Carbohydrates What are carbohydrates n n Carbohydrates include

Carbohydrates

What are carbohydrates? n n Carbohydrates include sugars, starches, cellulose and glycogen. Carbohydrates are needed for storage and liberation of energy. All carbohydrates contain the following elements: carbon, hydrogen, oxygen Carbohydrates may be used a sweeteners, thickeners, stabilizers, gelling agents, and fat replacers

Carbohydrates can be divided into three groups n n n Monosaccharide-”single sugars” eg glucose. Disaccharides –”double sugars” eg sucrose. Polysaccharides-”multiple sugars” eg starch n n Monosaccharide and disaccharides are sugars that are sweet and soluble. Polysaccharides are polymers that are not sweet or soluble.
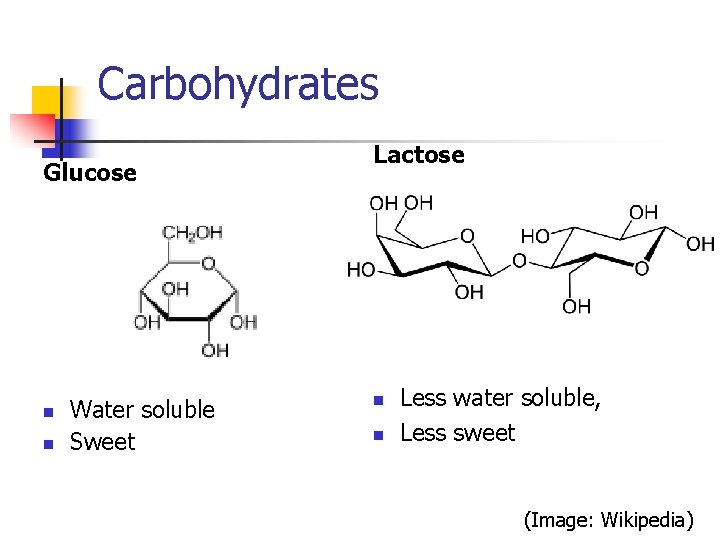
Carbohydrates Glucose n n Water soluble Sweet Lactose n n Less water soluble, Less sweet (Image: Wikipedia)
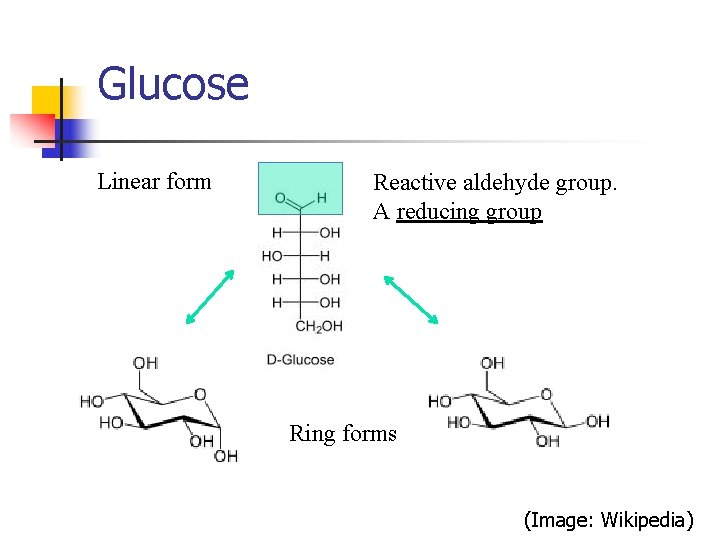
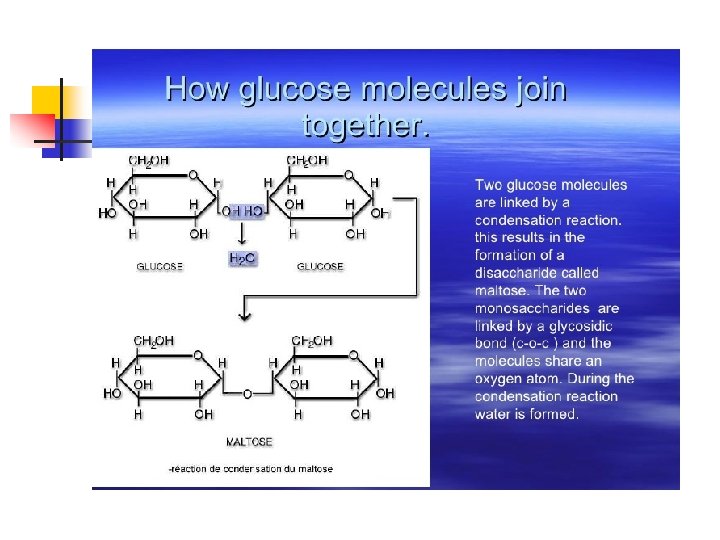
Glucose Linear form Reactive aldehyde group. A reducing group Ring forms (Image: Wikipedia)
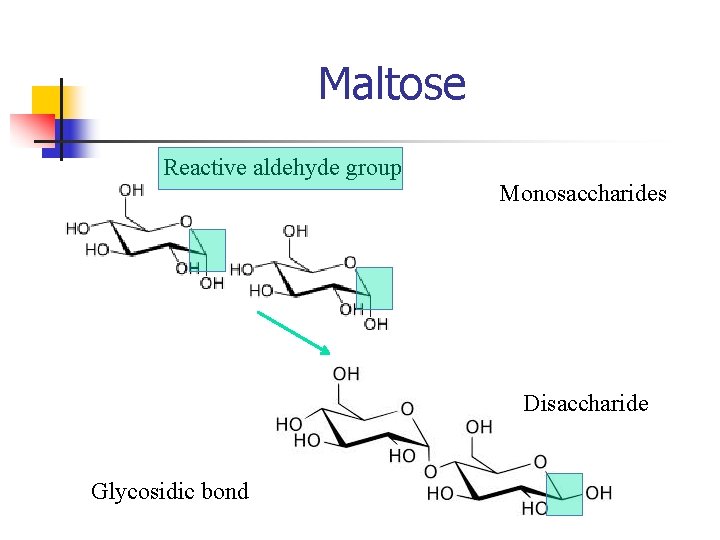
Maltose Reactive aldehyde group Monosaccharides Disaccharide Glycosidic bond
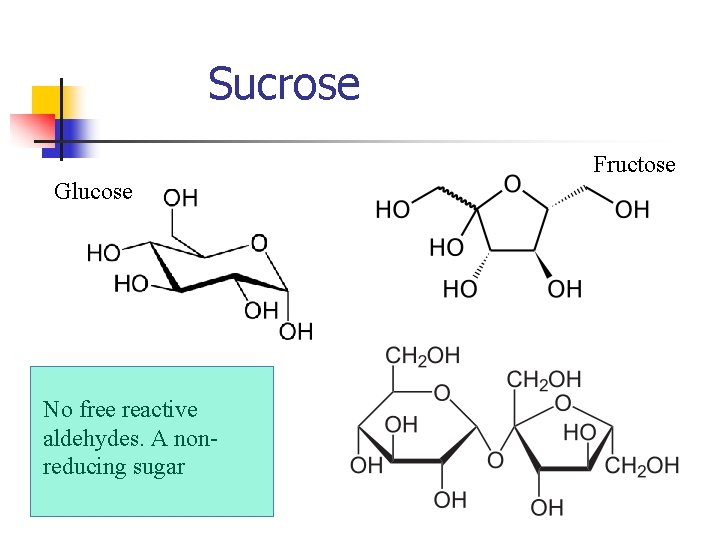
Sucrose Fructose Glucose No free reactive aldehydes. A nonreducing sugar

Polysaccharides n n Polysaccharides are formed from thousands of glucose molecules and are classified as polymers. There are three polysaccharides that need to be studied: n n n Starch Glycogen Cellulose
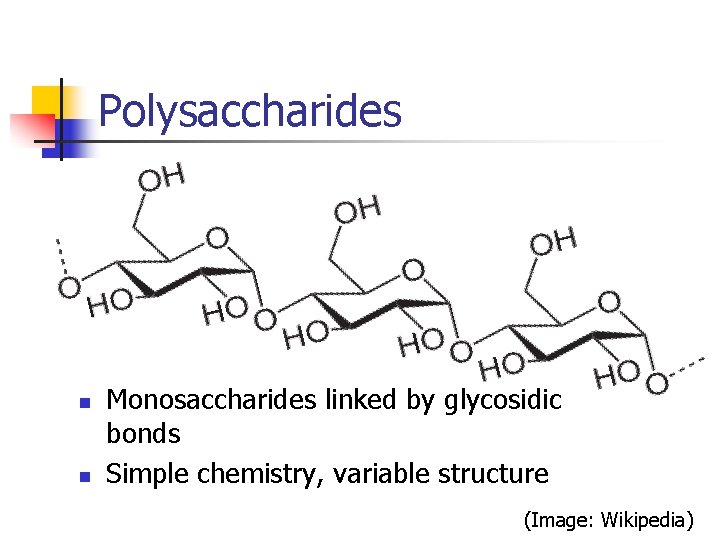
Polysaccharides n n Monosaccharides linked by glycosidic bonds Simple chemistry, variable structure (Image: Wikipedia)

Polysaccharide sources n n n n Higher plant sources: Starch, cellulose, pectin Physically modified: starch, cellulose Chemically modified: starch, cellulose Seaweeds: alginate, carageenan Seeds: guar, locust bean Tree extrudates: Gum arabic Microbial fermentations: xanthan gum
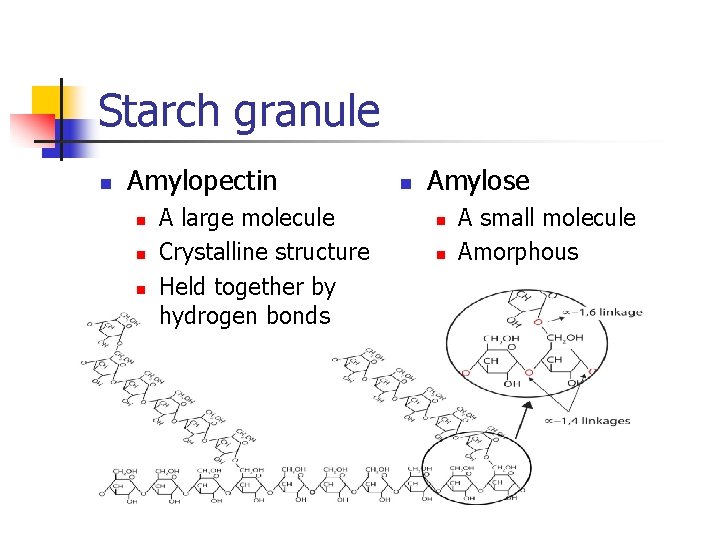
Starch granule n Amylopectin n A large molecule Crystalline structure Held together by hydrogen bonds n Amylose n n A small molecule Amorphous
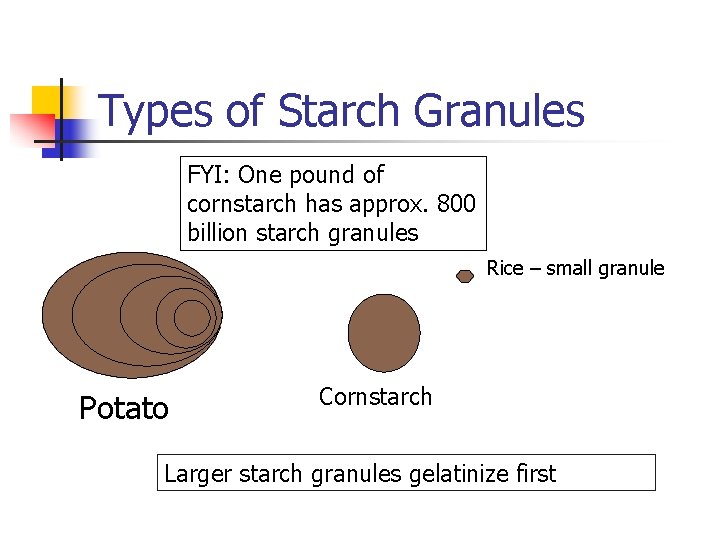
Types of Starch Granules FYI: One pound of cornstarch has approx. 800 billion starch granules Rice – small granule Potato Cornstarch Larger starch granules gelatinize first

Gelatinization n n Starch granules in water Heat – overcome the hydrogen bonds Swelling of starch granules https: //www. youtube. com/w atch? v=L 6 v. Yx. YE 1 j. Og
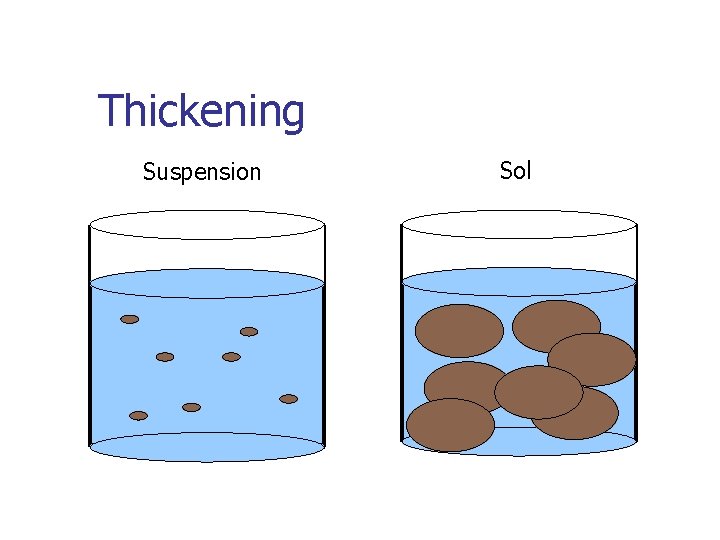
Thickening Suspension Sol

Thickening ability n Thickening ability of various starches n What starch has the greatest thickening ability? n Which on has the least? n At what temperature does thickening occur?

Q: What causes lumping? n Dry starch n n Exterior granules gelatinized first and clings together Interior granules remains dry

Ways to avoid lumping n Separation of starch granules n n n Agitation - stirring Mix with a fat - roux Mix with a sugar
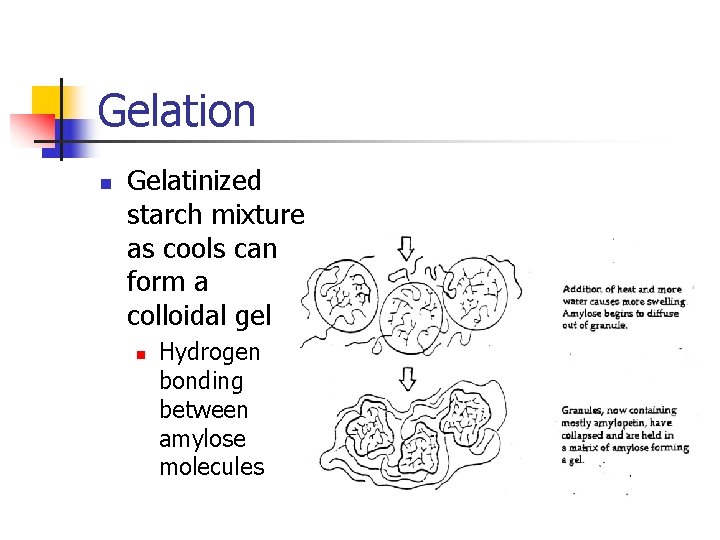
Gelation n Gelatinized starch mixture as cools can form a colloidal gel n Hydrogen bonding between amylose molecules

Gelled Dispersions Suspension Sol Gelled solid
Gel structure

Gels n n n May be clear and form at low concentrations Often stretch a long way before breaking Examples: gelatin, starch, carrageenan, pectin Image flickr user Furryscaly

Retrogradation and Syneresis n Retrogradation n The crystallization and matrix forming of starch molecules n Amylose retrogrades quickly n Starches with low levels of amylose do not form gels well As gel ages there is a continued drawing together of the gel and water is extruded. n n Syneresis n Separation of liquid from the gel

To Gel, or Not to Gel n n Starches High in Amylose content are able to gel Starches without amylose thicken, and do not gel

Waxy starch n Contains only amylopectin n Useful for making pies in which a thickened but ungelled consistency is desirable Waxy starches are derived from some natural strains of barley, corn, rice and sorghum Root starches are naturally low in amylose (cassava, arrowroot)

Modified Starches n n n Pregelatinized starch is gelatinized then dried (instant pudding) CWS may be gelling or non-gelling (low-fat items for mouthfeel) Cross-linked starches withstand low p. H, high shear, or high temperatures (bbq sauce, pizza sauce) Stabilized starches are used in frozen foods to prevent gelling and syneresis Acid-modified starch forms a strong gel upon cooling and less viscous when hot.

Pectin n n Pectinic acids are dispersible in water and can form gels It is not easy to form pectin gels: it requires a delicate balance of pectin, water, sugar, and acid Sugar competes for water, thus making less water available for the pectin molecules Acid makes the pectin molecules less likely to repel each other

Gums (hydrocolloids) n n n Complex hydrophilic carbohydrates containing thousands of monosaccharide units Do not form gels, but trap large amounts of water Thickening agent (replacing starch) Stabilizer of emulsions and maintain texture Mouthfeel of low-fat foods

Gums (hydrocolloids) n n Gums can be used synergistically to form gels n Guar and carrageenan n Locust bean and xanthan Seaweed polysaccharides n Can form gels under certain conditions (iota carrageenan with calcium) n Can also be used with other gums n Agar forms a heat-reversible gel n Alginates are used to produce specialized food products (synthetic fruits)
- Slides: 29