Carbon and organic molecules Carbon and its bonds

Carbon and organic molecules • Carbon and its bonds • Polymers and monomers - Carbohydrates - Proteins - Lipids - Nucleic acids Copyright © 2003 Pearson Education, Inc. publishing as Benjamin Cummings
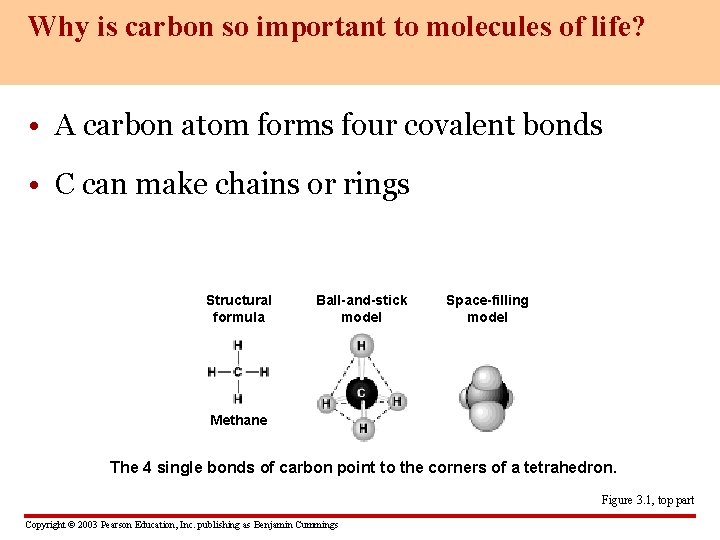
Why is carbon so important to molecules of life? • A carbon atom forms four covalent bonds • C can make chains or rings Structural formula Ball-and-stick model Space-filling model Methane The 4 single bonds of carbon point to the corners of a tetrahedron. Figure 3. 1, top part Copyright © 2003 Pearson Education, Inc. publishing as Benjamin Cummings
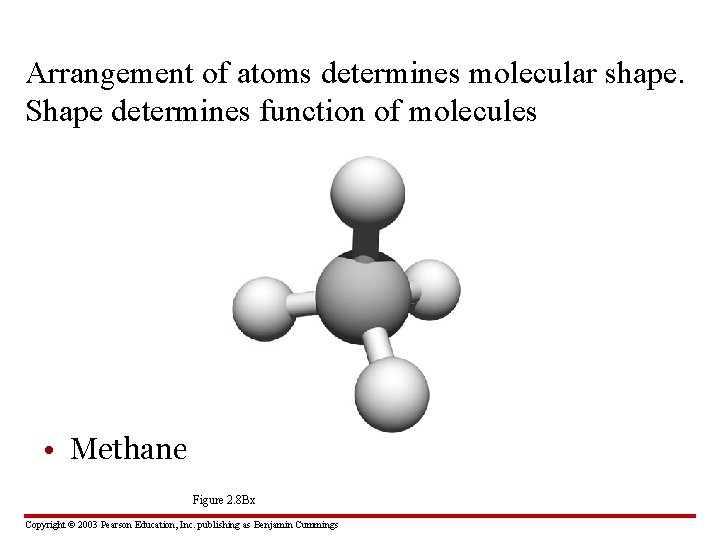
Arrangement of atoms determines molecular shape. Shape determines function of molecules • Methane, CH 4 Figure 2. 8 Bx Copyright © 2003 Pearson Education, Inc. publishing as Benjamin Cummings
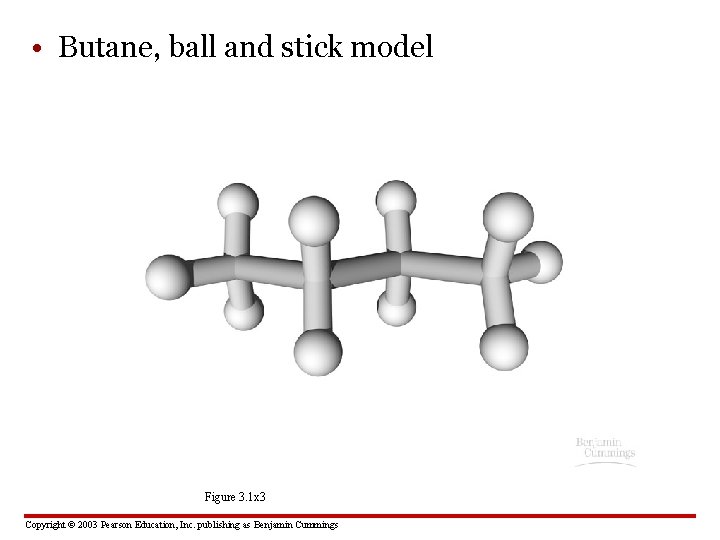
• Butane, ball and stick model Figure 3. 1 x 3 Copyright © 2003 Pearson Education, Inc. publishing as Benjamin Cummings
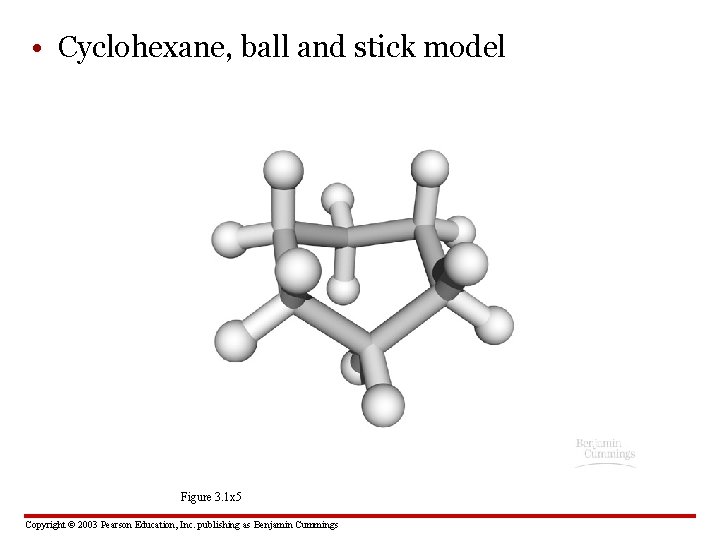
• Cyclohexane, ball and stick model Figure 3. 1 x 5 Copyright © 2003 Pearson Education, Inc. publishing as Benjamin Cummings

What are functional groups and what do they do? • groups of atoms that participate in chemical reactions • determine the chemical properties of molecules • Examples: acidity, solubility -OH -COOH Copyright © 2003 Pearson Education, Inc. publishing as Benjamin Cummings -NH 2 -CH 3

What affects solubility in water? • Molecules with +/- charge are usually hydrophilic or “water-loving” • Molecules with no charge and non-polar are usually hydrophobic and not soluble in water Copyright © 2003 Pearson Education, Inc. publishing as Benjamin Cummings

How do cells make so many different molecules that are needed for life? • Polymers are long chains of smaller molecular units called monomers • A huge number of different polymers can be made from a small number of monomers Copyright © 2003 Pearson Education, Inc. publishing as Benjamin Cummings

CARBOHYDRATES • Carbohydrates are a class of molecules – Monosaccharides: glucose, fructose, ribose – Disaccharides: maltose, sucrose, lactose – Polysaccharides: starch, glycogen Copyright © 2003 Pearson Education, Inc. publishing as Benjamin Cummings

Monosaccharides • are single-unit sugars • a multiple of CH 2 O • fuels for cellular work Copyright © 2003 Pearson Education, Inc. publishing as Benjamin Cummings
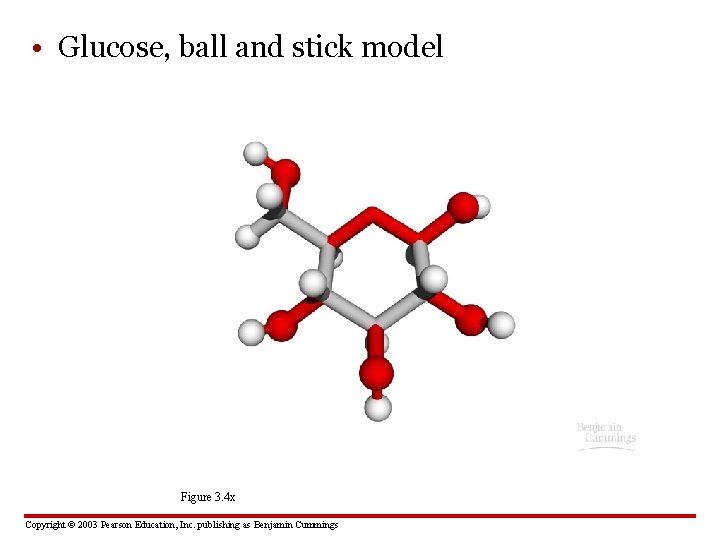
• Glucose, ball and stick model Figure 3. 4 x Copyright © 2003 Pearson Education, Inc. publishing as Benjamin Cummings
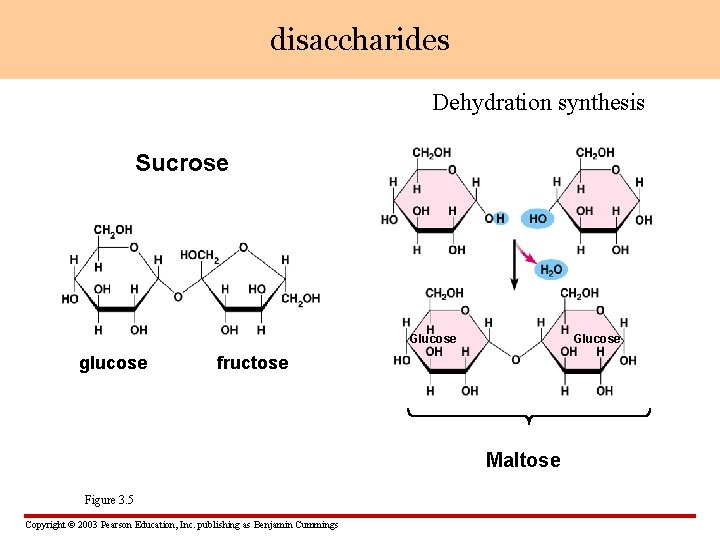
disaccharides Dehydration synthesis Sucrose Glucose glucose Glucose fructose Maltose Figure 3. 5 Copyright © 2003 Pearson Education, Inc. publishing as Benjamin Cummings

• Maltose, ball and stick model Figure 3. 5 x Copyright © 2003 Pearson Education, Inc. publishing as Benjamin Cummings
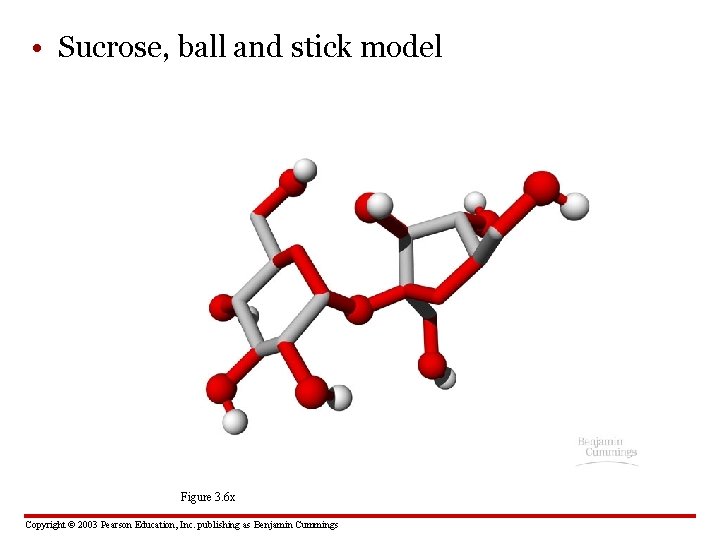
• Sucrose, ball and stick model Figure 3. 6 x Copyright © 2003 Pearson Education, Inc. publishing as Benjamin Cummings
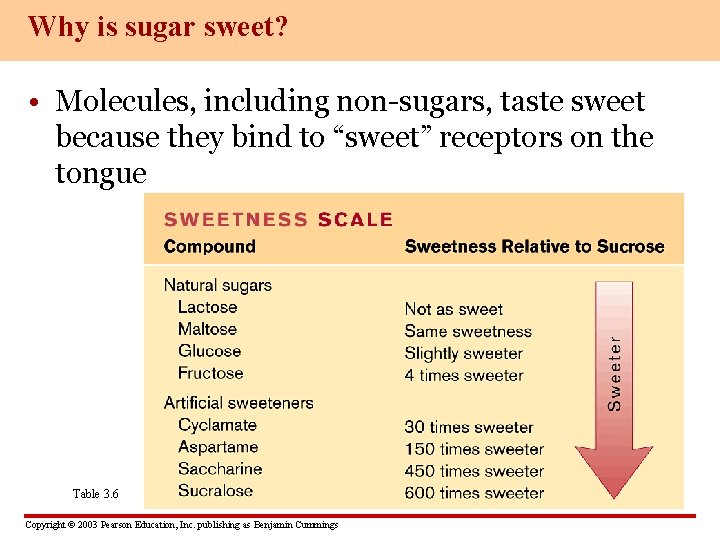
Why is sugar sweet? • Molecules, including non-sugars, taste sweet because they bind to “sweet” receptors on the tongue Table 3. 6 Copyright © 2003 Pearson Education, Inc. publishing as Benjamin Cummings

Polysaccharides are long chains of sugar units • Size: thousands of linked monosaccharides • purpose: energy storage, structural Copyright © 2003 Pearson Education, Inc. publishing as Benjamin Cummings
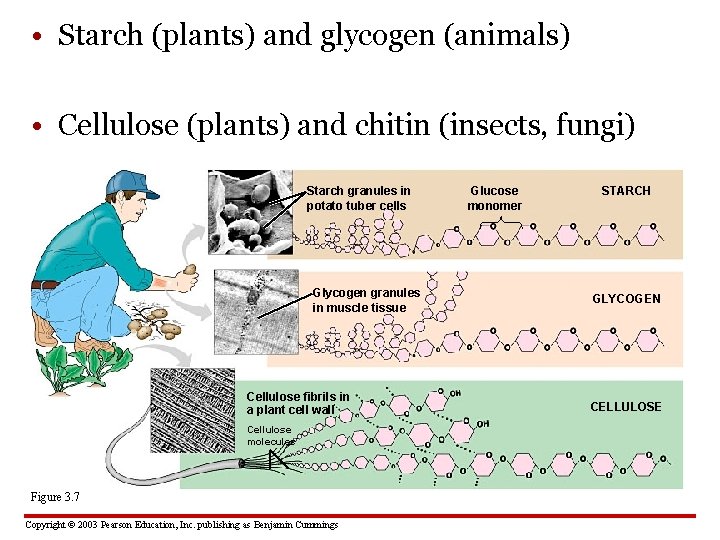
• Starch (plants) and glycogen (animals) • Cellulose (plants) and chitin (insects, fungi) Starch granules in potato tuber cells Glycogen granules in muscle tissue Cellulose fibrils in a plant cell wall Cellulose molecules Figure 3. 7 Copyright © 2003 Pearson Education, Inc. publishing as Benjamin Cummings Glucose monomer STARCH GLYCOGEN CELLULOSE
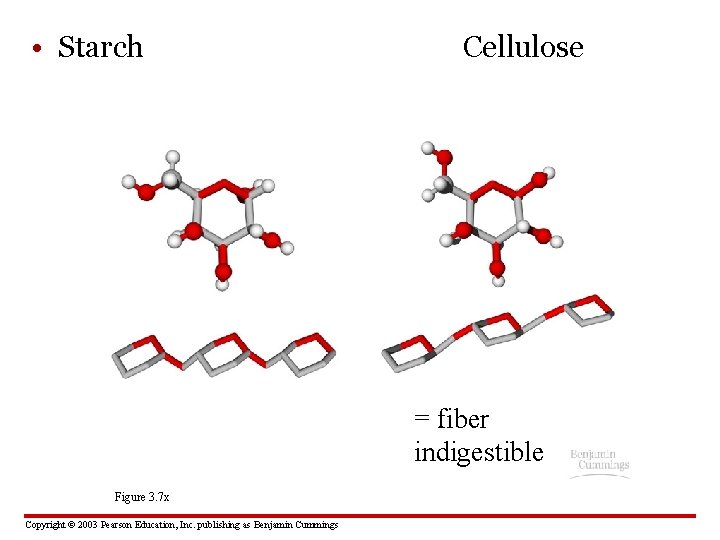
• Starch Cellulose = fiber indigestible Figure 3. 7 x Copyright © 2003 Pearson Education, Inc. publishing as Benjamin Cummings

Lipids include fats, oils, and steroids. • hydrophobic • composed largely of carbon and hydrogen • Purposes: - energy-storage - insulation, cushioning - membranes - signals Copyright © 2003 Pearson Education, Inc. publishing as Benjamin Cummings
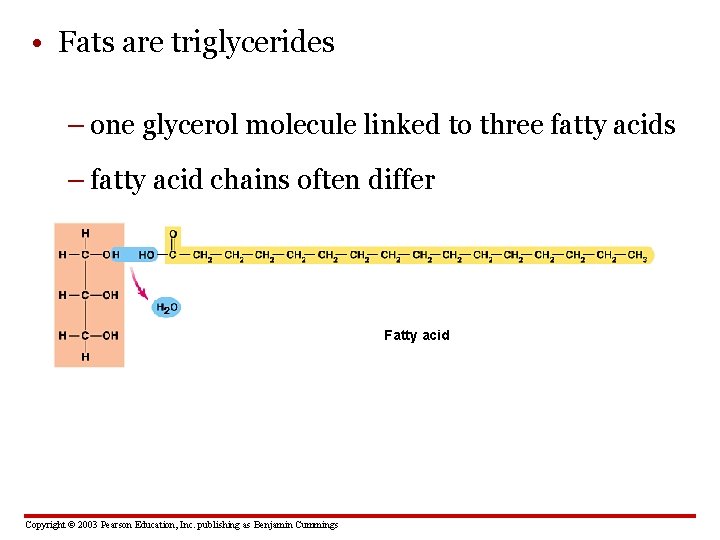
• Fats are triglycerides – one glycerol molecule linked to three fatty acids – fatty acid chains often differ Fatty acid Copyright © 2003 Pearson Education, Inc. publishing as Benjamin Cummings
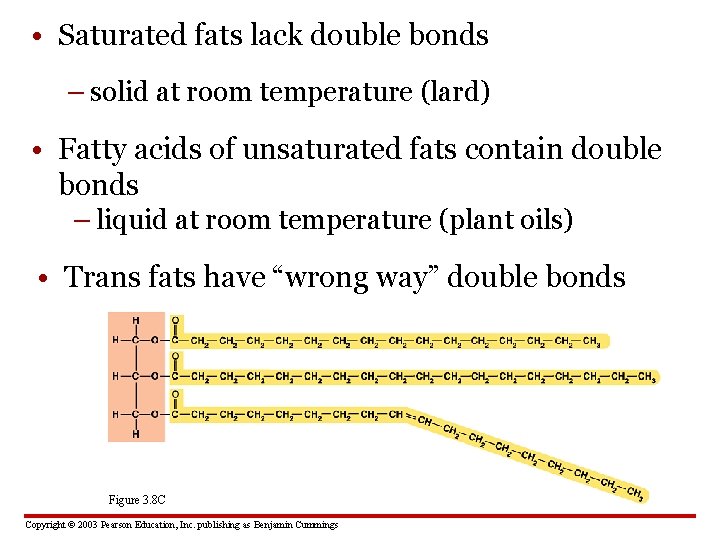
• Saturated fats lack double bonds – solid at room temperature (lard) • Fatty acids of unsaturated fats contain double bonds – liquid at room temperature (plant oils) • Trans fats have “wrong way” double bonds Figure 3. 8 C Copyright © 2003 Pearson Education, Inc. publishing as Benjamin Cummings

Phospholipids • both polar and nonpolar portions • major component of cell membranes Figure 3. 9 Copyright © 2003 Pearson Education, Inc. publishing as Benjamin Cummings

Waxes form waterproof coatings and can prevent organisms from drying out or getting wet Copyright © 2003 Pearson Education, Inc. publishing as Benjamin Cummings

Steroids are often hormones testosterone estrogen Anabolic steroids Hormone Replacement Therapy Copyright © 2003 Pearson Education, Inc. publishing as Benjamin Cummings
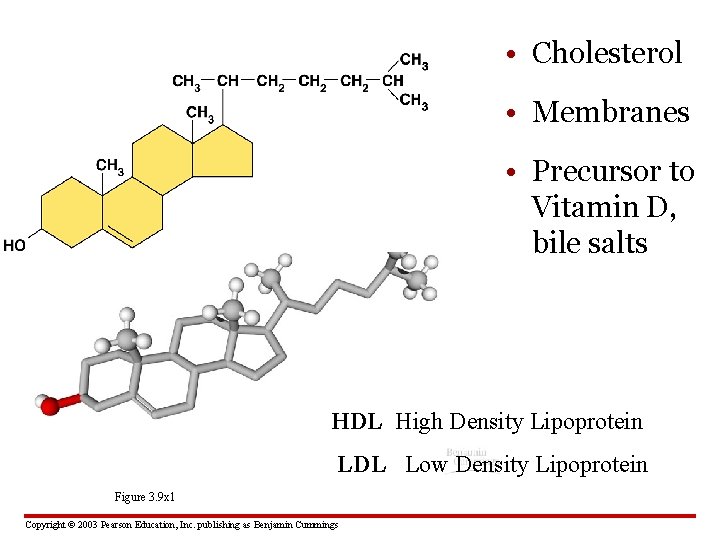
• Cholesterol • Membranes • Precursor to Vitamin D, bile salts HDL High Density Lipoprotein LDL Low Density Lipoprotein Figure 3. 9 x 1 Copyright © 2003 Pearson Education, Inc. publishing as Benjamin Cummings

Proteins are essential to the structures and activities of life • Proteins are involved in – cellular structure – movement – nutrition – defense – transport – communication • Enzymes regulate chemical reactions Copyright © 2003 Pearson Education, Inc. publishing as Benjamin Cummings Figure 3. 11

Proteins are the most structurally and functionally diverse of life’s molecules composed of 20 kinds of amino acids 9 a. a. must be consumed in food – Their diversity is based on different arrangements of amino acids Copyright © 2003 Pearson Education, Inc. publishing as Benjamin Cummings
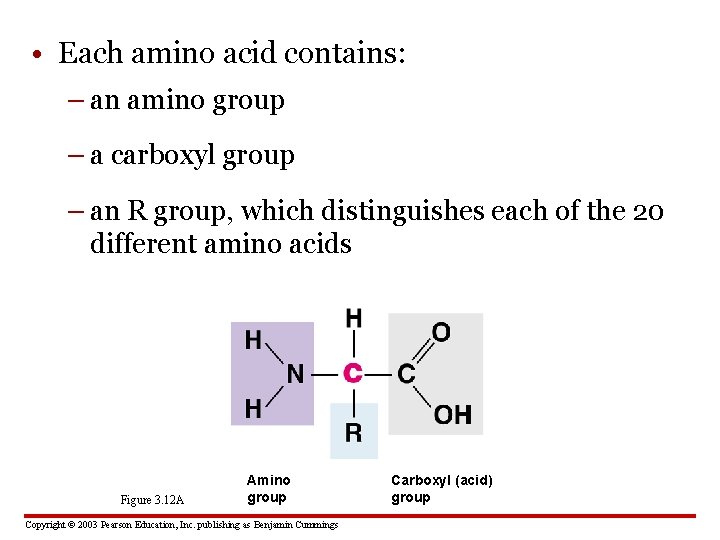
• Each amino acid contains: – an amino group – a carboxyl group – an R group, which distinguishes each of the 20 different amino acids Figure 3. 12 A Amino group Copyright © 2003 Pearson Education, Inc. publishing as Benjamin Cummings Carboxyl (acid) group
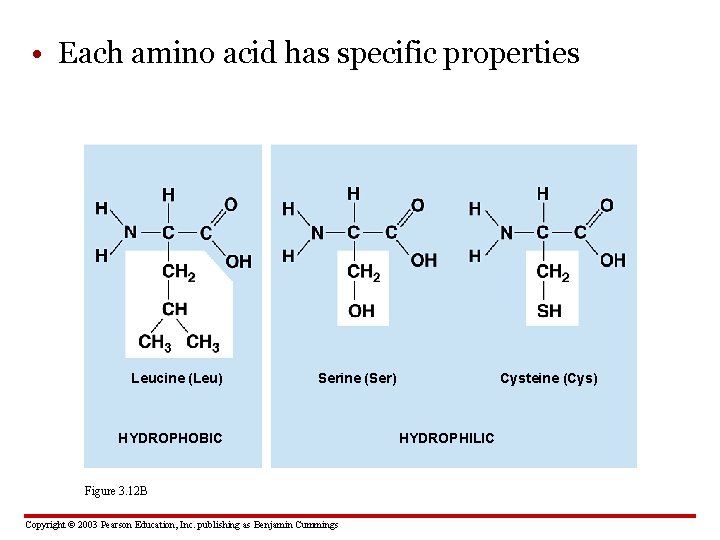
• Each amino acid has specific properties Leucine (Leu) Serine (Ser) HYDROPHOBIC Figure 3. 12 B Copyright © 2003 Pearson Education, Inc. publishing as Benjamin Cummings Cysteine (Cys) HYDROPHILIC
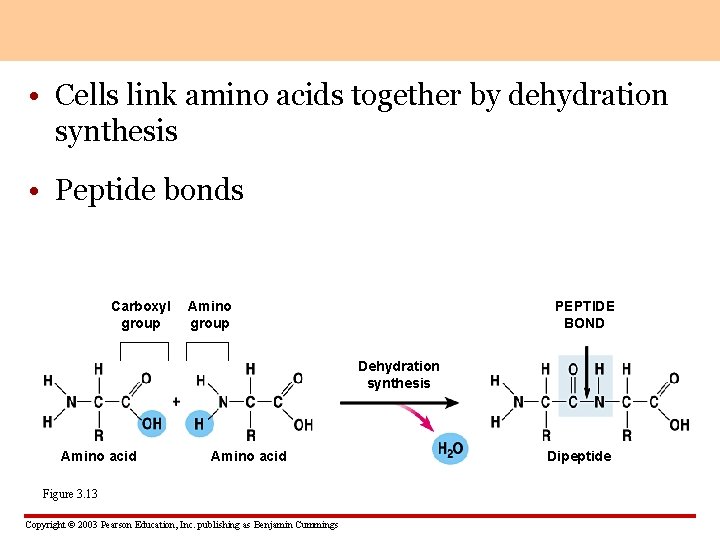
• Cells link amino acids together by dehydration synthesis • Peptide bonds Carboxyl group Amino group PEPTIDE BOND Dehydration synthesis Amino acid Figure 3. 13 Copyright © 2003 Pearson Education, Inc. publishing as Benjamin Cummings Dipeptide
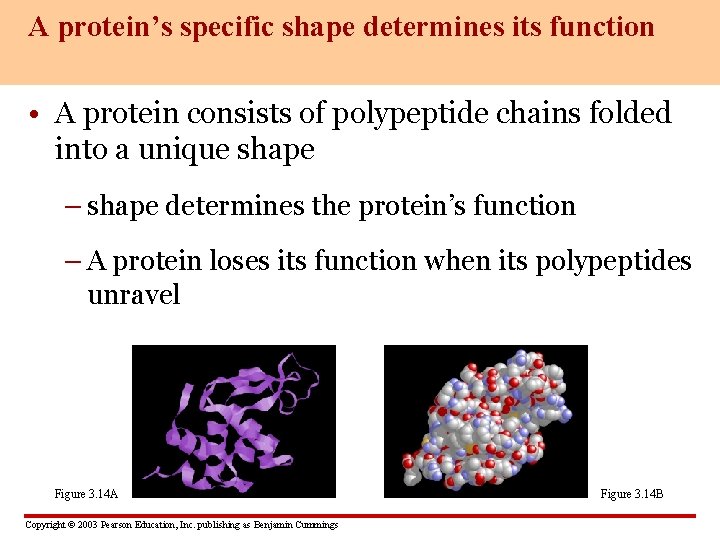
A protein’s specific shape determines its function • A protein consists of polypeptide chains folded into a unique shape – shape determines the protein’s function – A protein loses its function when its polypeptides unravel Figure 3. 14 A Copyright © 2003 Pearson Education, Inc. publishing as Benjamin Cummings Figure 3. 14 B
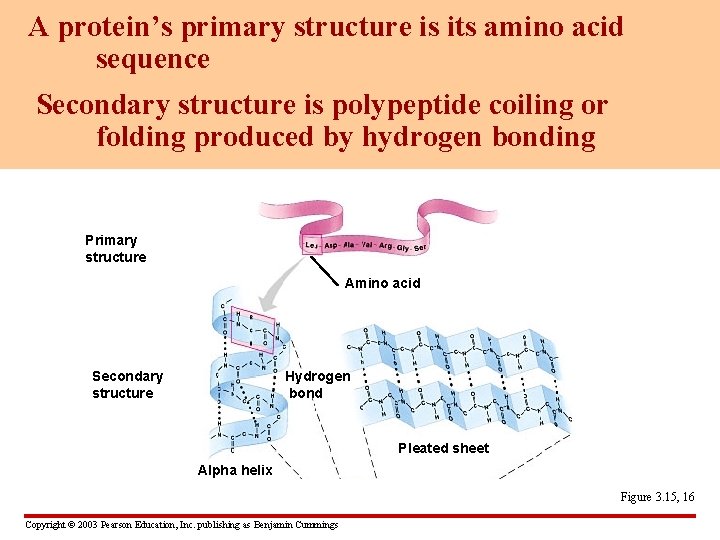
A protein’s primary structure is its amino acid sequence Secondary structure is polypeptide coiling or folding produced by hydrogen bonding Primary structure Amino acid Secondary structure Hydrogen bond Pleated sheet Alpha helix Figure 3. 15, 16 Copyright © 2003 Pearson Education, Inc. publishing as Benjamin Cummings
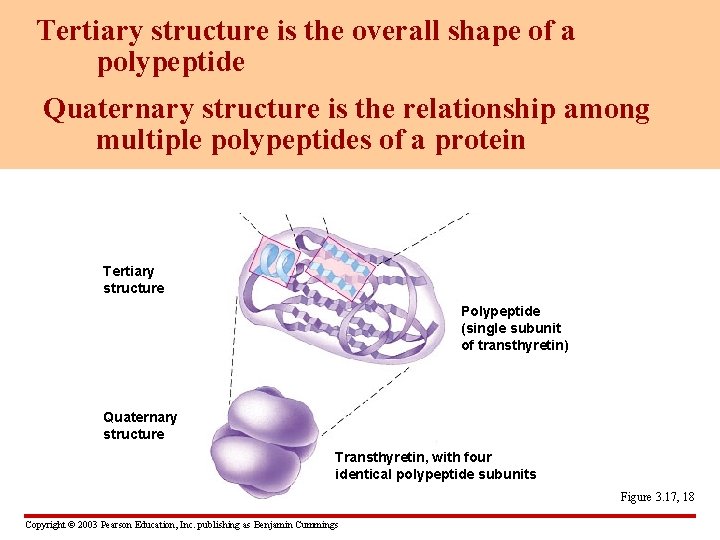
Tertiary structure is the overall shape of a polypeptide Quaternary structure is the relationship among multiple polypeptides of a protein Tertiary structure Polypeptide (single subunit of transthyretin) Quaternary structure Transthyretin, with four identical polypeptide subunits Figure 3. 17, 18 Copyright © 2003 Pearson Education, Inc. publishing as Benjamin Cummings

Linus Pauling contributed to our understanding of protein structure and function • 2 Nobel prizes - Chemistry and Peace Figure 3. 19 Copyright © 2003 Pearson Education, Inc. publishing as Benjamin Cummings

Nucleic acids are information-rich polymers of nucleotides • Nucleic acids such as DNA and RNA serve as the blueprints for proteins • They ultimately control the life of a cell • DNA sequence is inherited by progeny Copyright © 2003 Pearson Education, Inc. publishing as Benjamin Cummings
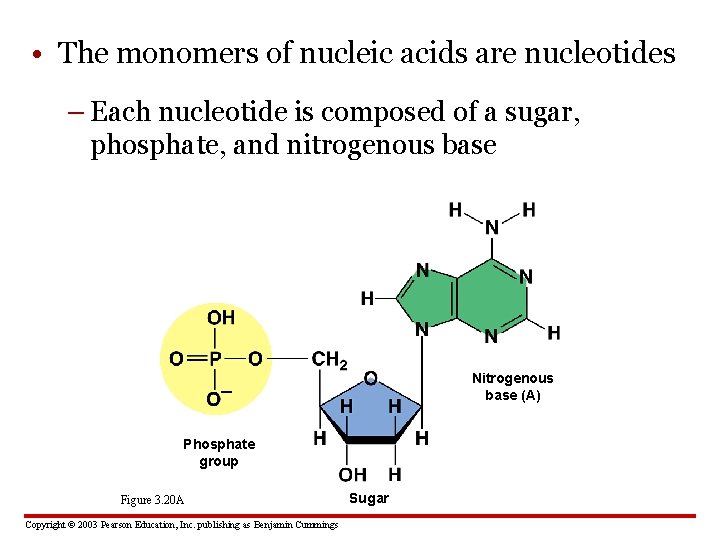
• The monomers of nucleic acids are nucleotides – Each nucleotide is composed of a sugar, phosphate, and nitrogenous base Nitrogenous base (A) Phosphate group Figure 3. 20 A Copyright © 2003 Pearson Education, Inc. publishing as Benjamin Cummings Sugar
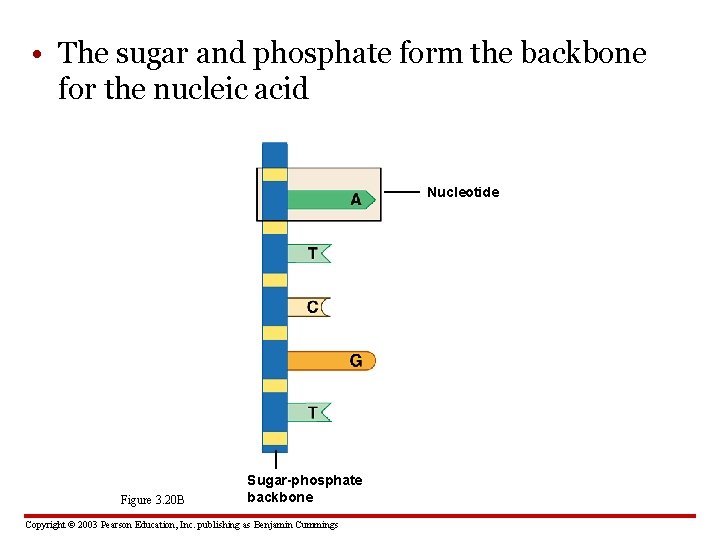
• The sugar and phosphate form the backbone for the nucleic acid Nucleotide Figure 3. 20 B Sugar-phosphate backbone Copyright © 2003 Pearson Education, Inc. publishing as Benjamin Cummings
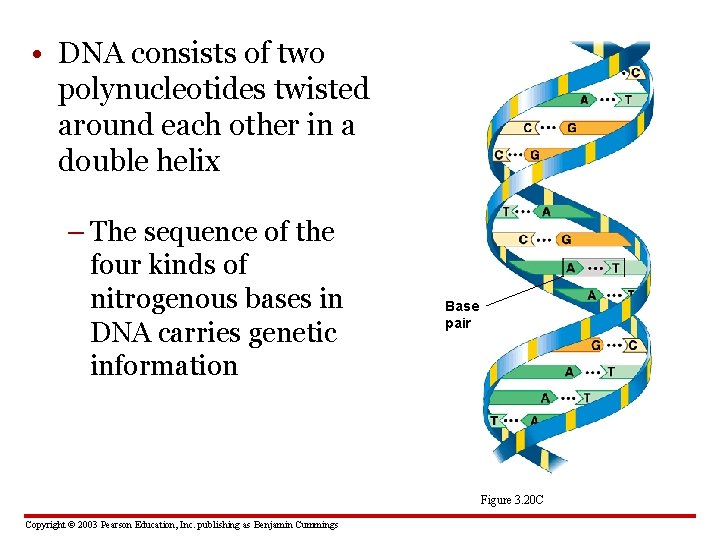
• DNA consists of two polynucleotides twisted around each other in a double helix – The sequence of the four kinds of nitrogenous bases in DNA carries genetic information Base pair Nitrogenous base (A) Figure 3. 20 C Copyright © 2003 Pearson Education, Inc. publishing as Benjamin Cummings
- Slides: 38