Carbon and oxygen combine to form carbon monoxide

Carbon and oxygen combine to form carbon monoxide (CO) and carbon dioxide (CO 2), but these two invisible gases are very different.
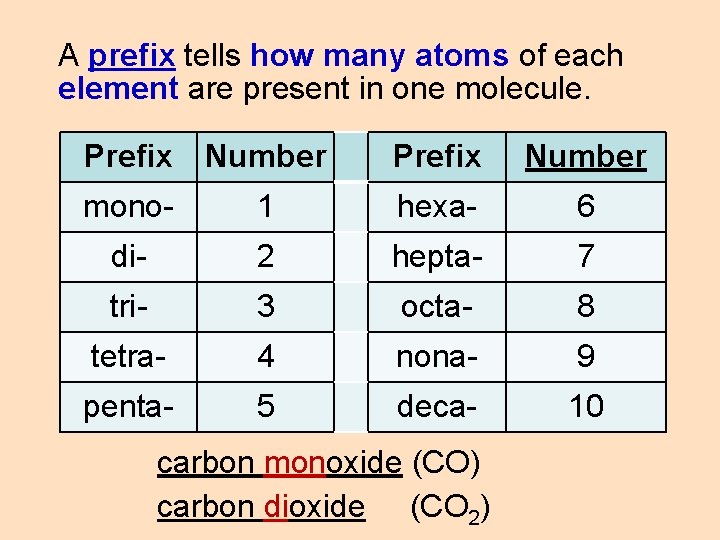
A prefix tells how many atoms of each element are present in one molecule. Prefix Number mono- 1 hexa- 6 di- 2 hepta- 7 tri- 3 octa- 8 tetra- 4 nona- 9 penta- 5 deca- 10 carbon monoxide (CO) carbon dioxide (CO 2)

Molecular Naming Rules: N 2 O dinitrogen monoxygen ide 1) Name elements in formula order. 2) Prefixes show the number of each atom. 3) Suffix of last element is –ide. 4) No prefix mono- for first element. CCl 4 carbon tetrachloride SO 3 sulfur trioxide H 2 O dihydrogen monoxide

Quick Quiz! 1. Which of the following compounds is named INCORRECTLY? A. CS 2 carbon disulfide B. BCl 3 boron trichloride C. IF 7 iodine heptafluoride D. PCl 5 phosphorus hexachloride

Quick Quiz. 2. Which of the following molecular compounds is named INCORRECTLY? A. PCl 3 phosphorus trichloride B. C 2 O 5 dicarbon pentoxide C. CF 4 carbon tetrafluoride D. H 3 P hydrogen monophosphide

Quick Quiz. 3. The correct formula for hexaphosphorus trisulfide is A. P 7 S 3 B. S 3 P 6 C. P 6 S 3 D. S 3 P 7
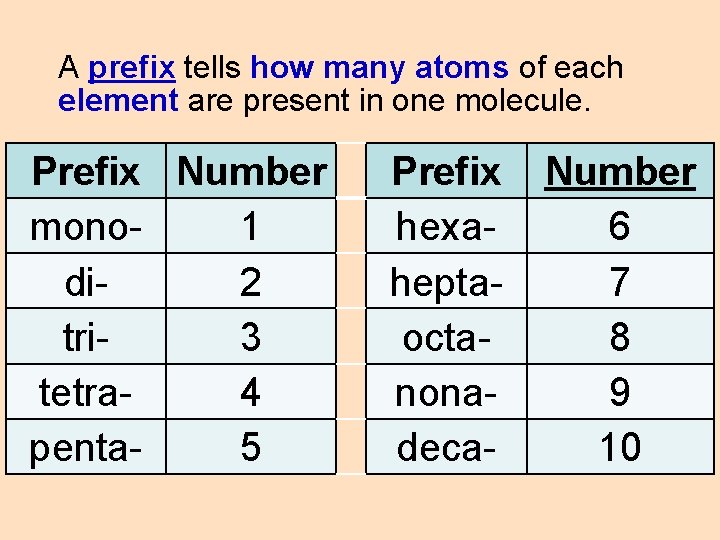
A prefix tells how many atoms of each element are present in one molecule. Prefix Number mono 1 di 2 tri 3 tetra 4 penta 5 Prefix hexaheptaoctanonadeca- Number 6 7 8 9 10
- Slides: 7