water to the system dissolution x density and viscosity difference between the CO 2 and brine x upward movement of CO 2 in the geological formation submit

test What contributes to the mixing of CO 2 in the native brine in a geological formation? x introduction of water to the system x dissolution x density and viscosity difference between the CO 2 and brine upward movement of CO 2 in the geological formation submit

test What contributes to the mixing of CO 2 in the native brine in a geological formation? x introduction of water to the system x dissolution density and viscosity difference between the CO 2 and brine x upward movement of CO 2 in the geological formation submit

test What contributes to the mixing of CO 2 in the native brine in a geological formation? x introduction of water to the system dissolution x density and viscosity difference between the CO 2 and brine x upward movement of CO 2 in the geological formation submit

test What contributes to the mixing of CO 2 in the native brine in a geological formation? introduction of water to the system x dissolution x density and viscosity difference between the CO 2 and brine x upward movement of CO 2 in the geological formation submit

test What contributes to the mixing of CO 2 in the native brine in a geological formation? x introduction of water to the system x dissolution x density and viscosity difference between the CO 2 and brine x upward movement of CO 2 in the geological formation submit
test unfortunately, this is not correct try again skip test

test What contributes to the mixing of CO 2 in the native brine in a geological formation? introduction of water to the system x dissolution x density and viscosity difference between the CO 2 and brine upward movement of CO 2 in the geological formation this is correct, good job

injectivity and pressure buildup Injection of CO 2 into a geological formations can lead to unacceptable pressure rise, geomechanical effects and opening or creation of new escape pathways. During CO 2 storage operation schemes care must be taken not to exceed the safe amount of pressure in the formation being used. § the safe pressure in the case of hydrocarbon fields is the initial pressure of the formation before production. § safe pressure of a saline aquifer may be more difficult to manage due to no fluids and/or gas being extracted prior to injection of CO 2 for storage. Sufficient injectivity (rate at which CO 2 can be injected into a geological formation) can be achieved if the permeability of the storage formation is high enough (e. g. more than 200 m. D).

injectivity and pressure buildup (cont. ) Displacement of pore fluids must be achieved in saline aquifer storage sites by either: § displacing the original pore fluid (so injection pressure must be greater than virgin pore fluid pressure) § allowing the displaced pore fluid to leak off. When injecting CO 2 into a saline aquifer there will be a progressive pore fluid pressure increase around the injection wells. Native pore fluids may therefore: § migrate to the ground surface/seabed § cause overpressuring in adjacent formations resulting in displacement of their pore fluids § result in movement of highly saline water into potable water supplies. Large-scale buildup of pressure during CO 2 injection can: § have an impact on caprock integrity § cause reactivation/propagation of faults § cause migration of CO 2 and/or brine into the shallow subsurface § affect hydrocarbon resources.

injectivity and pressure buildup (cont. ) Unacceptable pressures can bring a storage operation to a halt. If the pressure can’t be managed this can have an impact on the available storage capacity of potential storage formations. The magnitude and lateral extent of pressure increase during injection is determined by a number of parameters including (but not limited to): § porosity § permeability § thickness of reservoir § extent of the reservoir § CO 2 injection rate § number and placement of injection wells § barriers to fluid flow § water extracted from the reservoir.

relative injectivity of potential storage sites Saline water-bearing reservoir rocks (saline aquifers) § injectivity should be okay if the formation has reasonable permeability and is not compartmentalised Oil fields (enhanced oil recovery) § also should be okay due to lower pore fluid pressure after depletion; fluid recycling is often carried out during EOR Gas fields § injectivity should be good; most gas fields have low pressures once they are depleted Coal seams § injectivity can be a major issue due to low permeabilities – lots of wells would be needed to inject significant volumes of CO 2

pressure management If large volumes of fluid are to be displaced, the reservoir needs to be sufficiently permeable and also needs a connection, albeit tortuous, to the seabed or ground surface. Otherwise the pore fluid pressure in the reservoir will rise and eventually prevent further injection Pressure management is important to reduce the risk of caprock failure and the reduction in injection rates: § in ‘closed-system’ storage sites where there are lateral barriers preventing escape of native fluids § in ‘open-systems’ to ensure pressure increases don‘t adversly affect neighbouring storage sites. Understanding the pore pressure distribution is essential to ensure optimal storage efficiency. Monitoring pore pressure changes can detect deviations from the expected pressure response (predicted with computer models) from the storage site. Ensuring the long-term safety and conformance of the storage complex forms a fundamental prerequisite for an operators’ CCS investment decision.

pressure management – fluid production One method of managing the pressure is by extraction of native fluids from CO 2 storage sites [Birkholzer et al. , 2012]. There are two distinct categories of fluid production: § active fluid production involves pumping fluid from the reservoir through wells at a specific rate. It can commence before CO 2 injection is initiated. § passive fluid production is a method which allows fluid to be extracted from the reservoir, driven by pressure increases above hydrostatic values [Bergmo et al. , 2011]. This method benefits from not requiring pumping equipment or power at the site.

pressure management – fluid production (cont. ) The fluid production method may: § increase storage capacity § reduce the risk of failure § allow more control on the migration flow of the injected CO 2 (with careful placement of extraction wells). Fluid production increases the costs to any CO 2 storage operation due to the requirement to drill additional extraction wells plus installation of pumping equipment (in the case of active fluid production). This method also introduces the additional problem (particularly for offshore sites) of how to dispose of the extracted fluids.

pressure management – injection well placement Optimal distribution of CO 2 within a storage site is important for managing pressure during an injection operation. Computer models can be used to plan numbers and location of injection wells as well as the injection rates in each of those wells (this may be partly guided by supply rate of CO 2). These models have shown that the pressure footprint from an injection operation extends much further than the footprint of the CO 2; see diagram right [Noy et al. , 2012].
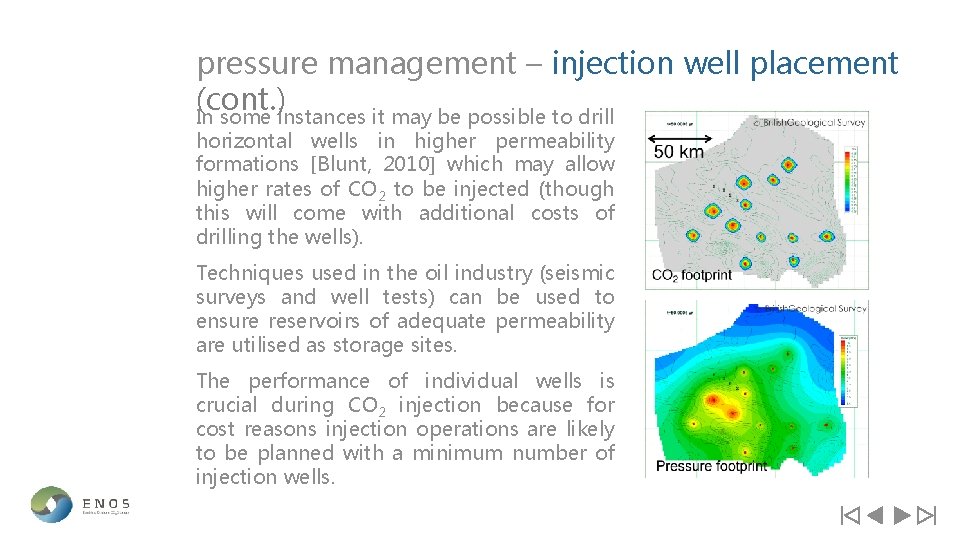
pressure management – injection well placement (cont. ) In some instances it may be possible to drill horizontal wells in higher permeability formations [Blunt, 2010] which may allow higher rates of CO 2 to be injected (though this will come with additional costs of drilling the wells). Techniques used in the oil industry (seismic surveys and well tests) can be used to ensure reservoirs of adequate permeability are utilised as storage sites. The performance of individual wells is crucial during CO 2 injection because for cost reasons injection operations are likely to be planned with a minimum number of injection wells.
test Which of these methods can help to manage pressure during injection of CO 2? fluid extraction injecting as fast as possible injecting water as well as CO 2 careful placement of injection wells skip test
test Which of these methods can help to manage pressure during injection of CO 2? x fluid extraction injecting as fast as possible injecting water as well as CO 2 careful placement of injection wells submit
test Which of these methods can help to manage pressure during injection of CO 2? fluid extraction x injecting as fast as possible injecting water as well as CO 2 careful placement of injection wells submit
test Which of these methods can help to manage pressure during injection of CO 2? fluid extraction injecting as fast as possible x injecting water as well as CO 2 careful placement of injection wells submit
test Which of these methods can help to manage pressure during injection of CO 2? fluid extraction injecting as fast as possible injecting water as well as CO 2 x careful placement of injection wells submit
test Which of these methods can help to manage pressure during injection of CO 2? x fluid extraction x injecting as fast as possible injecting water as well as CO 2 careful placement of injection wells submit
test Which of these methods can help to manage pressure during injection of CO 2? x fluid extraction injecting as fast as possible x injecting water as well as CO 2 careful placement of injection wells submit
test Which of these methods can help to manage pressure during injection of CO 2? x fluid extraction injecting as fast as possible injecting water as well as CO 2 x careful placement of injection wells submit
test Which of these methods can help to manage pressure during injection of CO 2? fluid extraction x injecting as fast as possible x injecting water as well as CO 2 careful placement of injection wells submit
test Which of these methods can help to manage pressure during injection of CO 2? fluid extraction x injecting as fast as possible injecting water as well as CO 2 x careful placement of injection wells submit
test Which of these methods can help to manage pressure during injection of CO 2? fluid extraction injecting as fast as possible x injecting water as well as CO 2 x careful placement of injection wells submit
test Which of these methods can help to manage pressure during injection of CO 2? x fluid extraction x injecting as fast as possible x injecting water as well as CO 2 careful placement of injection wells submit
test Which of these methods can help to manage pressure during injection of CO 2? x fluid extraction x injecting as fast as possible injecting water as well as CO 2 x careful placement of injection wells submit
test Which of these methods can help to manage pressure during injection of CO 2? x fluid extraction injecting as fast as possible x injecting water as well as CO 2 x careful placement of injection wells submit
test Which of these methods can help to manage pressure during injection of CO 2? fluid extraction x injecting as fast as possible x injecting water as well as CO 2 x careful placement of injection wells submit
test Which of these methods can help to manage pressure during injection of CO 2? x fluid extraction x injecting as fast as possible x injecting water as well as CO 2 x careful placement of injection wells submit
test unfortunately, this is not correct try again skip test

test Which of these methods can help to manage pressure during injection of CO 2? x fluid extraction injecting as fast as possible injecting water as well as CO 2 x careful placement of injection wells this is correct, keep it up

test Pressure buildup during injection into a geological formation can … result in little movement of the pore fluid have a positive effect in the caprock cause native fluids to move into neighbouring formations make storage of CO 2 more effective skip test

test Pressure buildup during injection into a geological formation can … x result in little movement of the pore fluid have a positive effect in the caprock cause native fluids to move into neighbouring formations make storage of CO 2 more effective submit

test Pressure buildup during injection into a geological formation can … result in little movement of the pore fluid x have a positive effect in the caprock cause native fluids to move into neighbouring formations make storage of CO 2 more effective submit

test Pressure buildup during injection into a geological formation can … result in little movement of the pore fluid have a positive effect in the caprock x cause native fluids to move into neighbouring formations make storage of CO 2 more effective submit

test Pressure buildup during injection into a geological formation can … result in little movement of the pore fluid have a positive effect in the caprock cause native fluids to move into neighbouring formations x make storage of CO 2 more effective submit
test unfortunately, this is not correct try again skip test
test Pressure buildup during injection into a geological formation can … result in little movement of the pore fluid have a positive effect in the caprock x cause native fluids to move into neighbouring formations make storage of CO 2 more effective this is correct, good job

trapping mechanisms Once in the reservoir rock the CO 2 will be retained by one or more trapping mechanisms. According to the Carbon Sequestration Leadership Forum (CSLF) there are two types of trapping mechanism that can occur during the geological storage of CO 2: § physical trapping § chemical trapping Injected CO 2 under certain conditions may migrate in the subsurface extremely slowly and could become trapped via a combination of physical or chemical trapping, this is described as hydrodynamic trapping by the CSLF [Bachu et al. , 2007]. These mechanisms occur over different timescales during the lifetime of CO 2 storage.
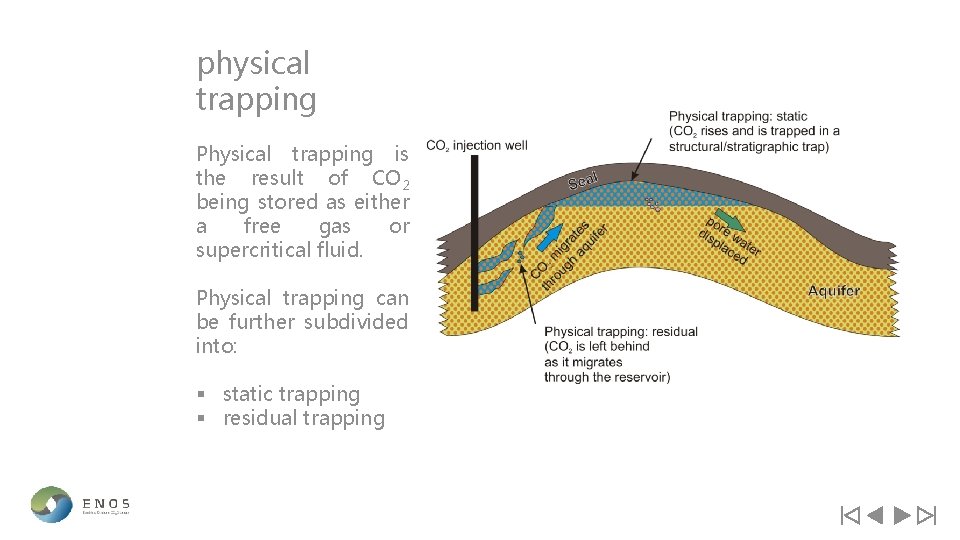
physical trapping Physical trapping is the result of CO 2 being stored as either a free gas or supercritical fluid. Physical trapping can be further subdivided into: § static trapping § residual trapping
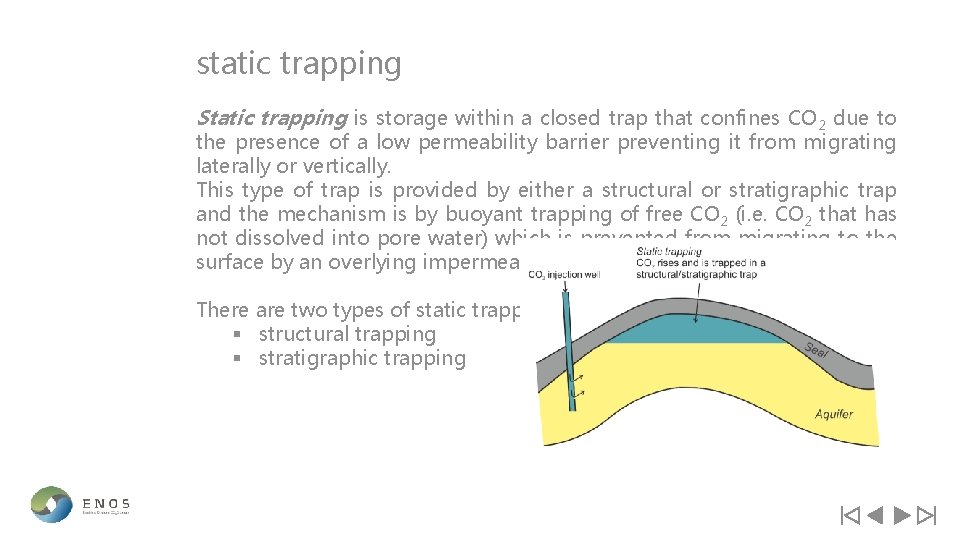
static trapping Static trapping is storage within a closed trap that confines CO 2 due to the presence of a low permeability barrier preventing it from migrating laterally or vertically. This type of trap is provided by either a structural or stratigraphic trap and the mechanism is by buoyant trapping of free CO 2 (i. e. CO 2 that has not dissolved into pore water) which is prevented from migrating to the surface by an overlying impermeable barrier. There are two types of static trapping: § structural trapping § stratigraphic trapping

residual-saturation traps Residual-saturation trapping in pore spaces occurs as a plume CO 2 moves through the reservoir. Some of the CO 2 remains within the pore spaces of rock due to capillary forces and small portions of CO 2 ‘snaps-off’ from the trailing edge of the migrating plume. Residual gas trapping is primarily linked with hydrodynamic trapping where CO 2 is left along the path of a migrating plume. This mainly takes place after injection has stopped [Bachu et al. , 2007] but while the plume of CO 2 is still migrating [Juanes et al. , 2006]. Diagram illustrating residual saturation trapping

residual-saturation traps (cont. ) Suitable geologic formations for residual trapping are water wet e. g. saline aquifers. They provide suitable conditions to initiate snap-off of the non-wetting gas phase at the trailing edge of the plume [Juanes et al. , 2006]. Injecting CO 2 at higher rates is more likely to initiate ‘snap-off’ as this allows more CO 2 to enter smaller pore spaces within the storage formation [Juanes et al. , 2006]. It has also been suggested that a combination of formation permeability, dip, and density difference between CO 2 and in-situ fluids all contribute to the amount that will be trapped residually Diagram illustrating residual saturation trapping

chemical trapping Chemical trapping is the result of a reaction between the CO 2, pore fluid and reservoir matrix. This may increase the amount and the effectiveness of CO 2 that can be stored. There are three types of chemical trapping: § solubility/dissolution trapping – CO 2 dissolves into the formation water § mineral trapping – some dissolved CO 2 may be converted to a stable mineral § adsorption trapping – preferential exchange of CO 2 for methane in coal

solubility/dissolution trapping Dissolution trapping is where CO 2 dissolves into the pore water (commonly a brine solution). This type of trapping is dependent on the temperature, pressure and salinity of the native pore fluid. CO 2 solubility (ability to dissolve) in formation water is decreased as temperature and salinity is increased [Metz et al. , 2005]. Dissolution may be quick at first but once the pore water is saturated the rate slows down and is controlled by diffusion and convection rates [Metz et al. , 2005]. As the CO 2 dissolves into the pore water it becomes more dense, at this point the bottom edge of the CO 2 becomes unstable and starts to form many small descending plumes/fingers; see images right of laboratory experiments performed during the EU CO 2 CARE project [Vosper et al. , 2014].

solubility/dissolution trapping (cont. ) In the experiments performed for the CO 2 CARE project the descending plumes displace CO 2 -poor water, and this rises to replace the descending water creating fluid circulation. This rising water then becomes saturated with CO 2 and also descends. Fluid circulation in the system contributes to this method of trapping. Once dissolved the CO 2 remains trapped in solution in the pore water. There is potential for large volumes of CO 2 to be trapped in this way, but it is dependent on the solubility of the CO 2 and whether the described mixing occurs within the reservoir. This method of trapping can take tens to thousands of years to dissolve the maximum amount of CO 2 into the pore water.

mineral trapping When CO 2 is dissolved into the pore water a weak acid is created. A chemical reaction may take place between the CO 2 -rich pore fluid and the reservoir rock resulting in formation of new carbonate minerals such as: § Calcite § Dolomite § Siderite § Dawsonite. Chemical reactions are dependent upon the pore water chemistry, rock mineralogy and the length of the migration path. § reactions with carbonate minerals can be rapid (a matter of days) § reactions with silicate minerals occur over much longer timescales (hundreds to thousands of years). Calcite under the microscope. Source – British Geological Survey

mineral/solubility trapping – geochemical issues A number of issues may arise due to the dissolution of CO 2 into the pore fluids: § corrosion of elements of the rock matrix by CO 2/fluid mixtures § this can result in seal failure § dehydration of the seal as CO 2 -rich fluid manages to flow into the caprock § this can result in seal failure § precipitation of minerals in the pore spaces of the reservoir rock § this may result in injection problems and therefore potential to exceed the threshold pore fluid pressure

hydrodynamic trapping Hydrodynamic trapping occurs via a combination of any of the above described mechanisms which may be simultaneous but at different rates and is not specific to physical or chemical trapping. This mechanism is active when CO 2 is injected into an unconfined aquifer (with no lateral barriers) rather than into a physical trap. The CO 2 will migrate in the same direction as the natural flow in the reservoir once away from the point of injection. Eventually, possibly after millions of years, no mobile free-phase CO 2 will remain in the system as it is trapped by residual-saturation trapping, dissolution and mineral precipitation [Bachu et al. , 2007].

adsorption trapping Adsorption trapping occurs where CO 2 is adsorbed onto mineral surfaces, e. g. in coal seams. The coal seam should be: § laterally continuous and vertically isolated from surrounding strata § minimally folded and faulted § have a permeability of at least one to five millidarcies. Adsorption trapping depends on: § coal composition § coal rank (amount of carbon content) – more CO 2 adsorbed with increasing rank § presence of non-organic material (ash) – reduces adsorption capacity § water content – reduces adsorption capacity.

interaction and relative importance of trapping mechanisms § Static trapping is the first important stage in trapping and ensures that the CO 2 cannot leave the storage formation once it has been injected. § Once immobilised in the storage site as long as conditions are right the CO 2 can then start to dissolve into the formation brine/fluid and can be contained via solubility trapping. § Fluid circulation in the storage site increases the amount of dissolution and therefore solubility trapping. § During migration through the storage formation there is potential for small amounts of the migrating plume to ‘snap-off’ allowing the CO 2 to be residually trapped. This trapping method increases the chances of dissolution/solubility trapping. § Once CO 2 is dissolved into the formation brine the likelihood of mineral trapping is increased because of the change in chemistry of the brine containing dissolved CO 2. Each of the methods for trapping are reliant upon at least one of the others occurring first and if conditions are right it could result in the
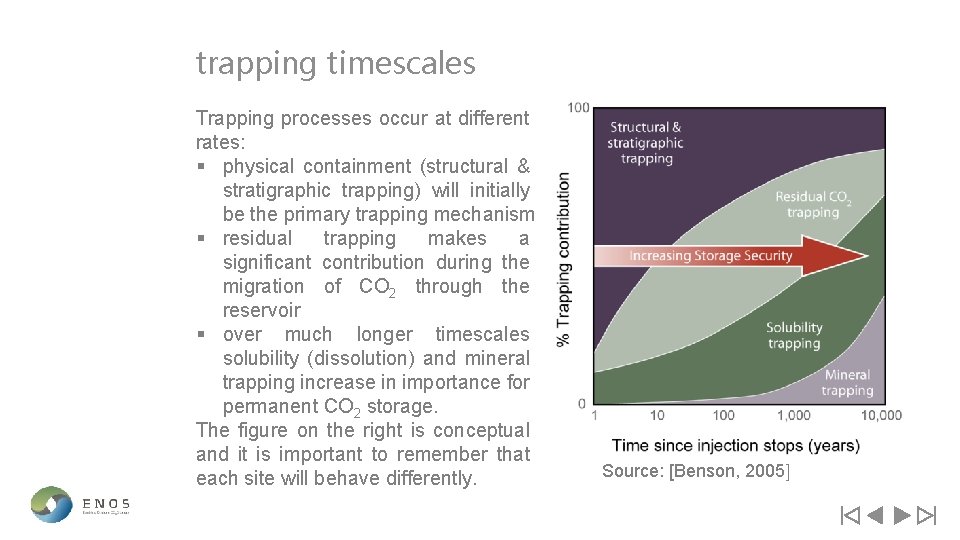
trapping timescales Trapping processes occur at different rates: § physical containment (structural & stratigraphic trapping) will initially be the primary trapping mechanism § residual trapping makes a significant contribution during the migration of CO 2 through the reservoir § over much longer timescales solubility (dissolution) and mineral trapping increase in importance for permanent CO 2 storage. The figure on the right is conceptual and it is important to remember that each site will behave differently. Source: [Benson, 2005]

test Which of these trapping mechanisms occur over the longest timescale? mineral trapping residual trapping solubility trapping static trapping skip test

test Which of these trapping mechanisms occur over the longest timescale? x mineral trapping residual trapping solubility trapping static trapping submit

test Which of these trapping mechanisms occur over the longest timescale? mineral trapping x residual trapping solubility trapping static trapping submit

test Which of these trapping mechanisms occur over the longest timescale? mineral trapping residual trapping x solubility trapping static trapping submit

test Which of these trapping mechanisms occur over the longest timescale? mineral trapping residual trapping solubility trapping x static trapping submit
test unfortunately, this is not correct try again skip test

test Which of these trapping mechanisms occur over the longest timescale? x mineral trapping residual trapping solubility trapping static trapping this is correct, keep it up
test What is dissolution trapping? where CO 2 snaps off and is left behind by the main migrating CO 2 plume a chemical reaction between the CO 2 and pore water results in the formation of a new mineral CO 2 is dissolved into the native pore fluid when CO 2 is trapped beneath an impermeable layer of rock skip test
test What is dissolution trapping? x where CO 2 snaps off and is left behind by the main migrating CO 2 plume a chemical reaction between the CO 2 and pore water results in the formation of a new mineral CO 2 is dissolved into the native pore fluid when CO 2 is trapped beneath an impermeable layer of rock submit
test What is dissolution trapping? where CO 2 snaps off and is left behind by the main migrating CO 2 plume x a chemical reaction between the CO 2 and pore water results in the formation of a new mineral CO 2 is dissolved into the native pore fluid when CO 2 is trapped beneath an impermeable layer of rock submit
test What is dissolution trapping? where CO 2 snaps off and is left behind by the main migrating CO 2 plume a chemical reaction between the CO 2 and pore water results in the formation of a new mineral x CO 2 is dissolved into the native pore fluid when CO 2 is trapped beneath an impermeable layer of rock submit
test What is dissolution trapping? where CO 2 snaps off and is left behind by the main migrating CO 2 plume a chemical reaction between the CO 2 and pore water results in the formation of a new mineral CO 2 is dissolved into the native pore fluid x when CO 2 is trapped beneath an impermeable layer of rock submit
test unfortunately, this is not correct try again skip test
test What is dissolution trapping? where CO 2 snaps off and is left behind by the main migrating CO 2 plume a chemical reaction between the CO 2 and pore water results in the formation of a new mineral x CO 2 is dissolved into the native pore fluid when CO 2 is trapped beneath an impermeable layer of rock this is correct, good job

summary § There a number of options for storage of CO 2 as long as they have: § porous and permeable reservoir at a suitable depth § effective seal § method to trap CO 2. § Coal seams may not be as attractive as hydrocarbon fields and saline aquifers as storage sites due to their low permeability. § CO 2 stored at suitable depths will occupy much less space as a supercritical fluid than at surface conditions as a gas. § Careful pressure management and injection planning is essential for a CO 2 storage operation. This will be site specific due to variations in reservoir characteristics so detailed computer models and simulations must be used.

summary (cont. ) § Density and viscosity of supercritical CO 2 are important properties contributing to circulation of fluids in a storage site to promote dissolution of CO 2 into the formation fluids. § There are several trapping mechanisms that could take place during a storage operation. They occur over different timescales and one could be dependent upon another. § With the right conditions a successful storage operation could result in the permanent storage of CO 2 if minerals are precipitated as part of the trapping mechanism. § It is important to remember that not every site will perform the same so careful investigation must be carried out to understand what might be expected before any operation commences.

parameters m metres mm millimetres m. D millidarcies p. H figure for acidity or alkalinity, where 7 is neutral, lower values are more acid and higher values are more alkaline.

glossary chemical ENOS Enabling Onshore CO 2 Storage in Europe brine water saturated with salt CCS Carbon Dioxide Capture and Storage capillary a tube or pathway with a diameter similar to that of a strand of hair capillary forcesthe caprock preventing pressure the within CO 2 to enter it cleat joint and fractures found within coal seams physical, diagenetic sedimentary (diagenesis) rocks caused by temperatures and pressures as they become consolidated diapiric (diapir) geological structure comprising mobile material often associated with salt in the formation of salt domes dissolution the process of being dissolved

glossary (cont. ) at geomechanical behaviour of rock mechanics lithology physical characteristics of a rock permeability the measure of the ability to flow or transmit liquid or gas through a porous material porosity the measure of the empty spaces in a rock saturated containing the maximum amount of a solute capable of being dissolved under given conditions soluble capable of being dissolved solute a substance dissolved to create a solution substance supercritical fluid a pressure above its critical point where distinct liquid and gas phases do not exist viscosity the property of a fluid that resists the force that causes the fluid to flow

references Bachu, S. , D. Bonijoly, J. Bradshaw, R. Burruss, N-P. Christensen, S. Holloway, and OM. Mathiassen, Estimation of CO 2 Storage Capacity in Geological Media - Phase 2 - Prepared by the Task Force on CO 2 Storage Capacity Estimation for the Technical Group (TG) of the Carbon Sequestration Leadership Forum (CSLF), 2007 Benson, S. and P. Cook (eds. ), Chapter 5: Underground geological storage, In: IPCC Special Report on Carbon dioxide Capture and Storage, 195− 276, 2005 Bergmo, P. , A. A. Grimstad, and E. Lindeberg, Simultaneous CO 2 injection and water production to optimise aquifer storage capacity, International Journal of Greenhouse Gas Control, 5, 555– 564, 2011 Birkholzer, J. T. , A. Cihan, and Q. Zhou, Impact-driven pressure management via targeted brine extraction – Conceptual studies of CO 2 storage in saline formations, International Journal of Greenhouse Gas Control, 7, 168– 180, 2012 Blunt, M. , Carbon dioxide storage, Grantham Institute for Climate Change, Briefing paper No. 4, Imperial College London, 2010 Chadwick, A. , R. Arts, C. Bernstone, F. May, S. Thibeau, and P. Zweigal, Best practice for the storage of CO 2 in saline aquifers, British Geological Survey Occasional Publication No. 14, Keyworth, Nottingham, 2008 Eke, P. E. , M. Naylor, S. Haszeldine, and A. Curtis, CO 2/Brine surface dissolution and injection: CO 2 storage enhancement, SPE Projects, Facilities & Construction, Society of Petroleum Engineers, 2011

references (2) Ennis-King, J. and L. Paterson, Rate of Dissolution Due to Convective Mixing in the Underground Storage of Carbon Dioxide, Proceedings of the 6 th International Conference on Greenhouse Gas Control Technologies, 2003 Gammer, D. , A. Green, S. Holloway, and G. Smith, The Energy Technologies Institute's UK CO 2 storage appraisal project (UKSAP), In: SPE Offshore Europe Oil and Gas Conference, Aberdeen, Scotland, 6– 8 Sept 2011, Society of Petroleum Engineers, 2011 Heidaryan, E. , T. Hatami, M. Rahimi, and J. Moghadasic, Viscosity of pure carbon dioxide at supercritical region: Measurement and correlation approach, Journal of Supercritical Fluids, 56, 144– 151, 2011 Juanes, R. , E. J. Spiteri, F. M. Orr Jr. , and M. J. Blunt, Impact of relative permeability hysteresis on geological CO 2 storage, Water Resources Research, 42, 2006 Kaldi, J. , Storage 1 – Site Selection: Capacity and Injectivity, Presentation for IEAGHG CCS Summer School, Svalbard, 23– 27 August, 2010 Mc. Bride-Wright, M. , G. C. Maitland, and J. P. M. Trusler, Viscosity and Density of Aqueous Solutions of Carbon Dioxide at Temperatures from (274 to 449) K and at Pressures up to 100 Mpa, Journal of Chemical Engineering Data, 60, 171− 180, 2015 Metz, B. , O. Davidson, H. de Coninck, M. Loos, and L. Meyer (eds. ), Special Report on Carbon dioxide Capture and Storage, Intergovernmental Panel on Climate Change, 2005

references (3) Noy, D. J. , S. Holloway, R. A. Chadwick, J. D. O. Williams, S. A. Hannis, and R. W. Lahann, Modelling large-scale carbon dioxide injection into the Bunter Sandstone in the UK Southern North Sea, International Journal of Greenhouse Gas Control, 9, 220− 233, 2012 Vosper, H. , K. Kirk, C. Rochelle, D. Noy and R. A. Chadwick, Does numerical modelling of the onset of dissolution-convection reliably reproduce this key stabilization process in CO 2 Storage? , Energy Procedia, 63, 5341– 5348, 2014 Wang, S. and A. F. Clarens, The effects of CO 2 -brine rheology on leakage processes in geologic carbon sequestration, Water Resources Research, 48, 2012

acknowledgement + disclaimer This lecture was completed with the support of the European Commission and European Union’s Horizon 2020 research and innovation programme under grant agreement No 653718. The contents of this publication do not necessarily reflect the Commission's own position. The document reflects only the author's views and the European Union and its institutions are not liable for any use that may be made of the information contained here. Lecture template by Dorothee Rebscher (BGR)

Carbon Capture and Storag e-book

Carbon Capture and Storag e-book
- Slides: 170