Carbon and the Molecular Diversity of Life Life
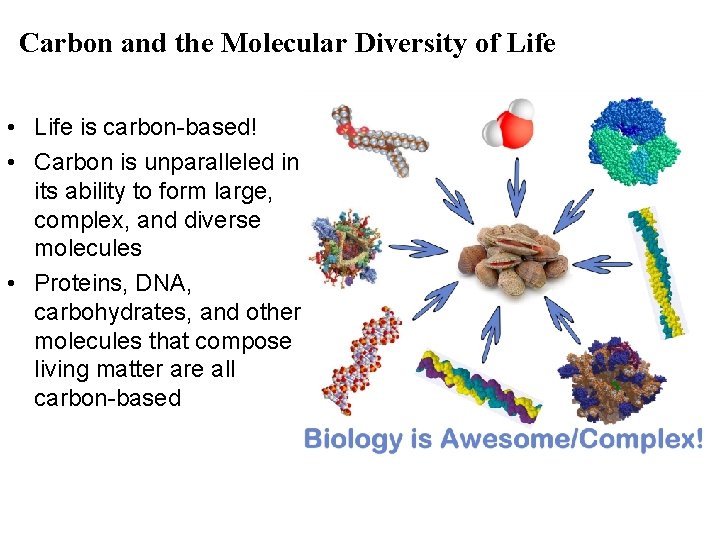
Carbon and the Molecular Diversity of Life • Life is carbon-based! • Carbon is unparalleled in its ability to form large, complex, and diverse molecules • Proteins, DNA, carbohydrates, and other molecules that compose living matter are all carbon-based
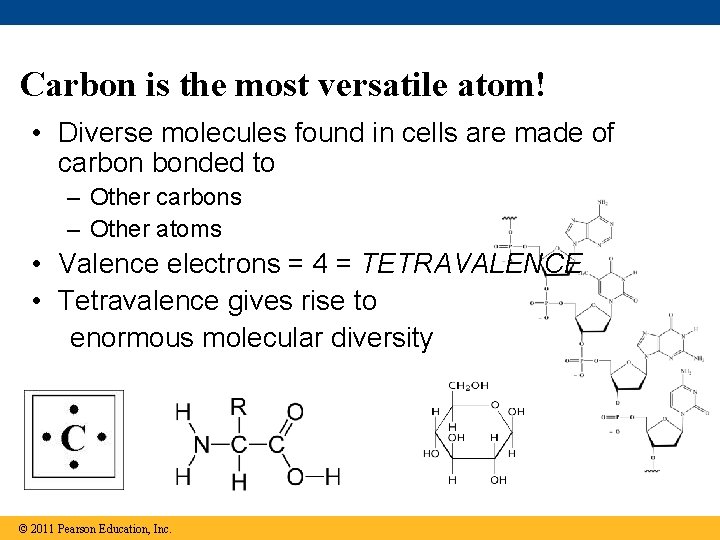
Carbon is the most versatile atom! • Diverse molecules found in cells are made of carbon bonded to – Other carbons – Other atoms • Valence electrons = 4 = TETRAVALENCE • Tetravalence gives rise to enormous molecular diversity © 2011 Pearson Education, Inc.

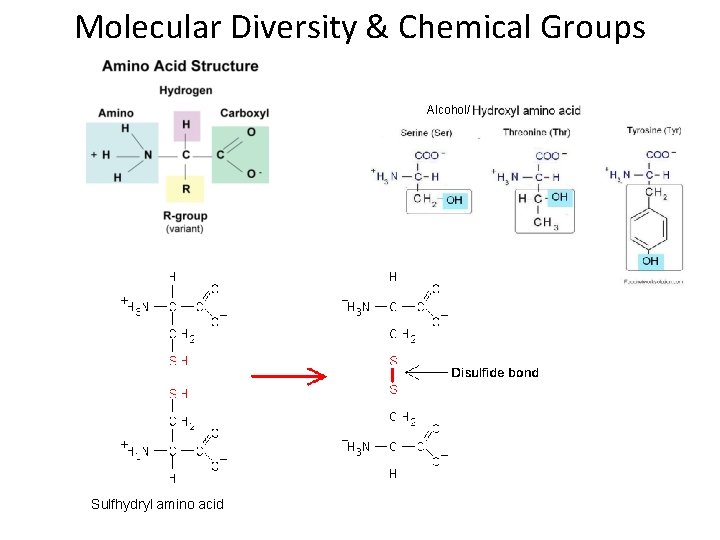
Molecular Diversity & Chemical Groups Alcohol/ Sulfhydryl amino acid
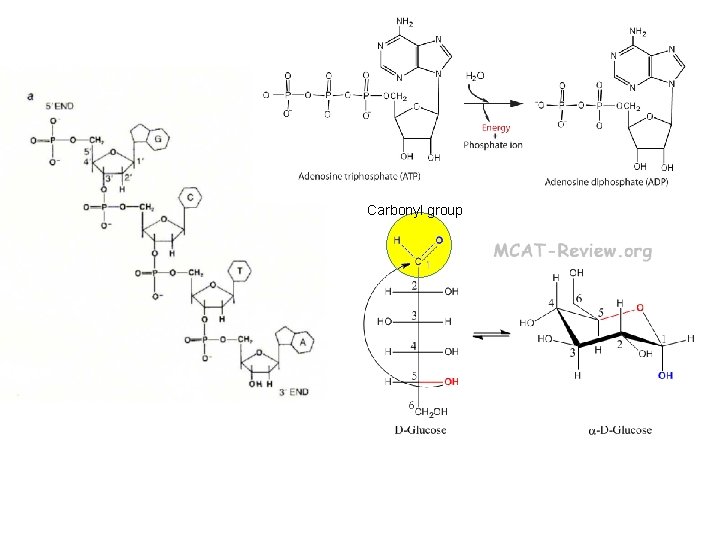
Carbonyl group
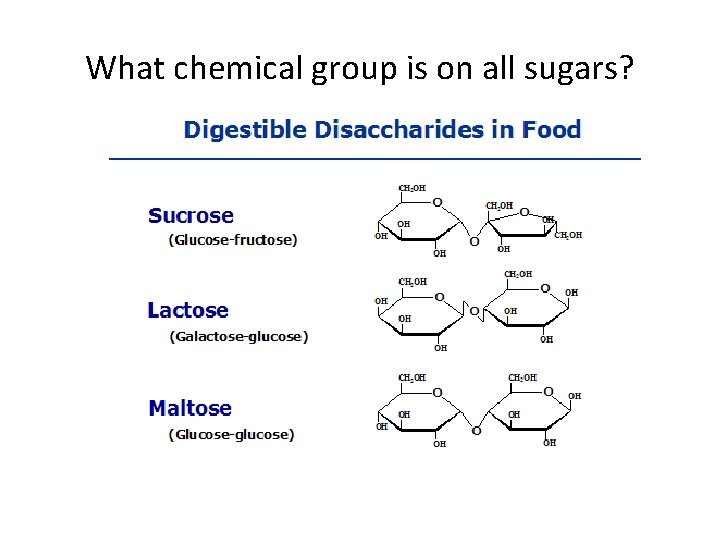
What chemical group is on all sugars?
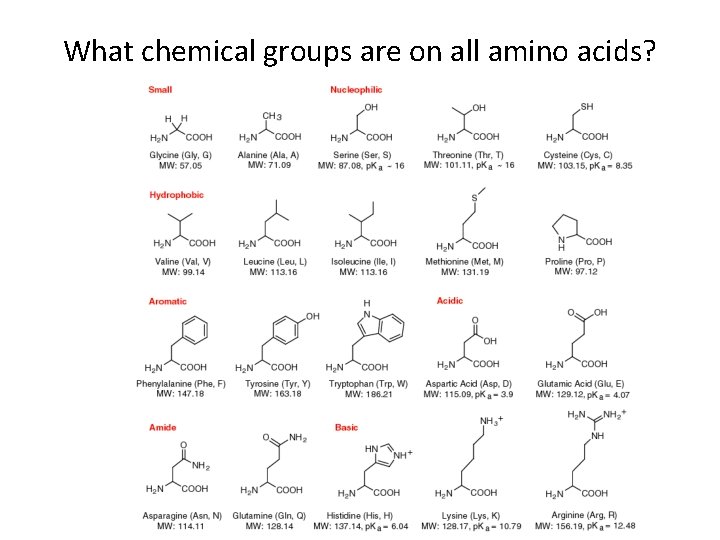
What chemical groups are on all amino acids?
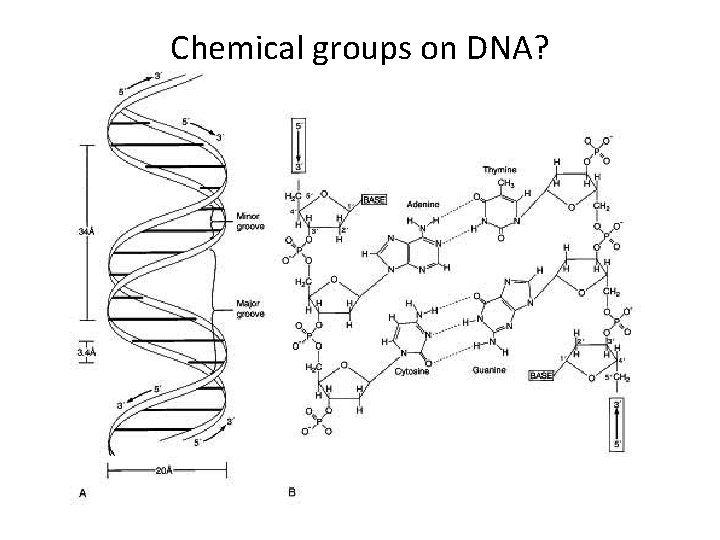
Chemical groups on DNA?
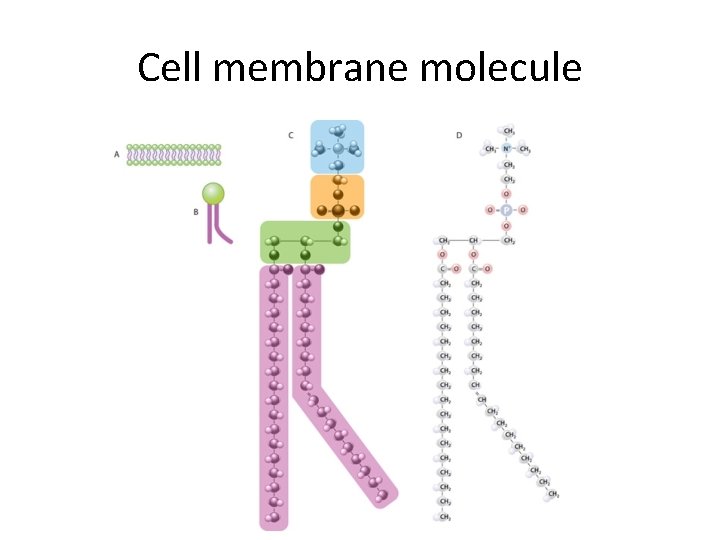
Cell membrane molecule
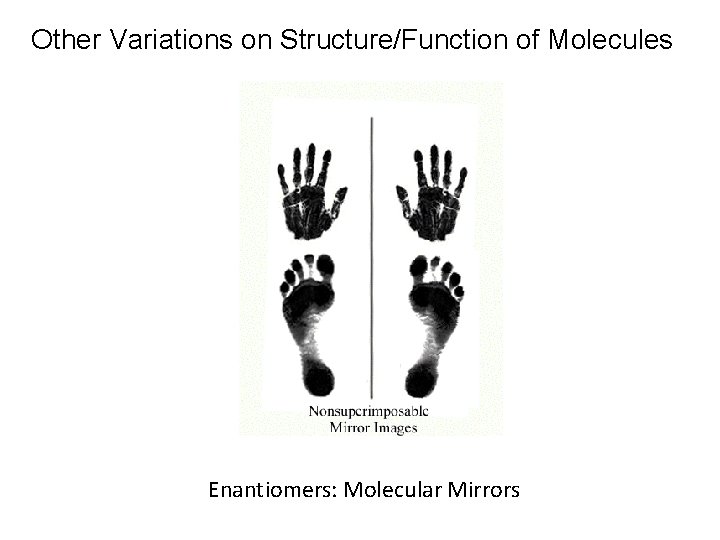
Other Variations on Structure/Function of Molecules Enantiomers: Molecular Mirrors
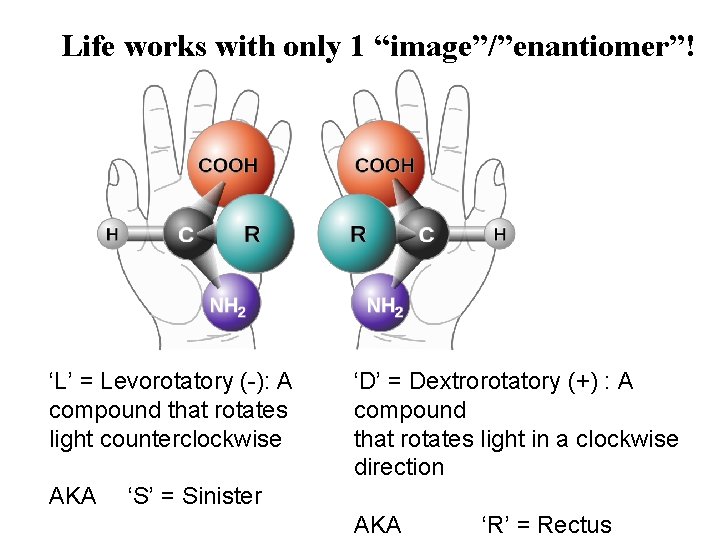
Life works with only 1 “image”/”enantiomer”! ‘L’ = Levorotatory (-): A compound that rotates light counterclockwise AKA ‘D’ = Dextrorotatory (+) : A compound that rotates light in a clockwise direction ‘S’ = Sinister AKA ‘R’ = Rectus

The “Dark Side” of Enantiomers • Enantiomers critical in the pharmaceutical industry • Two enantiomers of a drug may have different effects • Usually one isomer is biologically active
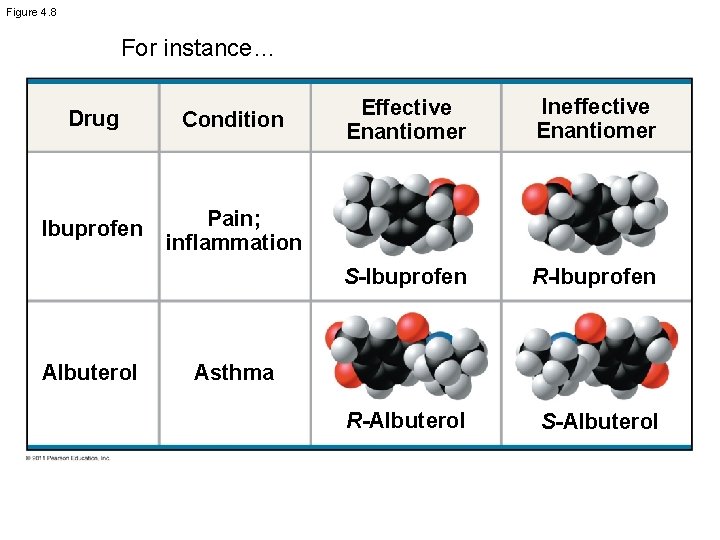
Figure 4. 8 For instance… Drug Condition Ibuprofen Pain; inflammation Albuterol Effective Enantiomer Ineffective Enantiomer S-Ibuprofen R-Albuterol S-Albuterol Asthma
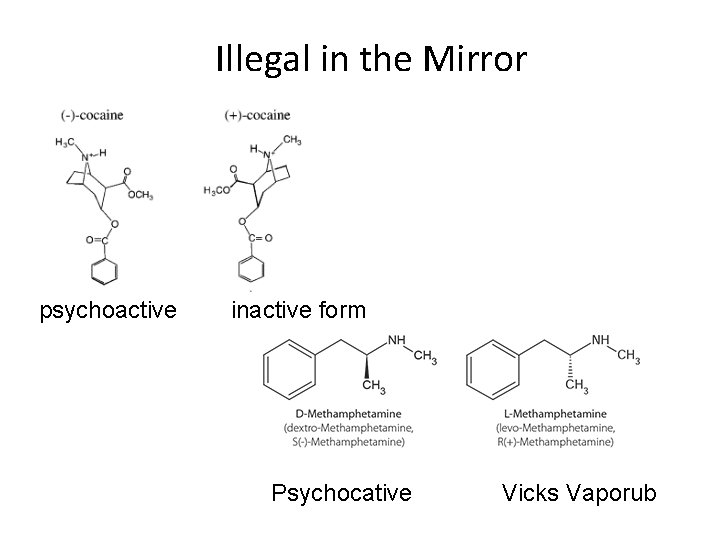
Illegal in the Mirror psychoactive inactive form Psychocative Vicks Vaporub
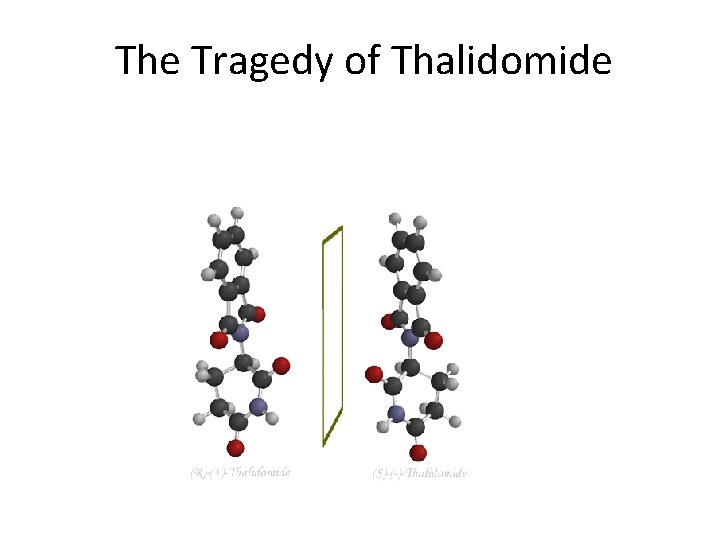
The Tragedy of Thalidomide

Thalidomide’s History • • • Thalidomide was marketed in Europe from 1957 into the early 1960’s to pregnant women to combat many of the symptoms associated with morning sickness. Thalidomide was hailed as a "wonder drug" that provided a "safe, sound sleep” for pregnant women. At the time, many doctors thought that drugs/alcohol could not go from mother to fetus No clinical testing was available to show that Thalidomide molecules could cross the placental wall affecting the fetus until it was too late. Thalidomide became notorious as the killer and disabler of thousands of babies.

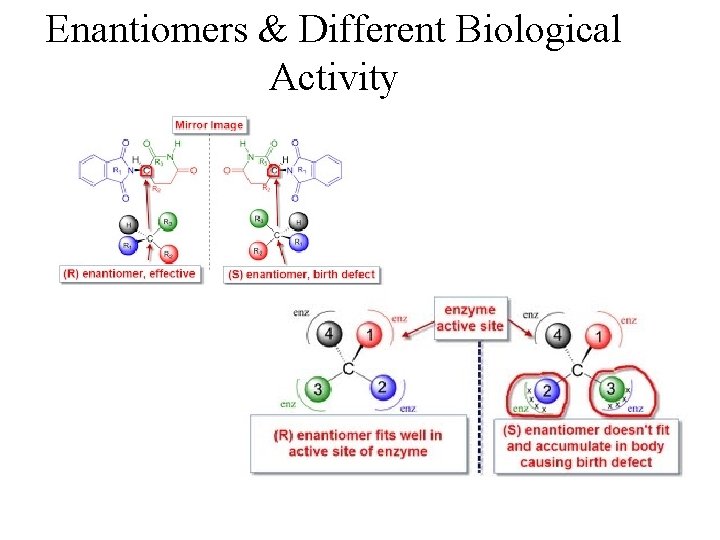
Enantiomers & Different Biological Activity
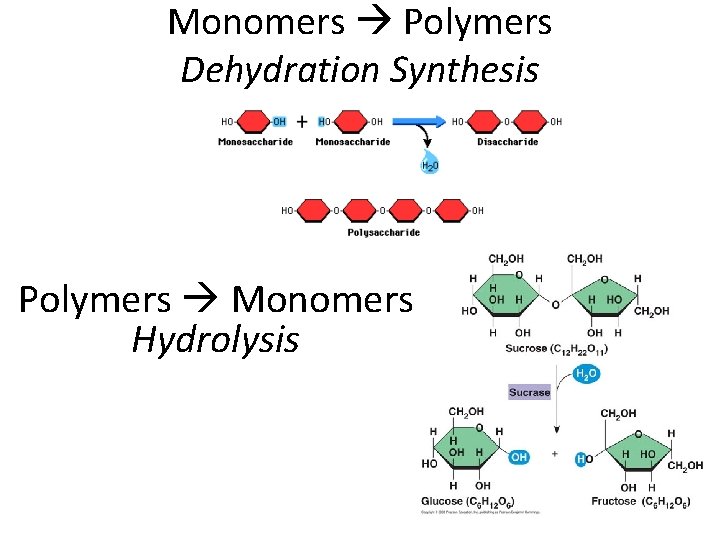
Monomers Polymers Dehydration Synthesis Polymers Monomers Hydrolysis
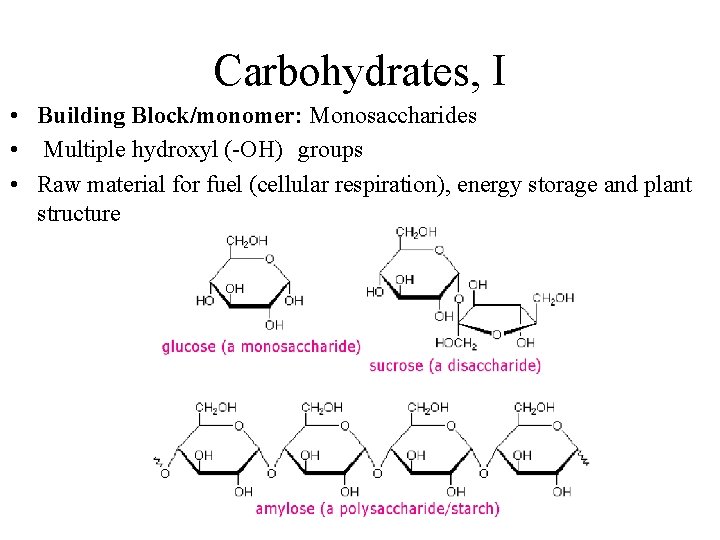
Carbohydrates, I • Building Block/monomer: Monosaccharides • Multiple hydroxyl (-OH) groups • Raw material for fuel (cellular respiration), energy storage and plant structure
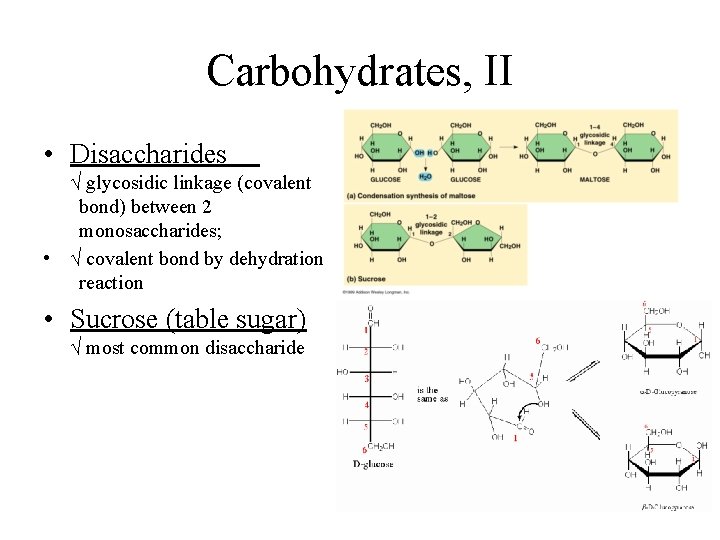
Carbohydrates, II • Disaccharides √ glycosidic linkage (covalent bond) between 2 monosaccharides; • √ covalent bond by dehydration reaction • Sucrose (table sugar) √ most common disaccharide
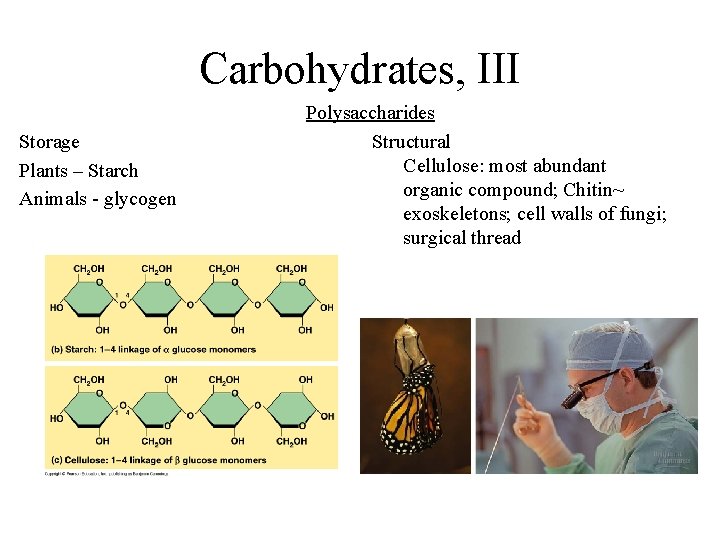
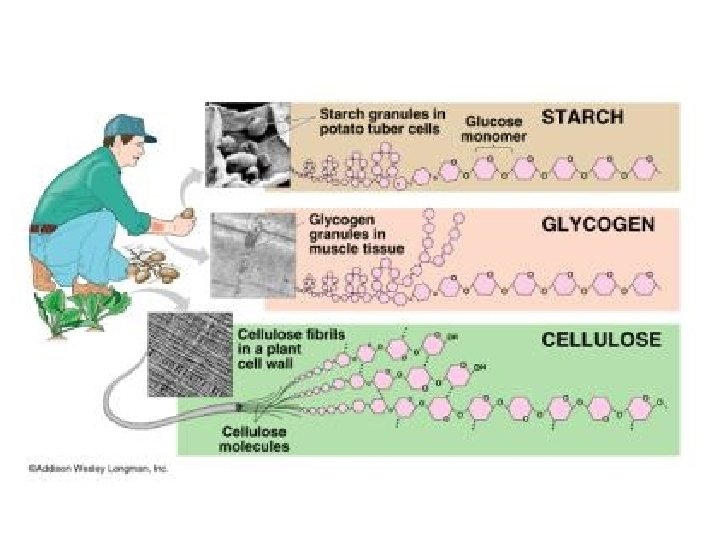
Carbohydrates, III Storage Plants – Starch Animals - glycogen Polysaccharides Structural Cellulose: most abundant organic compound; Chitin~ exoskeletons; cell walls of fungi; surgical thread

Lipids 1: Fats • Fats, phospholipids, steroids • Insulation & long-term energy storage • No polymers; glycerol and long chain fatty acid • Hydrophobic; H bonds in water exclude fats • Non-polar C-H bonds in fatty acid ‘tails’ • Saturated vs. unsaturated fats; single vs. double bonds
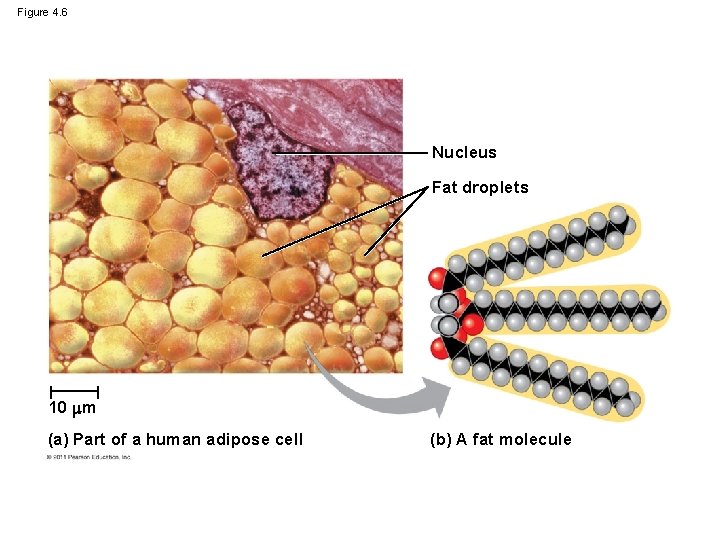
Figure 4. 6 Nucleus Fat droplets 10 m (a) Part of a human adipose cell (b) A fat molecule
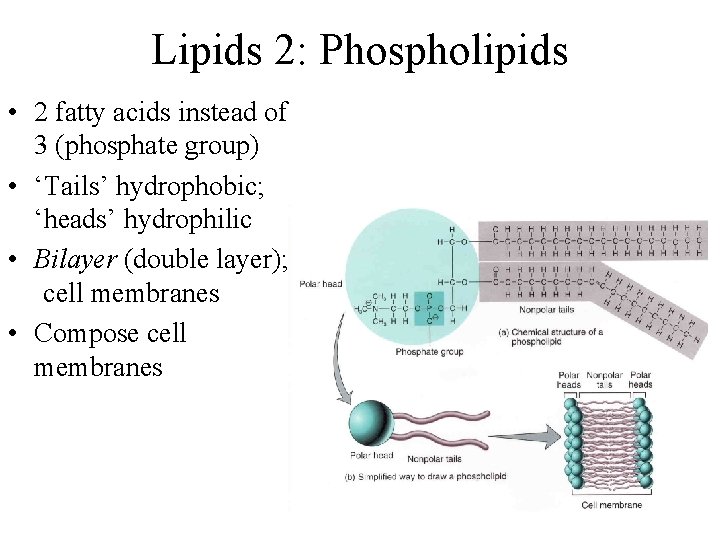
Lipids 2: Phospholipids • 2 fatty acids instead of 3 (phosphate group) • ‘Tails’ hydrophobic; ‘heads’ hydrophilic • Bilayer (double layer); cell membranes • Compose cell membranes
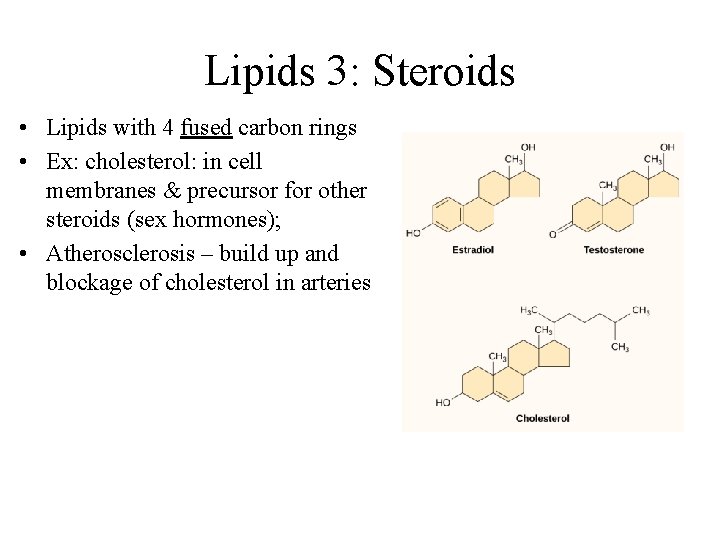
Lipids 3: Steroids • Lipids with 4 fused carbon rings • Ex: cholesterol: in cell membranes & precursor for other steroids (sex hormones); • Atherosclerosis – build up and blockage of cholesterol in arteries
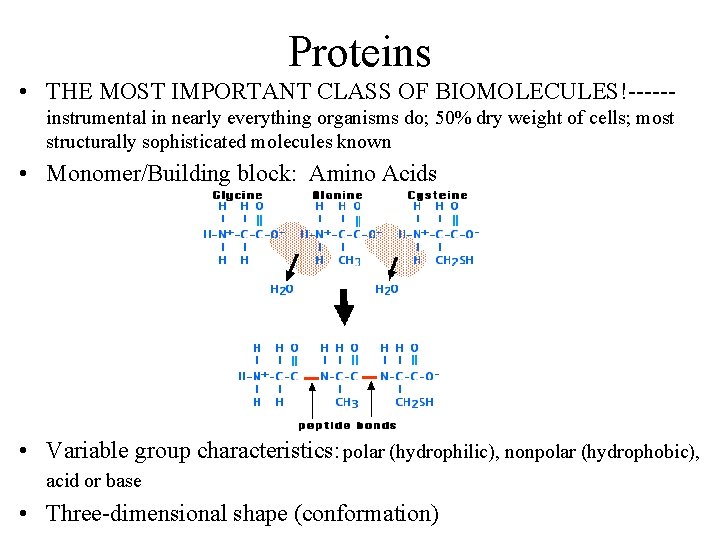
Proteins • THE MOST IMPORTANT CLASS OF BIOMOLECULES!-----instrumental in nearly everything organisms do; 50% dry weight of cells; most structurally sophisticated molecules known • Monomer/Building block: Amino Acids • Variable group characteristics: polar (hydrophilic), nonpolar (hydrophobic), acid or base • Three-dimensional shape (conformation)

Primary Structure • Conformation: Linear structure; sequence of amino acids • Molecular Biology: each type of protein has a unique primary structure of amino acids • Amino acid substitution: hemoglobin sickle-cell anemia
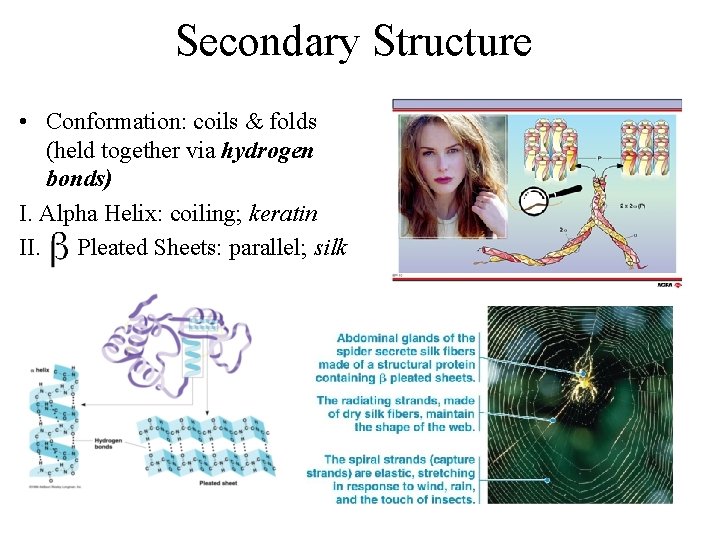
Secondary Structure • Conformation: coils & folds (held together via hydrogen bonds) I. Alpha Helix: coiling; keratin II. Pleated Sheets: parallel; silk
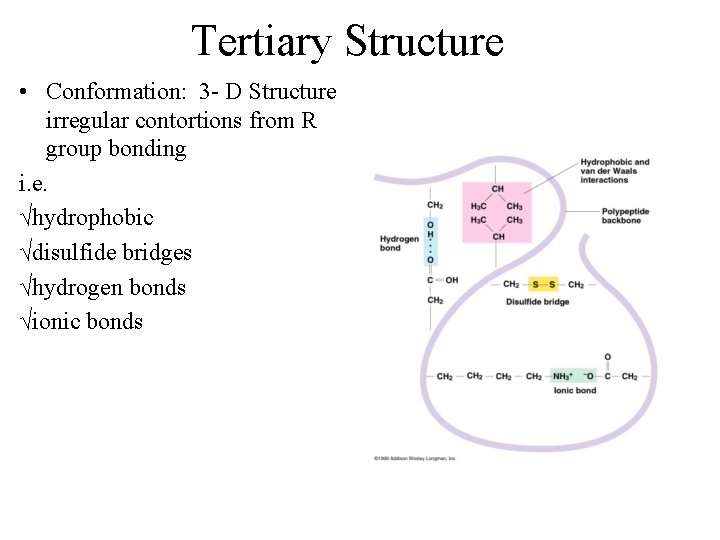
Tertiary Structure • Conformation: 3 - D Structure irregular contortions from R group bonding i. e. √hydrophobic √disulfide bridges √hydrogen bonds √ionic bonds
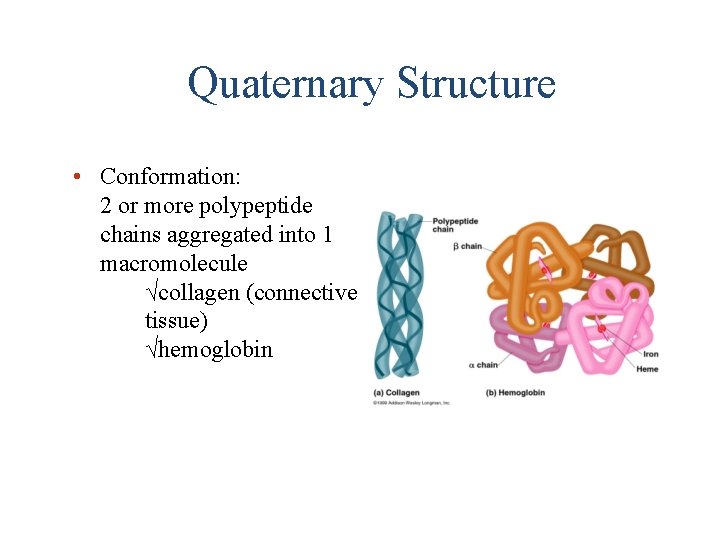
Quaternary Structure • Conformation: 2 or more polypeptide chains aggregated into 1 macromolecule √collagen (connective tissue) √hemoglobin
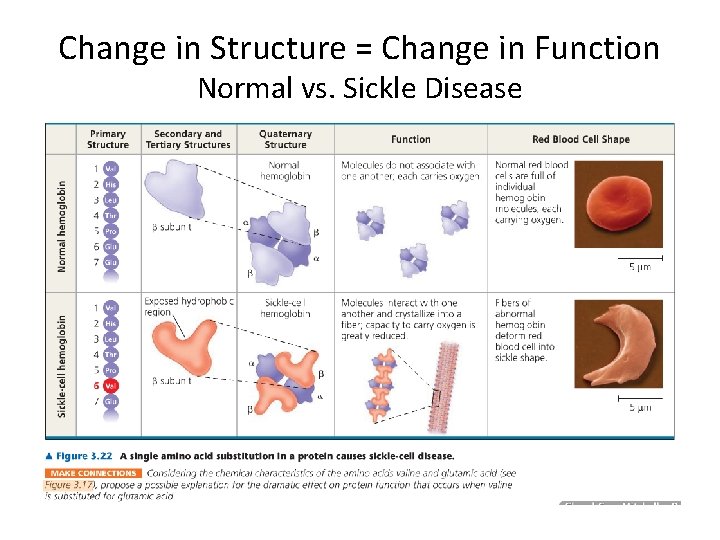
Change in Structure = Change in Function Normal vs. Sickle Disease
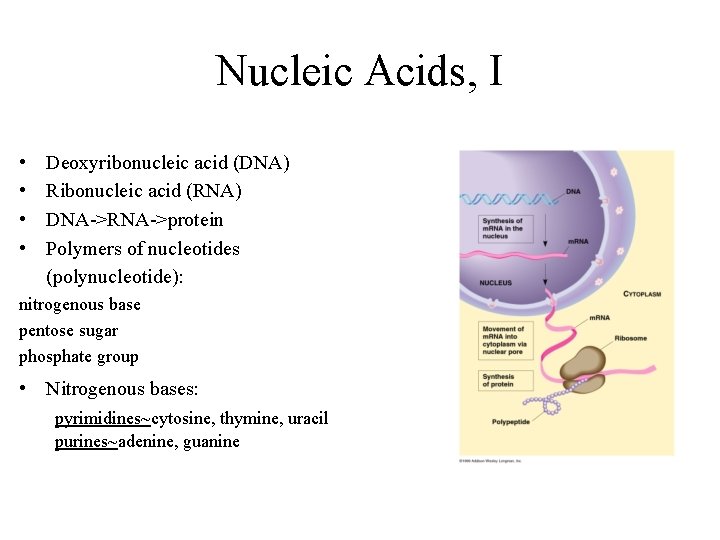
Nucleic Acids, I • • Deoxyribonucleic acid (DNA) Ribonucleic acid (RNA) DNA->RNA->protein Polymers of nucleotides (polynucleotide): nitrogenous base pentose sugar phosphate group • Nitrogenous bases: pyrimidines~cytosine, thymine, uracil purines~adenine, guanine
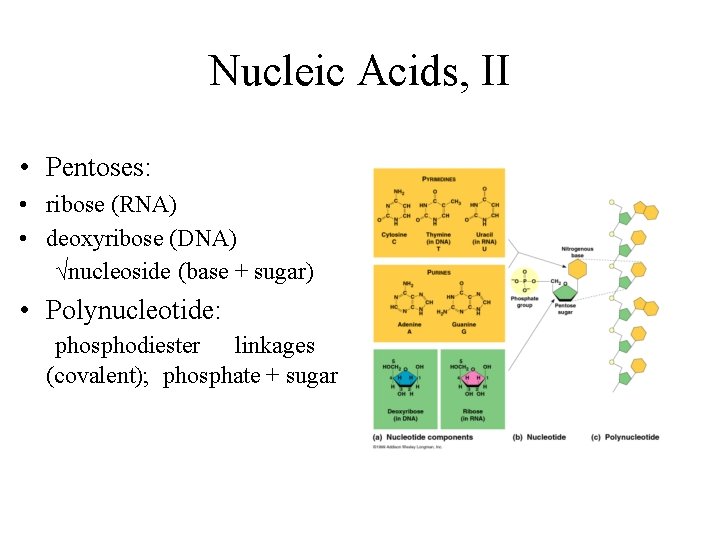
Nucleic Acids, II • Pentoses: • ribose (RNA) • deoxyribose (DNA) √nucleoside (base + sugar) • Polynucleotide: phosphodiester linkages (covalent); phosphate + sugar

Nucleic Acids, III • Inheritance based on DNA replication • Double helix (Watson & Crick 1953) H bonds~ between paired bases van der Waals~ between stacked bases • A to T; C to G pairing • Complementary
- Slides: 36