Carbon and the Molecular Diversity of Life Chapter

Carbon and the Molecular Diversity of Life Chapter 4 notes

Introduction • Although cells are 70 -95% water, the rest consists mostly of carbon-based compounds. • Proteins, DNA, carbohydrates, and other molecules that distinguish living matter from inorganic materials are all composed of carbon atoms bonded to each other and to atoms of other elements. – These other elements include hydrogen, oxygen, nitrogen, sulfur, and phosphorous.

What is organic chemistry? • The study of carbon (organic) compounds • These compounds can come from biological processes or they can be synthesized by non-living reactions. – Organic compounds can range from simple molecules, such as CO 2 or CH 4, to complex molecules like proteins, that may weigh over 100, 000 daltons.

Carbon atoms are versatile! • With a total of 6 electrons, a carbon atom has 2 in the first shell and 4 in the second shell. – Carbon usually does NOT form ionic bonds by losing or gaining 4 electrons. – Carbon usually completes its valence shell by sharing electrons with other atoms in four covalent bonds. – This tetravalence by carbon makes large, complex molecules possible.
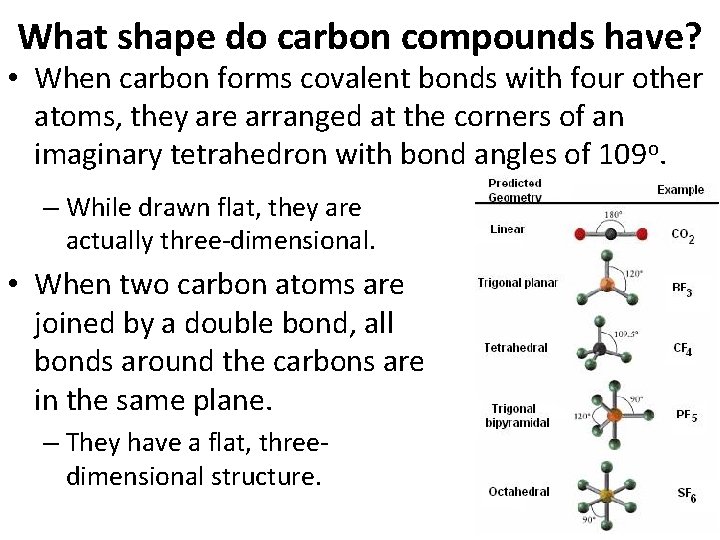
What shape do carbon compounds have? • When carbon forms covalent bonds with four other atoms, they are arranged at the corners of an imaginary tetrahedron with bond angles of 109 o. – While drawn flat, they are actually three-dimensional. • When two carbon atoms are joined by a double bond, all bonds around the carbons are in the same plane. – They have a flat, threedimensional structure.
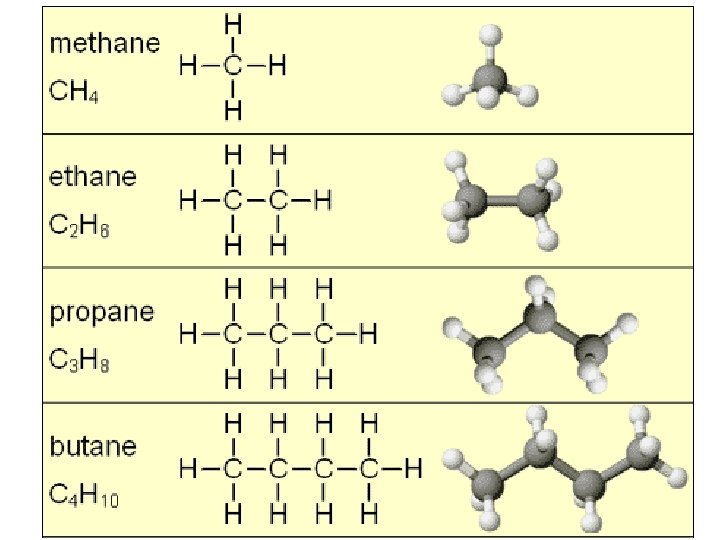
Carbon Models
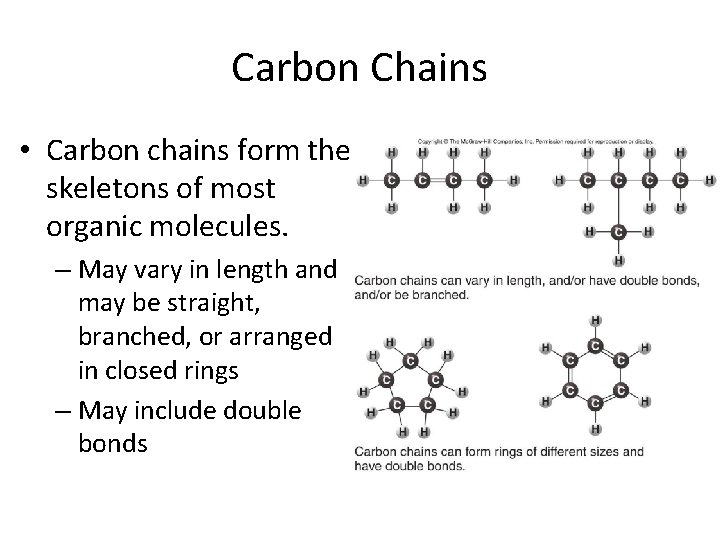
Carbon Chains • Carbon chains form the skeletons of most organic molecules. – May vary in length and may be straight, branched, or arranged in closed rings – May include double bonds

Hydrocarbons • Hydrocarbons are organic molecules that consist of only carbon and hydrogen atoms. – major component of petroleum • Fats are biological molecules that have long hydrocarbon tails attached to a nonhydrocarbon component.

Isomers • Isomers are compounds that have the same molecular formula but different structures and therefore different chemical properties. • Sort of like twins • 3 types…
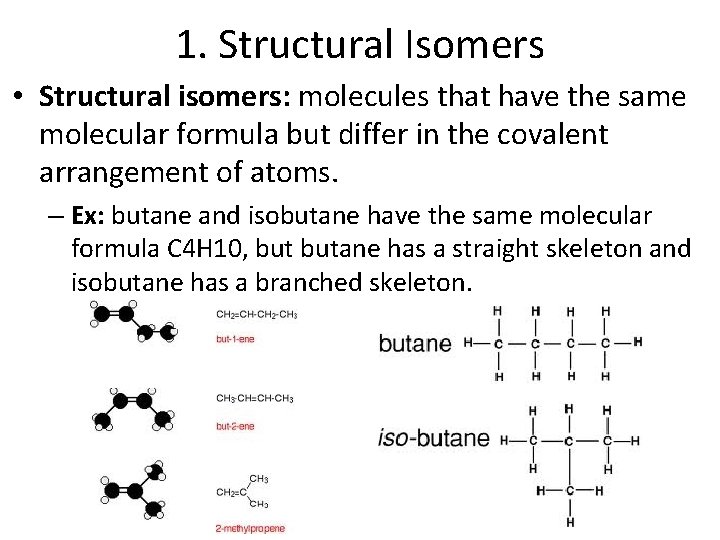
1. Structural Isomers • Structural isomers: molecules that have the same molecular formula but differ in the covalent arrangement of atoms. – Ex: butane and isobutane have the same molecular formula C 4 H 10, butane has a straight skeleton and isobutane has a branched skeleton.
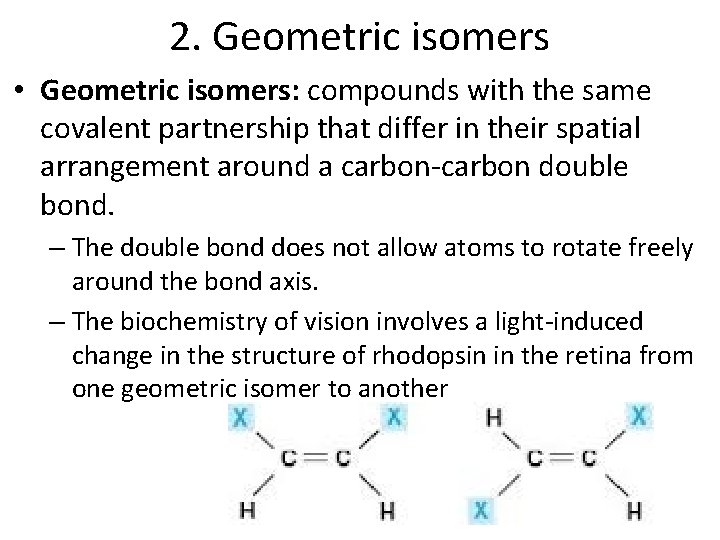
2. Geometric isomers • Geometric isomers: compounds with the same covalent partnership that differ in their spatial arrangement around a carbon-carbon double bond. – The double bond does not allow atoms to rotate freely around the bond axis. – The biochemistry of vision involves a light-induced change in the structure of rhodopsin in the retina from one geometric isomer to another

3. Enantiomers • Enantiomers: molecules that are mirror images of each other. – possible if there are four different atoms or groups of atoms bonded to a carbon. – Like left-handed and right-handed versions – Usually one is biologically active, the other inactive.

Enantiomer examples • Even the subtle structural differences in two enantiomers have important functional significance because of emergent properties from the specific arrangements of atoms. – One enantiomer of the drug thalidomide reduced morning sickness, its desired effect, but the other isomer caused severe birth defects. • The L-Dopa isomer is an effective treatment of Parkinson’s disease, but the D-Dopa isomer is inactive.

Model Carbon Molecules • With a partner, make a model of each of the following: 1. CH 4 (tetrahedron) 2. Structural Isomers: butane and isobutane (you only need to build the Carbon molecules) 3. Geometric Isomers: you design your own 4. Enantiomer Isomers: Use the CH 4 from #1 and change one H to something else. Then make the enantiomer of that molecule.
- Slides: 14