CARBOHYDRATES Types of Carbohydrates Monosacchrides Disaccharides Contain 2

CARBOHYDRATES

Types of Carbohydrates • Monosacchrides • Disaccharides Contain 2 monosacchride units • Polysacchrides Contain many monosacchride units 2

MONOSACCHARIDES Empirical formula: CH 2 O Monosaccharides contain: a) C=O group and b) at least two OH- groups C 5 H 10 O 5 pentoses (e. g. ridose) C 6 H 10 O 6 hexoses (e. g. glucose) They have between 3 and 6 carbon atoms. They have many structural isomers. Several carbon atoms are chiral and give rise to optical isomerism. Chain and ring structures are possible.

Monosaccharides • Three Carbons = Triose • Four Carbons = Tetrose • Five Carbons = Pentose • Six Carbons = Hexose 4

Monosaccharides • Aldoses are monosacchrides with an aldehyde group and many hydroxyl (-OH) groups. • Ketoses are monosacchrides with a ketone group and many hydroxyl (-OH) groups. 5
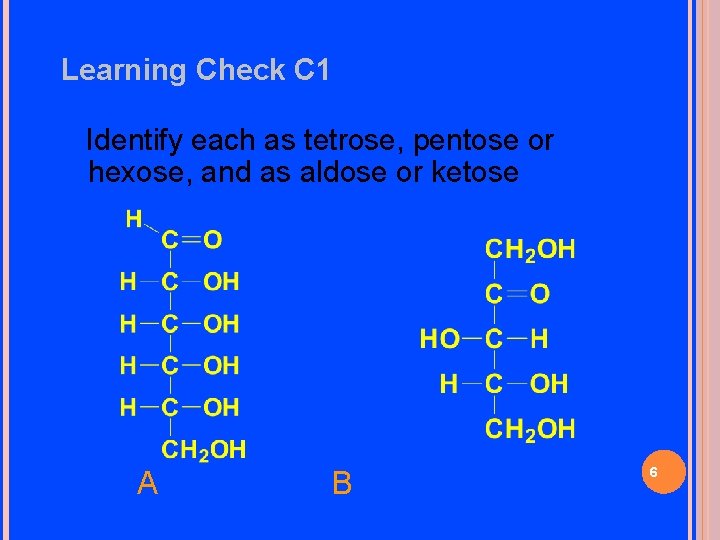
Learning Check C 1 Identify each as tetrose, pentose or hexose, and as aldose or ketose A B 6
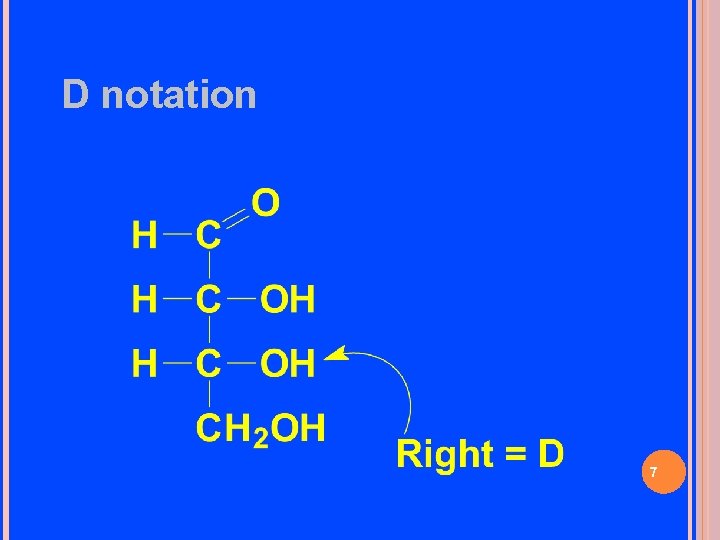
D notation 7

D-GLUCOSE The form of glucose that is found in nature is known as D-glucose. To form the cyclic structure, connect the first and the fifth carbon (condensation) Figure 1: Straight chain formula of Dglucose.
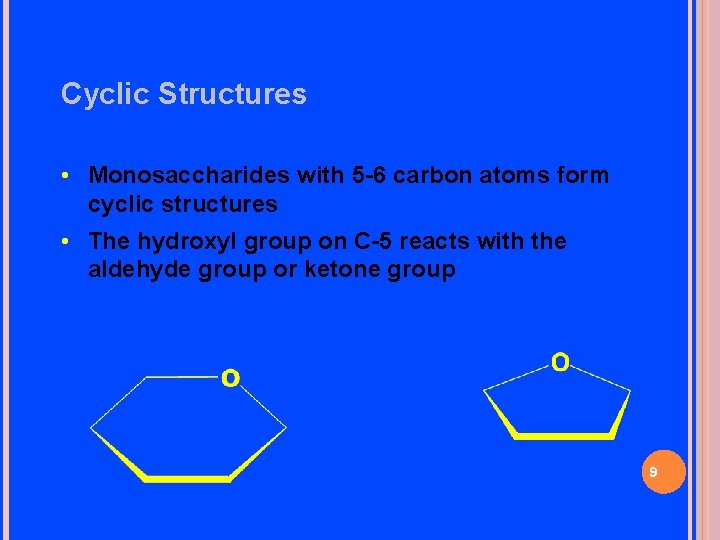
Cyclic Structures • Monosaccharides with 5 -6 carbon atoms form cyclic structures • The hydroxyl group on C-5 reacts with the aldehyde group or ketone group 9
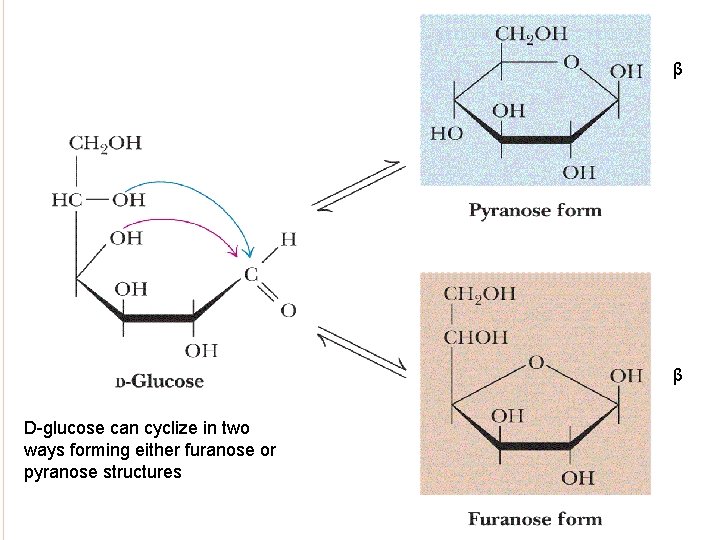
β β D-glucose can cyclize in two ways forming either furanose or pyranose structures
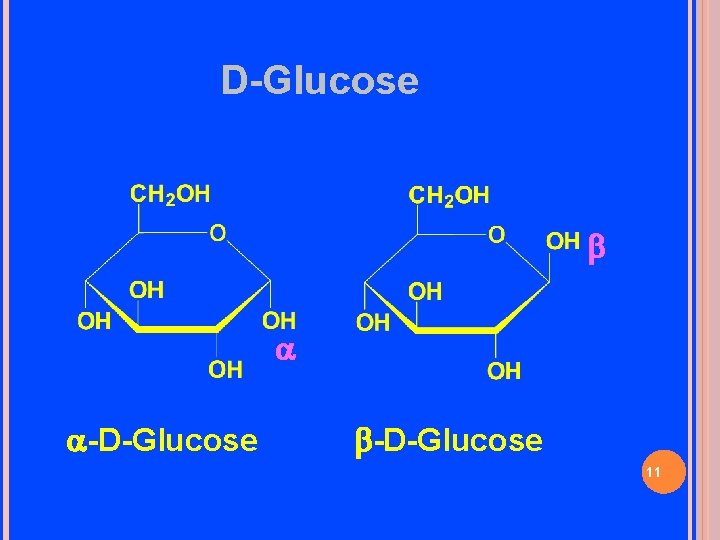
D-Glucose -D-Glucose 11
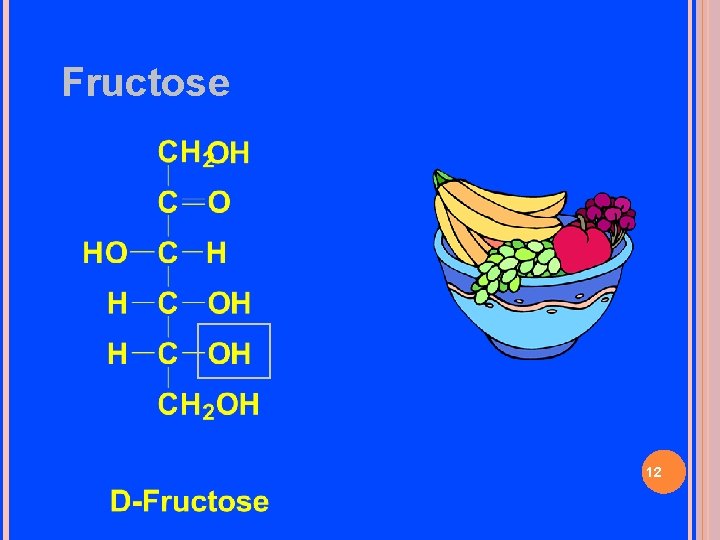
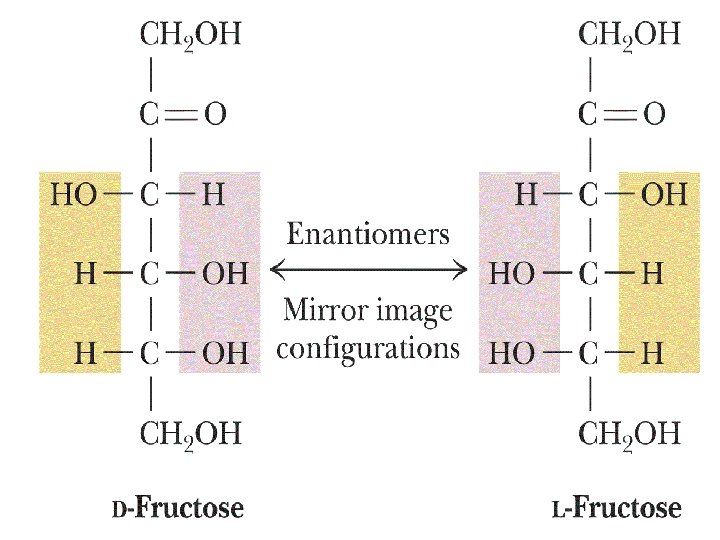
Fructose 12
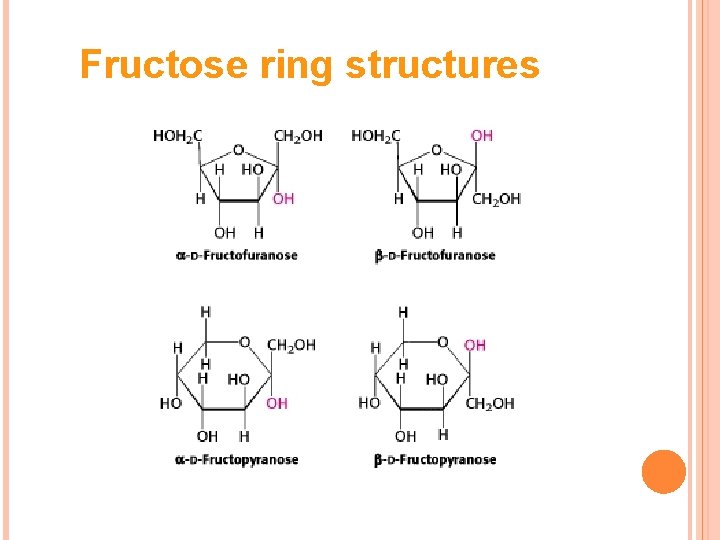
Fructose ring structures

POLYSACCHARIDES o Monosaccharides can undergo condensation reaction to form disaccharides and polysaccharides. Monosaccharides Condensation Disaccharides Condensation Polysaccharides
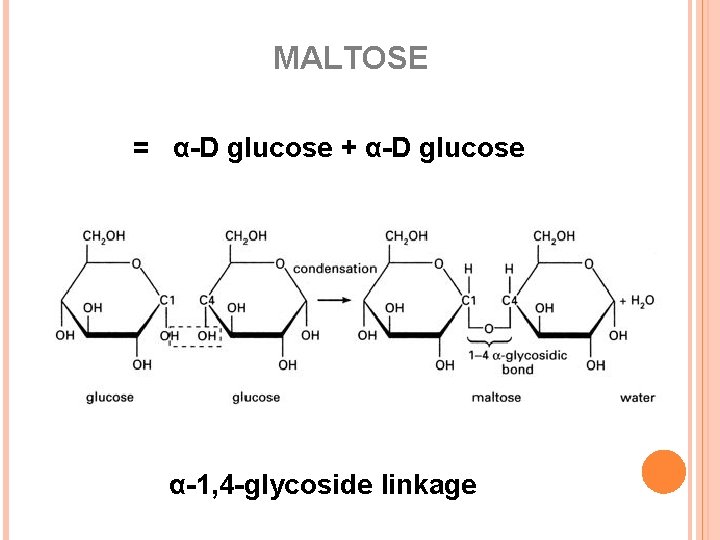
MALTOSE = α-D glucose + α-D glucose α-1, 4 -glycoside linkage

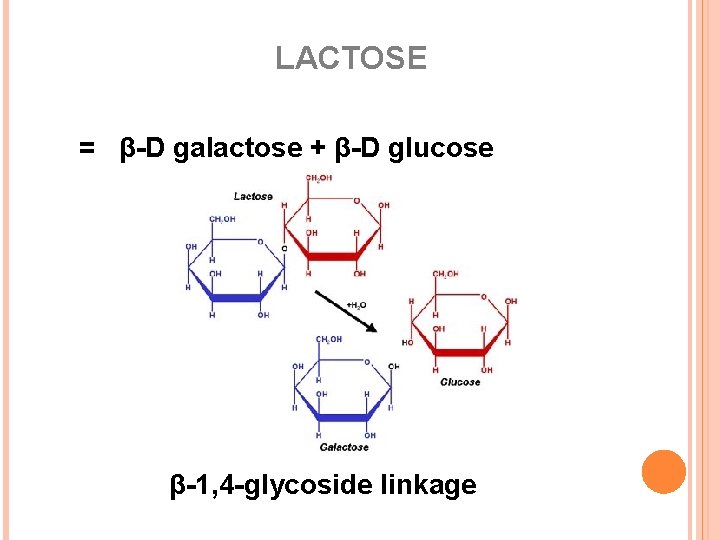
LACTOSE = β-D galactose + β-D glucose β-1, 4 -glycoside linkage

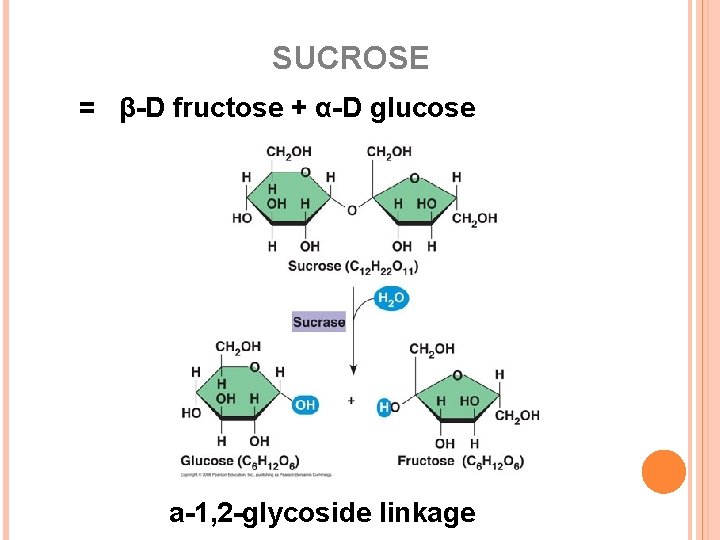
SUCROSE = β-D fructose + α-D glucose a-1, 2 -glycoside linkage


POLYSACCHARIDES o Monosaccharides can undergo condensation reaction to form disaccharides and polysaccharides. Monosaccharides Condensation Disaccharides Condensation Polysaccharides

POLYSACCHARIDES Starch Amylose Straight chain Amylopectin Branched Water soluble α-1, 4 α-glucose Glycogen α-glucose More branched Cellulose β-glucose Straight chain α-1, 4+α-1, 6 Water insoluble β-1, 4+H-bonds Water insoluble Hydrolyzed by cellulose > found only in plant eating mammals > cannot be digested by humans
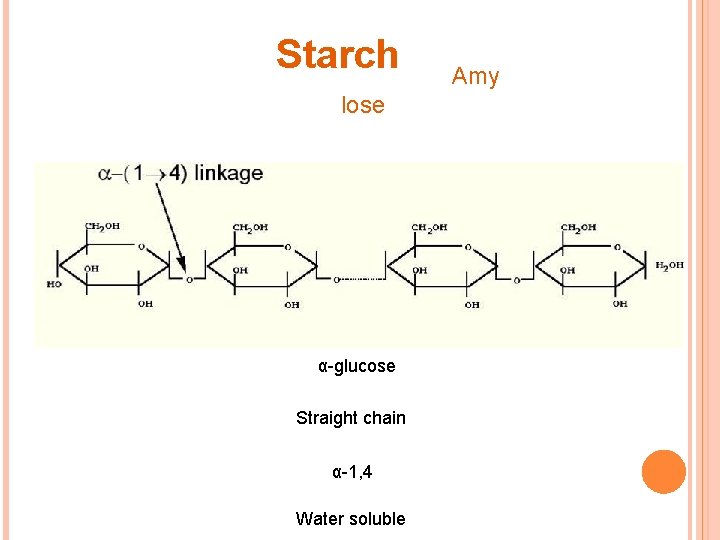
Starch lose α-glucose Straight chain α-1, 4 Water soluble Amy
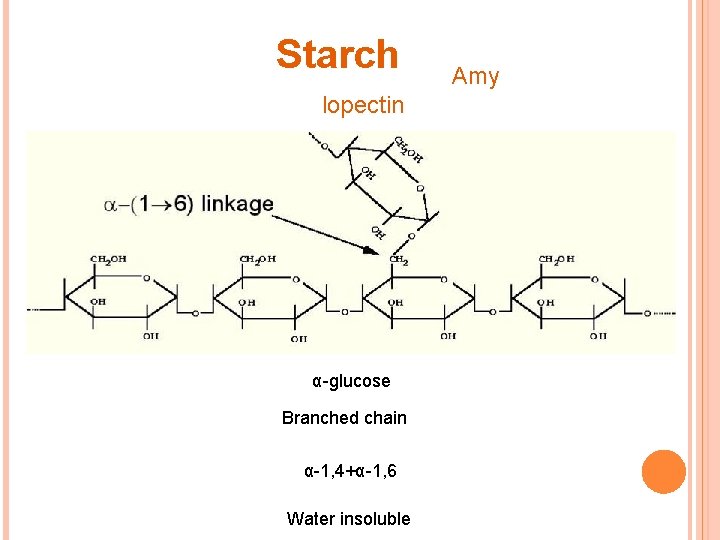
Starch lopectin α-glucose Branched chain α-1, 4+α-1, 6 Water insoluble Amy
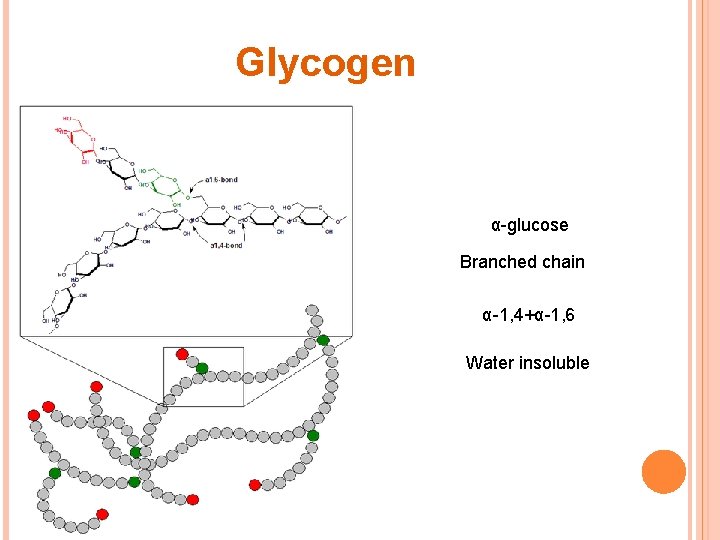
Glycogen α-glucose Branched chain α-1, 4+α-1, 6 Water insoluble
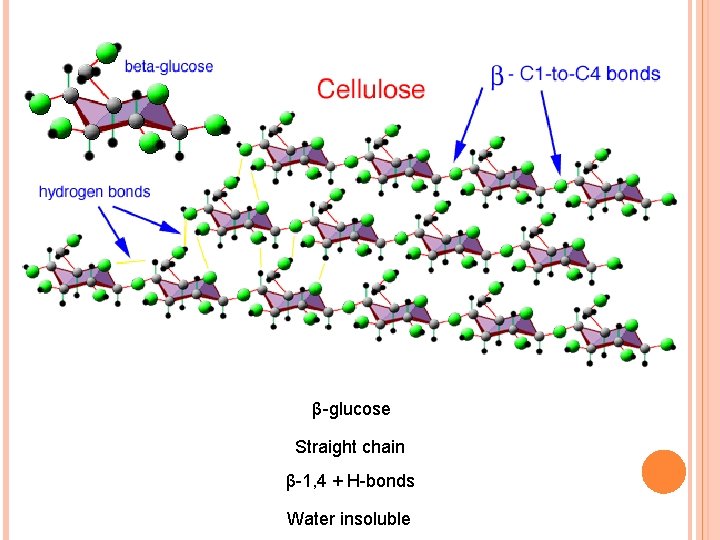
β-glucose Straight chain β-1, 4 + H-bonds Water insoluble
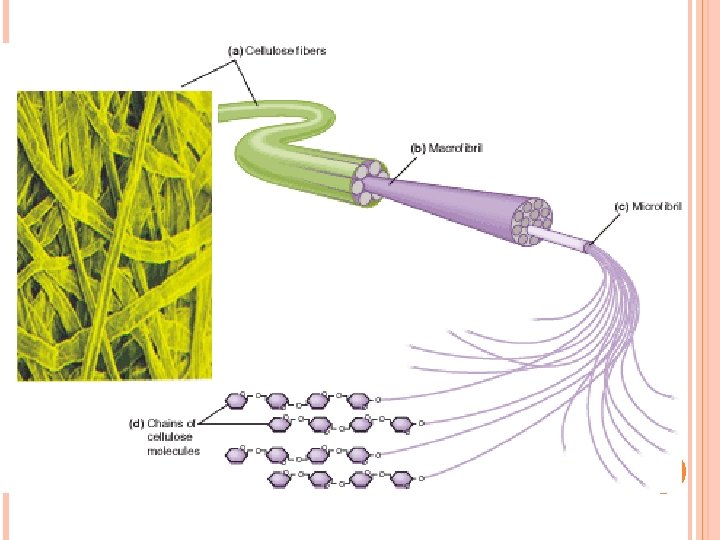

MAJOR FUNCTIONS OF POLYSACCHARIDES IN THE HUMAN BODY Carbohydrates have many uses: a)They provide energy: bread, corn, cereals, rice, potatoes, fruits. b)The store energy: starch is stored in the liver of the animals in the form of glycogen. Glycogen is hydrolyzed to glucose. c)They used as precursors for other important biological molecules (nucleic acids) d)They are used as dietary fibres: cellulose, lignin, pectin in fruits, grains, vegetables


Dietary fibre Water insoluble Cellulose, lignin Whole grain foods, brown rice, carrots, tomatoes Absorb water Bulk moving food through digestive system Water soluble Pectin Oatmeal, beans, peas, lentils, most fruits Fermented by bacteria to carboxylic acids Stabilize blood glucose levels (prevent diabetes) Stimulate production of antibodies Prevent: Constipation Irritable bowel Obesity Haemorrhoids
- Slides: 31