Carbon By Gabriela Segura Alexis Guadarrama Birth of

Carbon By: Gabriela Segura & Alexis Guadarrama

Birth of Your Element: • Symbol: C • This element was known to ancients. • Who and where it was discovered is unknown.

Pictures:

Structure: • Atomic mass: 12. 0107 amu • Atomic Number: 6 • Isotopes: 13 whose half-lives are unknown.

Siblings: • Carbon is located in group number 14. • One famous sibling of carbon is Lead. • Some common properties of elements in this group are that they are very brittle & cannot be rolled into wires.
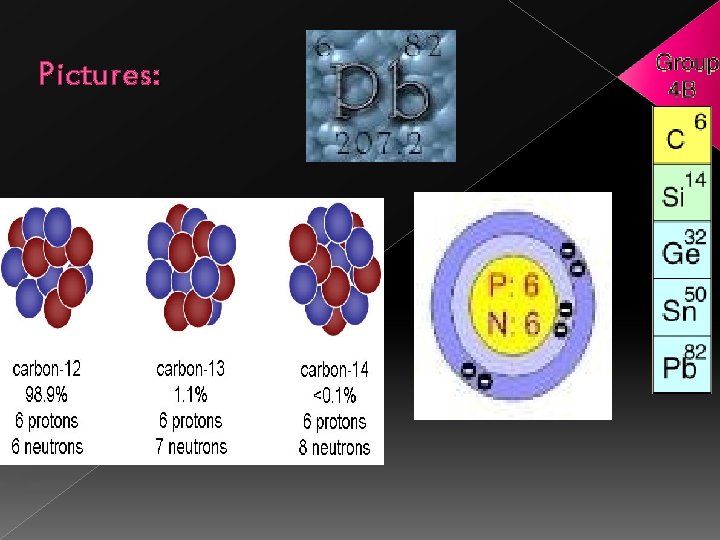
Pictures:

Chemical and physical properties: • • Density: 2. 2670 g per cm cube Color: Black & Transparent Melting Point: 3550 • C State in room temperature: Solid It’s a Nonmetal Common Charges of ion: 4 Common compounds found in Carbon are: Carbon-Dioxide & Carbon-Monoxide.

Pictures: Carbon-Dioxide Carbon-Monoxide

How does Carbons position in the periodic table relate to its properties? • The radii increase as you move down the group, and ionization energies decrease. • Metallic properties increase as you move down the group. • Its position tells us that carbon has only two electrons in the p orbital.

Home: • Carbon is found in nature as a form of amorphous (lampblack, or boneblack), graphite, diamonds, and white carbon. • Diamonds are prized as a gemstone and is used for cutting, drilling, and as a bearing. • Graphite is used as a crucible for melting metals, in pencils, for rust production, for lubrication, and as a moderator for slowing neutrons for atomic fission. • Amorphous carbon is used for removing taste and odors. • Carbon is found everywhere. For instance in water, human beings, and the atmosphere.

Pictures:

Why is this Carbon important? • All living things are based on Carbon. • Carbon makes up only 0. 032% of the Earths crust. • Its also the main component of fuels.

Nickname for this element: • Black Rocky

Interesting facts: • Much of the Carbon on Earth exists in combination with other elements. • Carbon compounds make up the living tissue of all plants and animals. • Most organic compounds consists mainly of carbon combined with hydrogen, nitrogen, and oxygen, in various proportions.

Pictures:

Essay: We would like to adopt Carbon because he’s very interesting. He has many qualities and he’s very important in the earth. Also his very valuable especially found in diamonds. He’s found as carbon-dioxide and carbon-monoxide. He’s very special by the fact that he’s found in every single human being.

Works Cited Bentor, Y. (2012, June 5). Periodic table: Carbon. Retrieved from http: //chemicalelements. com/ "Carbon". (2012, June 5). Carbon element facts. Retrieved from http: //www. chemicool. com/elements/carbon. html Winter, M. (1993 -2012). Carbon: The essentials. Retrieved from http: //www. webelements. com/carbon/
- Slides: 17