Carbohydrates Structure and Biological Function Monosaccharides Carbohydrates in

Carbohydrates Structure and Biological Function Monosaccharides Carbohydrates in Cyclic Structures Reactions of Glucose and Other Monosaccharides Polysaccharides Glycoproteins 1

Carbohydrates Compounds containing C, H and O General formula : Cx(H 2 O)y All have C=O and -OH functional groups. Classified based on • Size of base carbon chain • Number of sugar units • Location of C=O • Stereochemistry 2

Types of carbohydrates Classifications based on number of sugar units in total chain. Monosaccharides Disaccharides Oligosaccharides Polysaccharides - single sugar unit - two sugar units - 2 to 10 sugar units - more than 10 units Chaining relies on ‘bridging’ of oxygen atoms glycoside bonds 3
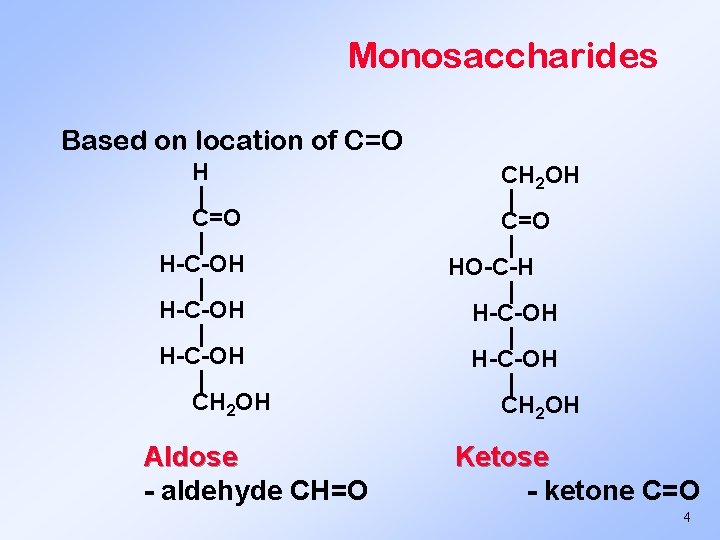
Monosaccharides Based on location of C=O H | C=O | H-C-OH | CH 2 OH Aldose - aldehyde CH=O CH 2 OH | C=O | HO-C-H | H-C-OH | CH 2 OH Ketose - ketone C=O 4
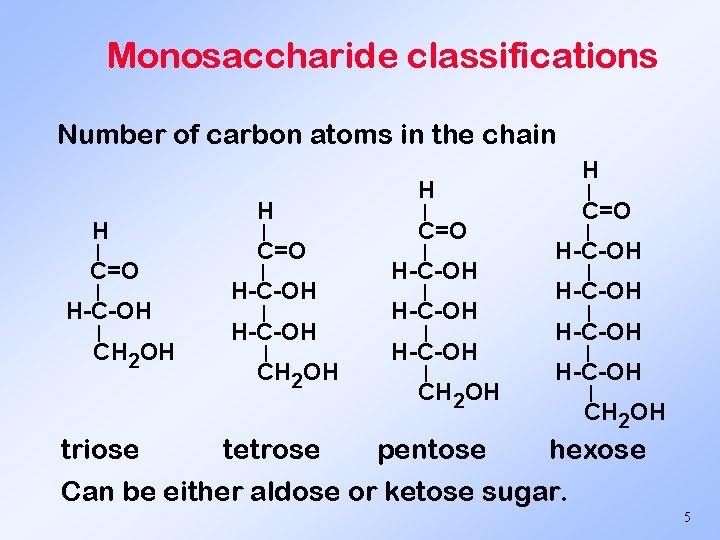
Monosaccharide classifications Number of carbon atoms in the chain H | C=O | H-C-OH | CH 2 OH triose H | C=O | H-C-OH | CH 2 OH tetrose H H | C=O | H-C-OH | CH 2 OH pentose | H-C-OH | CH 2 OH hexose Can be either aldose or ketose sugar. 5
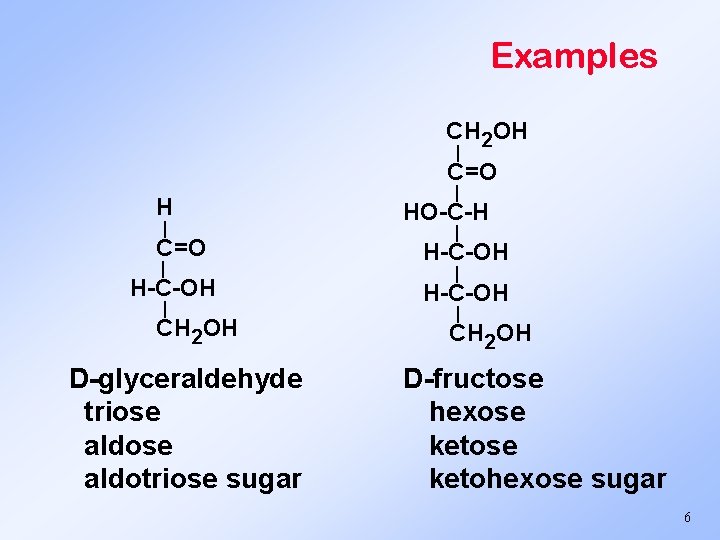
Examples CH 2 OH | C=O | H-C-OH | CH 2 OH D-glyceraldehyde triose aldotriose sugar | HO-C-H | H-C-OH | CH 2 OH D-fructose hexose ketohexose sugar 6
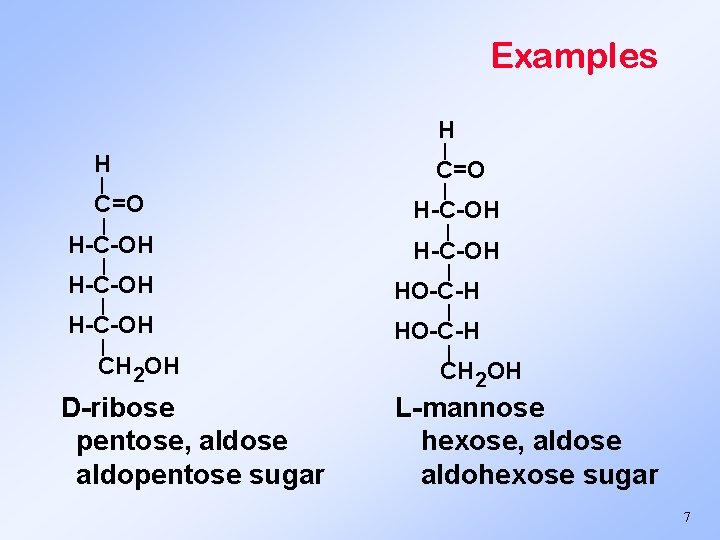
Examples H H | C=O | H-C-OH | CH 2 OH D-ribose pentose, aldose aldopentose sugar | C=O | H-C-OH | HO-C-H | CH 2 OH L-mannose hexose, aldose aldohexose sugar 7

Stereoisomers Stereochemistry Study of the spatial arrangement of molecules. Stereoisomers have • the same order and types of bonds. • different spatial arrangements. • different properties. Many biologically important chemicals, like sugars, exist as stereoisomers. Your body can tell the difference. 8

Enantiomers Pairs of stereoisomers Designated by D- or L- at the start of the name. They are mirror images that can’t be overlapped. If you don’t believe it, give it a try! 9
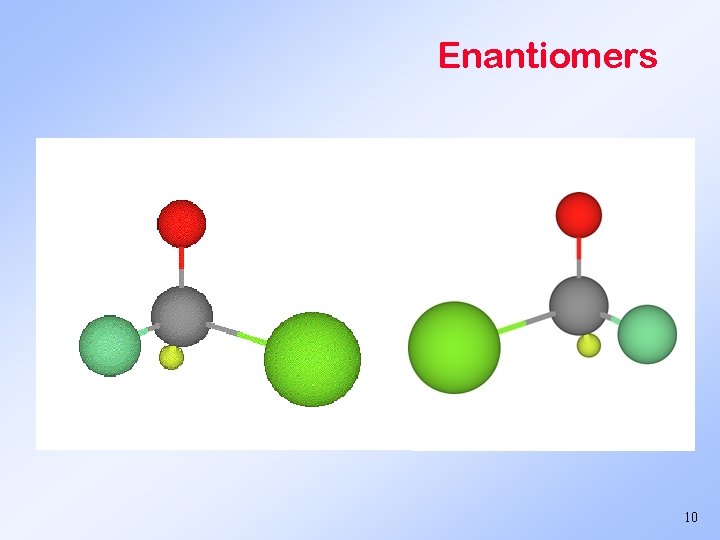
Enantiomers 10
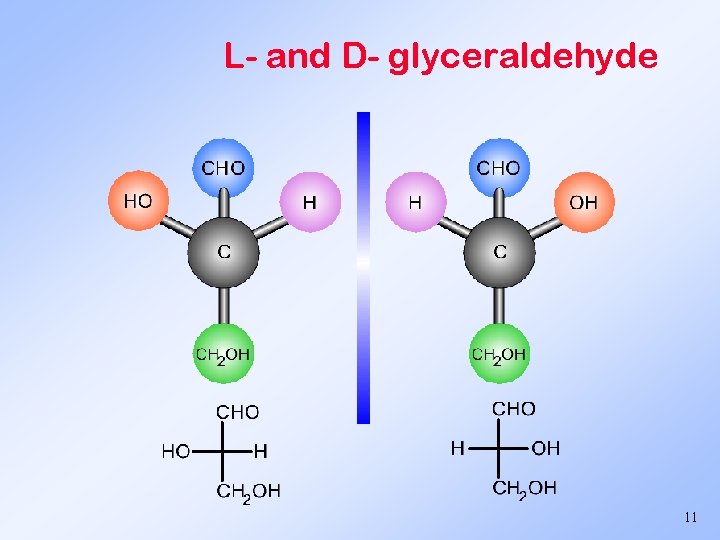
L- and D- glyceraldehyde 11

Enantiomers Chiral center. Asymmetric carbon - 4 different things are attached to it. Cl | I- C - F | Chiral center Br You must have at least one asymmetric carbon to have stereoisomers. 12
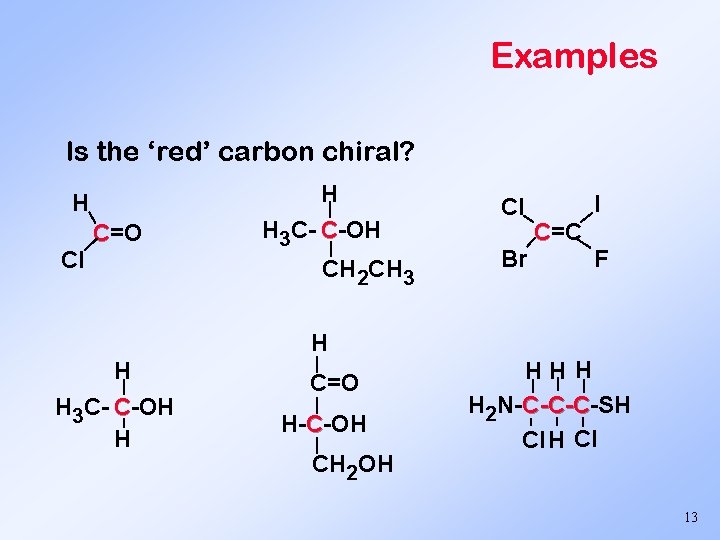
Examples Is the ‘red’ carbon chiral? H Cl C=O H H 3 C- C-OH CH 2 CH 3 Cl Br I C=C F H H H 3 C- C-OH H | C=O | H-C-OH | CH 2 OH HH H H 2 N-C-C-C-SH C-C-C Cl H Cl 13

Physical properties Optical activity ability to rotate plane polarized light. dextrorotatory - rotate to right - use + symbol - usually D isomers levorotatory - rotate to left - use - symbol - usually L isomers 14
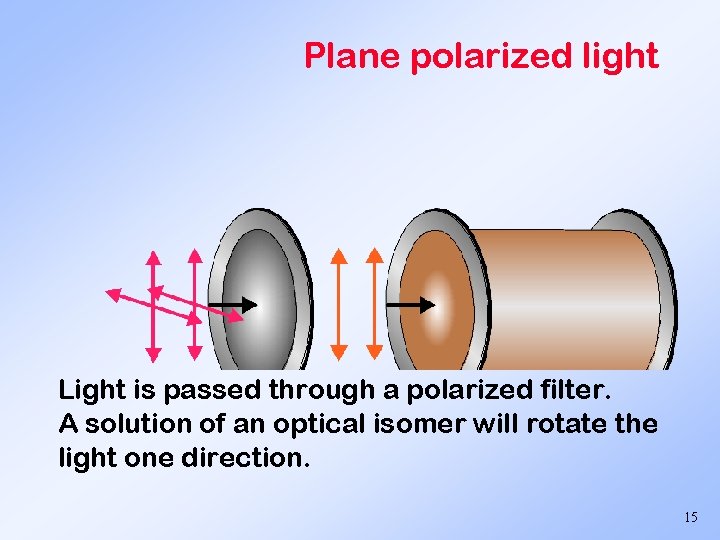
Plane polarized light Light is passed through a polarized filter. A solution of an optical isomer will rotate the light one direction. 15

Stereochemistry Properly drawing enantiomers in 3 -D is hard. Use Fischer Projections Specific type of formula that designates the orientation of groups. H H | C=O | H-C-OH | CH 2 OH 16
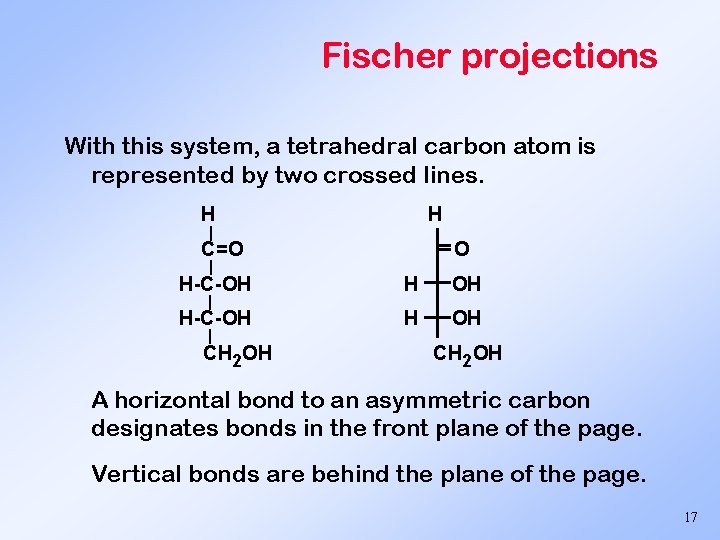
Fischer projections With this system, a tetrahedral carbon atom is represented by two crossed lines. H H | O C=O | H-C-OH H OH | | CH 2 OH A horizontal bond to an asymmetric carbon designates bonds in the front plane of the page. Vertical bonds are behind the plane of the page. 17
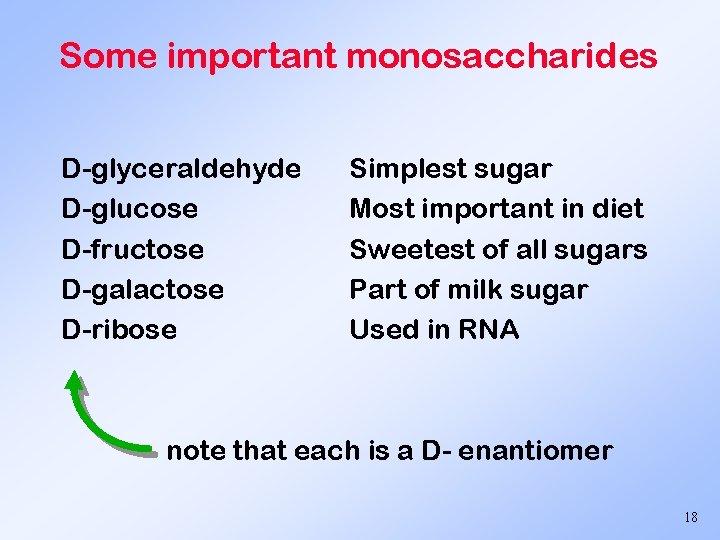
Some important monosaccharides D-glyceraldehyde D-glucose D-fructose D-galactose D-ribose Simplest sugar Most important in diet Sweetest of all sugars Part of milk sugar Used in RNA note that each is a D- enantiomer 18
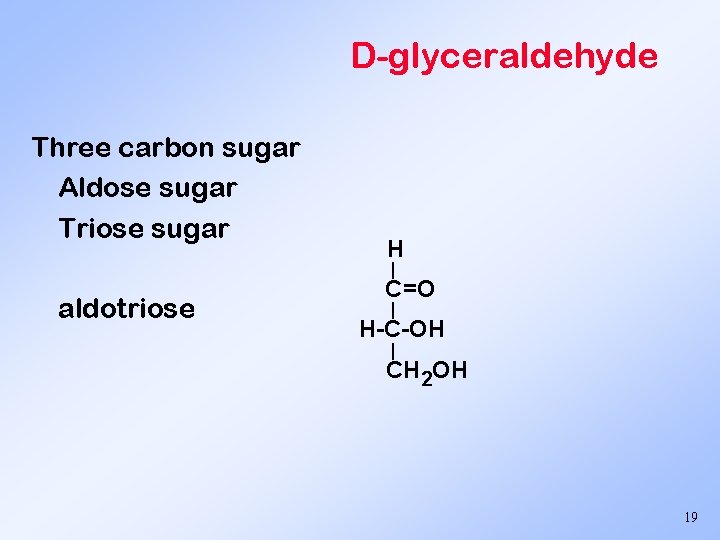
D-glyceraldehyde Three carbon sugar Aldose sugar Triose sugar H | aldotriose C=O | H-C-OH | CH 2 OH 19
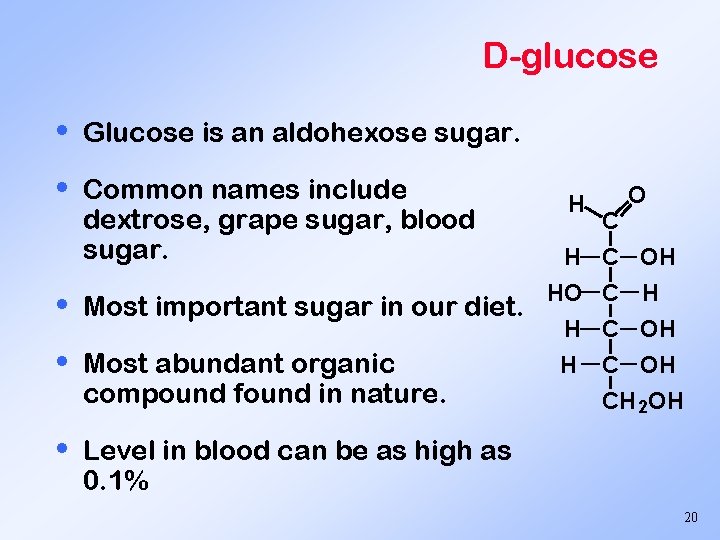
D-glucose • Glucose is an aldohexose sugar. • Common names include dextrose, grape sugar, blood sugar. • Most important sugar in our diet. • Most abundant organic compound found in nature. • H C OH HO C H H C OH CH 2 OH Level in blood can be as high as 0. 1% 20

D-fructose (levulose Rotation is left) Another common sugar. CH 2 OH | C=O | It is a ketohexose. HO-C-H | H-C-OH | Sweetest of all sugars. H-C-OH | CH 2 OH 21
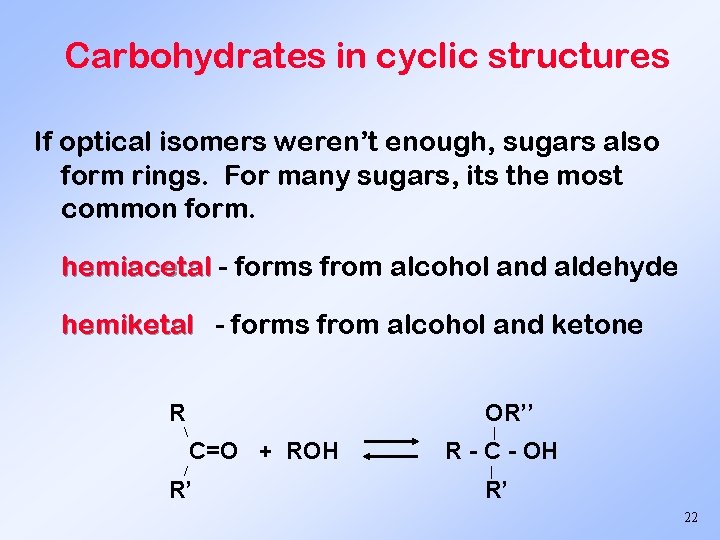
Carbohydrates in cyclic structures If optical isomers weren’t enough, sugars also form rings. For many sugars, its the most common form. hemiacetal - forms from alcohol and aldehyde hemiketal - forms from alcohol and ketone R / OR’’ C=O + ROH R’ | R - C - OH | R’ 22
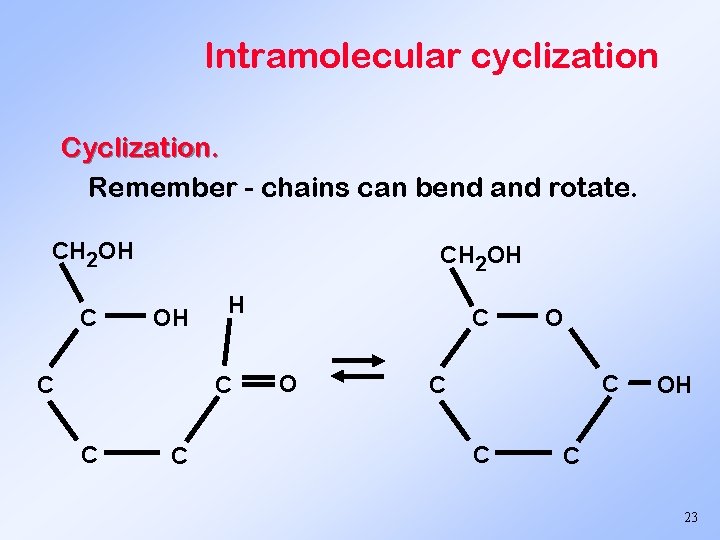
Intramolecular cyclization Cyclization. Remember - chains can bend and rotate. CH 2 OH C CH 2 OH OH C C C C O O C C C OH C 23

Intramolecular cyclization The -OH group that forms can be above or below the ring resulting in two forms anomers and are used to identify the two forms. · - OH group is down compared to CH 2 OH (trans). · - OH group is up compared to CH 2 OH (cis). 24
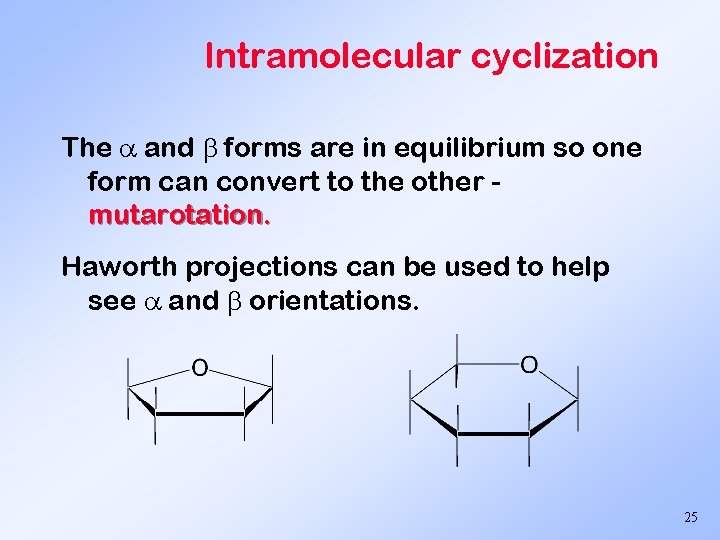
Intramolecular cyclization The and forms are in equilibrium so one form can convert to the other mutarotation. Haworth projections can be used to help see and orientations. 25
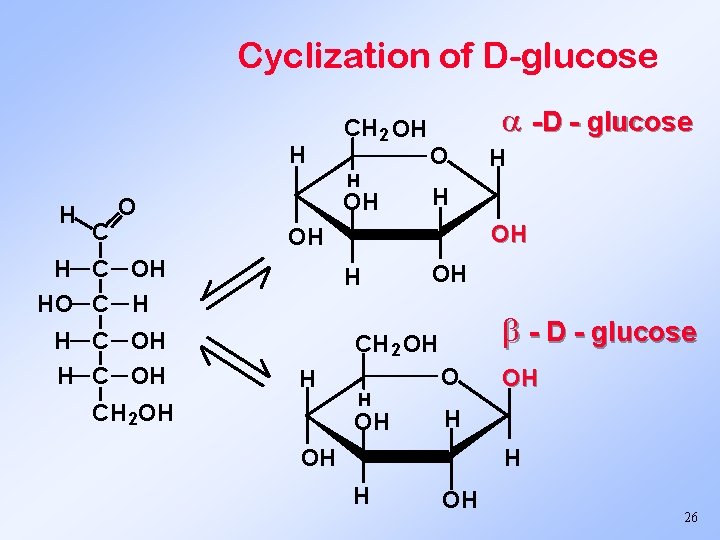
Cyclization of D-glucose H CH 2 OH H H C OH O O OH H OH - D - glucose CH 2 OH H CH 2 OH H H OH H C OH HO C H H C OH -D - glucose H OH O OH H H OH 26
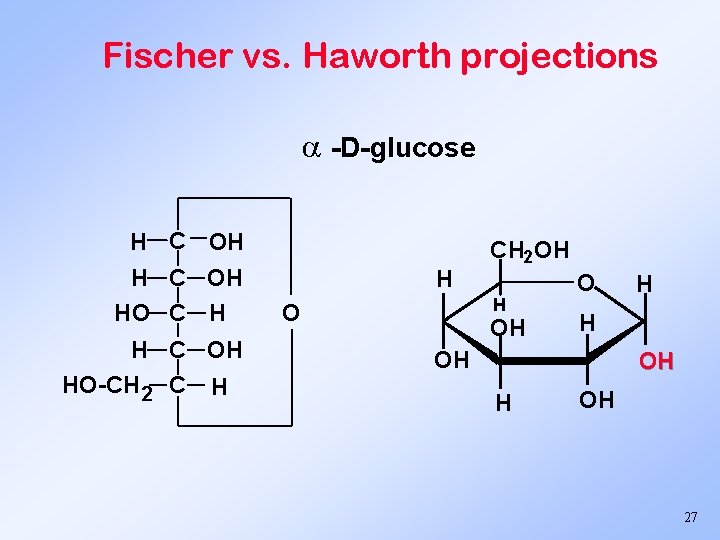
Fischer vs. Haworth projections -D-glucose H C OH HO C H H C OH HO-CH 2 C H H CH 2 OH H O OH O H H OH OH 27
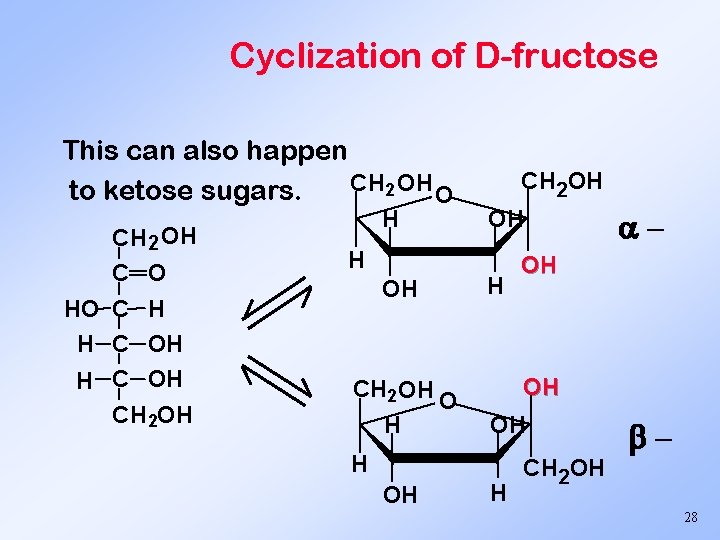
Cyclization of D-fructose This can also happen CH 2 OH O to ketose sugars. CH 2 OH C O HO C H H C OH CH 2 OH H H OH CH 2 OH O H CH 2 OH OH H CH 2 OH 28
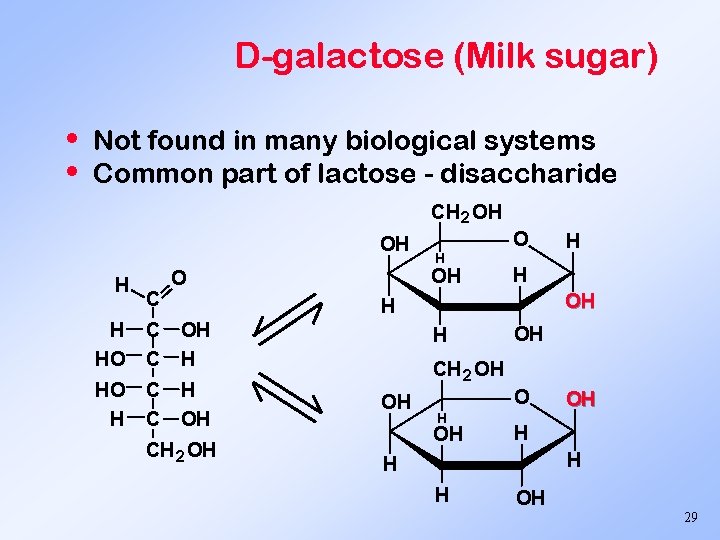
D-galactose (Milk sugar) • • Not found in many biological systems Common part of lactose - disaccharide CH 2 OH OH H C O H OH H H HO C C OH H HO H C C H OH CH 2 OH H H OH CH 2 OH OH OH H H OH 29

D-glucose vs. D-galactose D-glucose H C O D-galactose H C OH HO C H H C OH CH 2 OH Can you find a difference? Your body can! You can’t digest galactose - it must be converted to glucose first. 30

D-ribose An important sugar used in genetic material. This sugar is not used as an energy source but is a part of the backbone of RNA. When the C-2 OH is removed, the sugar becomes deoxyribose which is used in the backbone of DNA. H | C=O | H-C-OH | CH 2 OH 31

Reactions of glucose and other monosaccharides Oxidation-Reduction. Required for their complete metabolic breakdown. Esterification. Production of phosphate esters. Amino derivatives. Used to produce structural components and glycoprotein. Glycoside formation. Linkage of monosaccharides to form polysaccharides. 32
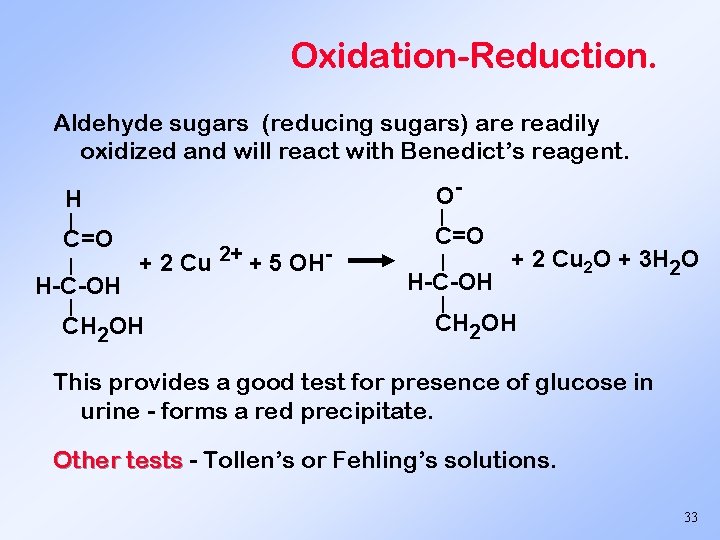
Oxidation-Reduction. Aldehyde sugars (reducing sugars) are readily oxidized and will react with Benedict’s reagent. H O- C=O | | | H-C-OH | + 2 Cu 2+ + 5 OH- CH 2 OH | H-C-OH + 2 Cu 2 O + 3 H 2 O | CH 2 OH This provides a good test for presence of glucose in urine - forms a red precipitate. Other tests - Tollen’s or Fehling’s solutions. 33

Benedict’s reagent glucose 0. 5% 2% Benedict's Reagent 34
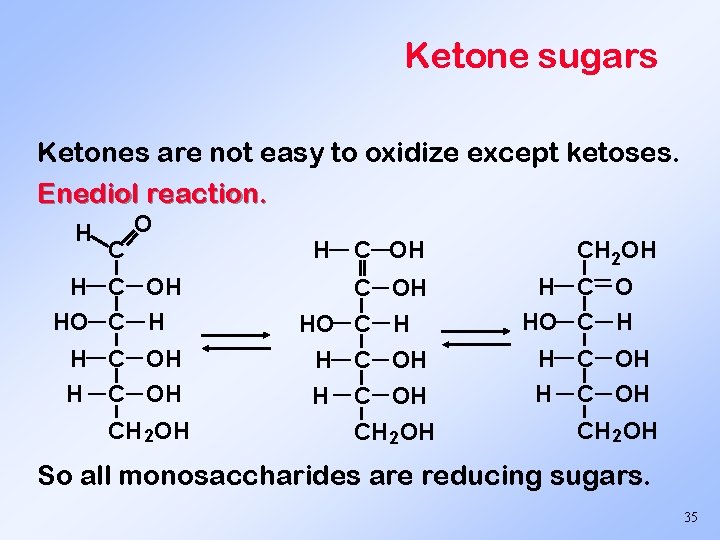
Ketone sugars Ketones are not easy to oxidize except ketoses. Enediol reaction. H C OH HO C H H C OH CH 2 OH H C O HO C H H C OH CH 2 OH So all monosaccharides are reducing sugars. 35

Esterification Esters are formed by reaction of hydroxyl groups (alcohols) with acids. The hydroxyl groups of carbohydrates react similarly to alcohols. 36

Esterification The most important biological esters of carbohydrates are phosphate esters. Example. Phosphoryl group from ATP forms an ester with D-glucose, catalyzed by kinases. kinase D-glucose + ATP D-glucose-6 -phosphate + ADP 37
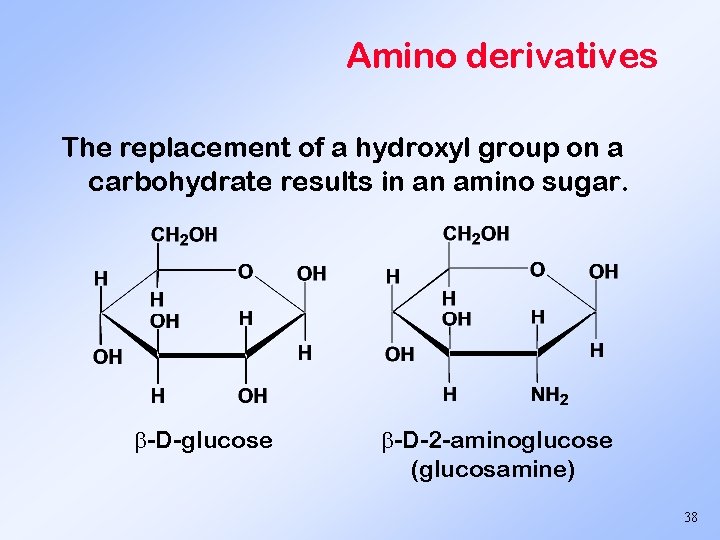
Amino derivatives The replacement of a hydroxyl group on a carbohydrate results in an amino sugar. -D-glucose -D-2 -aminoglucose (glucosamine) 38

Amino derivatives Uses for amino sugars. Structural components of bacterial cell walls. As a component of chitin, a polymer found in the exoskeleton of insects and crustaceans. A major structural unit of chondroitin sulfate - a component of cartilage. Component of glycoprotein and glycolipids. 39
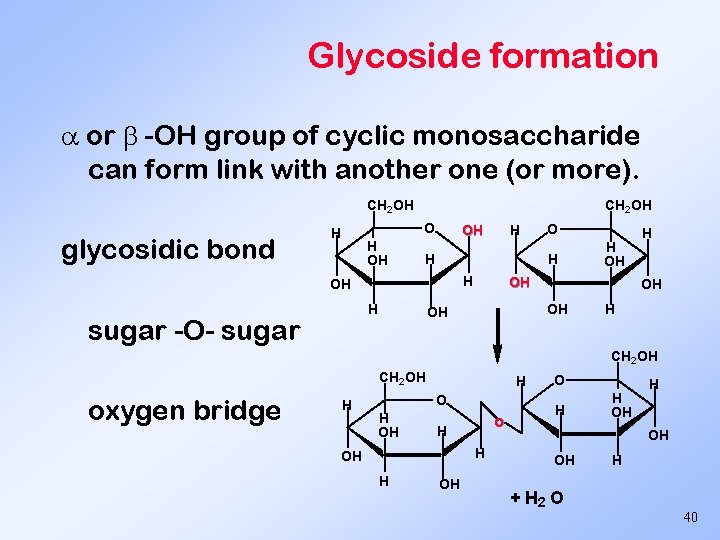
Glycoside formation or -OH group of cyclic monosaccharide can form link with another one (or more). CH 2 OH glycosidic bond CH 2 OH O H H OH OH H O H OH sugar -O- sugar H H OH OH OH H H CH 2 OH oxygen bridge H H OH o H H OH OH + H 2 O H 40

Glycosidic bonds Type is based on the position of the C-1 OH glycosidic bond - linkage between a C-1 OH and a C-4 OH bonds O O C-4 end can be either up or down depending on the orientation of the monosaccharide. 41
Glycosidic bonds O O O 42 O

Glycosidic bonds General format used to describe bond. OH type ( or ) ( carbon# of first sugar carbon# of second sugar ) As we work through the next few examples this will become clear. For disaccharides - the sugar is either or based on form of the remaining C-1 OH. 43
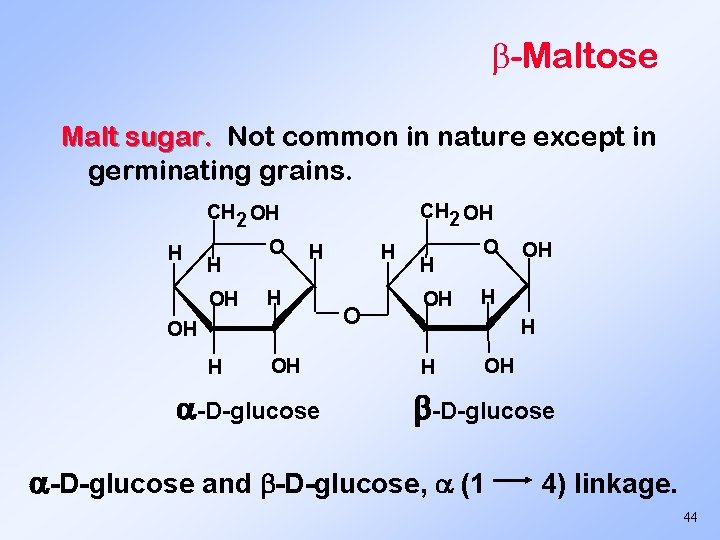
-Maltose Malt sugar. Not common in nature except in germinating grains. CH 2 OH H H OH O H H OH -D-glucose H OH O OH H OH -D-glucose and -D-glucose, (1 4) linkage. 44
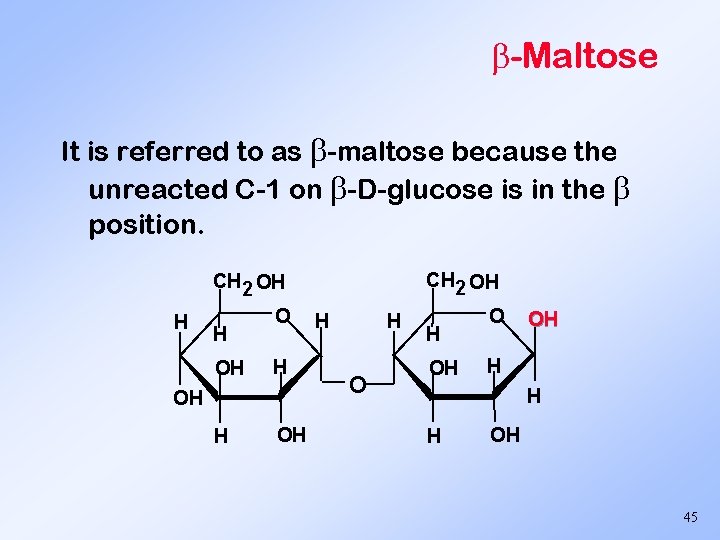
-Maltose It is referred to as -maltose because the unreacted C-1 on -D-glucose is in the position. CH 2 OH H H OH O H OH H H OH O OH H OH 45

-Maltose Uses for -maltose Ingredient in infant formulas. Production of beer. Flavoring - fresh baked aroma. It is hydrolyzed the in body by: maltose + H 2 O maltase 2 glucose 46
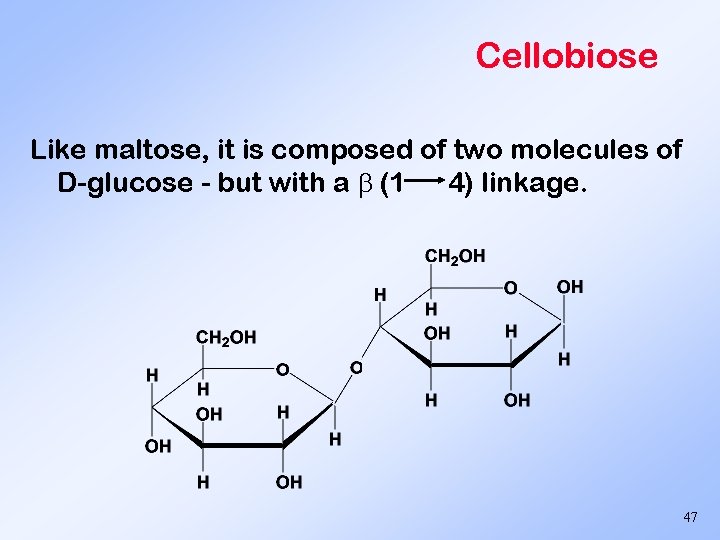
Cellobiose Like maltose, it is composed of two molecules of D-glucose - but with a (1 4) linkage. 47
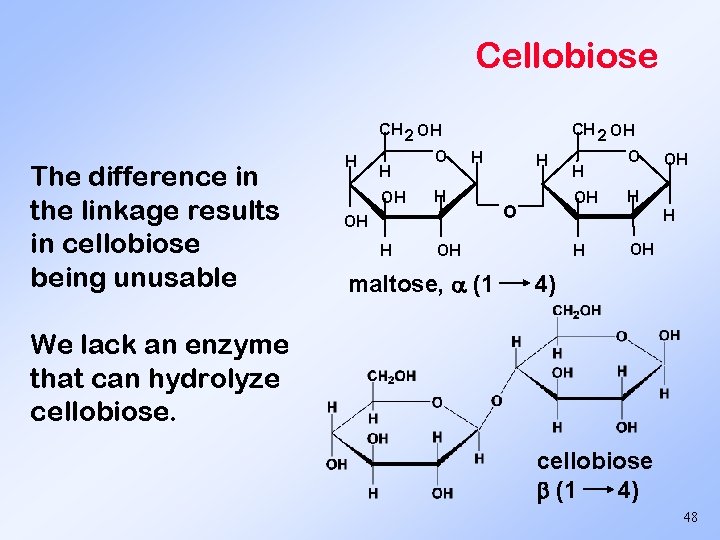
Cellobiose CH 2 OH The difference in the linkage results in cellobiose being unusable H H OH O OH H H OH maltose, (1 H OH 4) We lack an enzyme that can hydrolyze cellobiose (1 4) 48
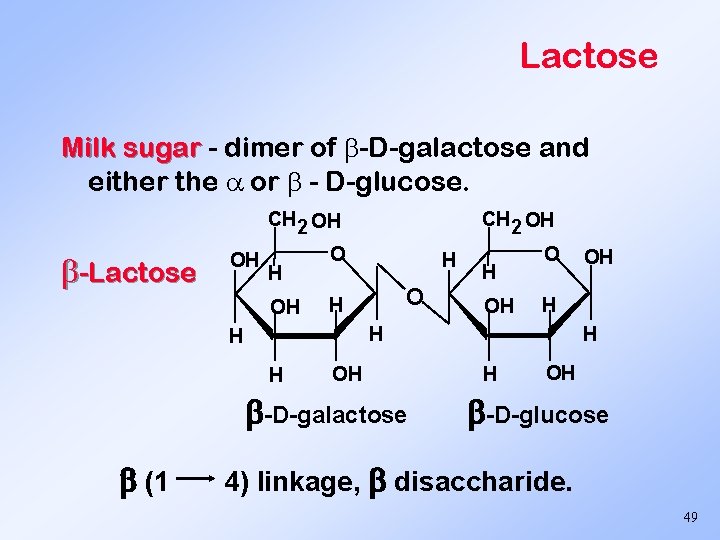
Lactose Milk sugar - dimer of -D-galactose and either the or - D-glucose. CH 2 OH -Lactose OH H OH O H OH -D-galactose OH H H (1 H OH -D-glucose 4) linkage, disaccharide. 49

Lactose We can’t directly use galactose. It must be converted to a form of glucose. Galactosemia - absence of needed enzymes needed for conversion. Build up of galactose or a metabolite like dulcitol (galactitol) causes toxic effects. Can lead to retardation, cataracts, death. 50

Lactose Lactase Enzyme required to hydrolyze lactose. Lactose intolerance Lack or insufficient amount of the enzyme. If lactase enters lower GI, it can cause gas and cramps. 51
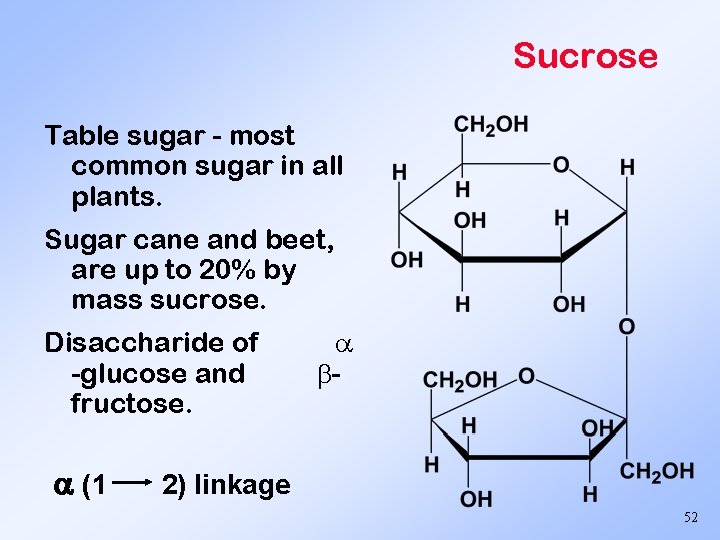
Sucrose Table sugar - most common sugar in all plants. Sugar cane and beet, are up to 20% by mass sucrose. Disaccharide of -glucose and fructose. (1 - 2) linkage 52
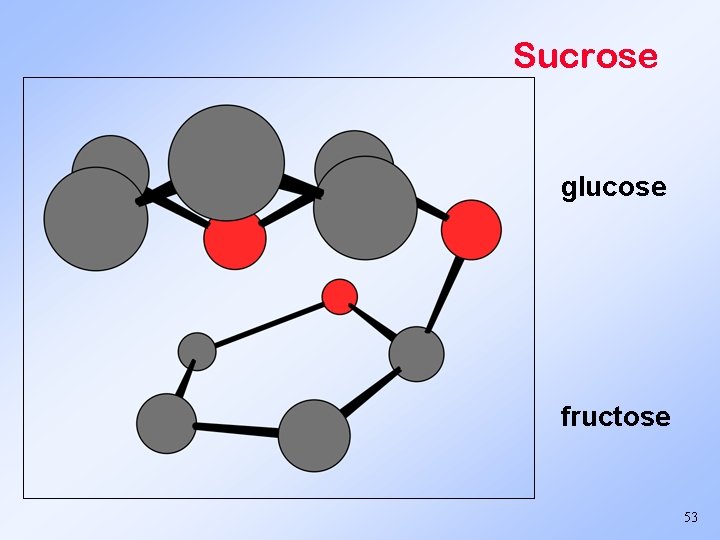
Sucrose glucose fructose 53
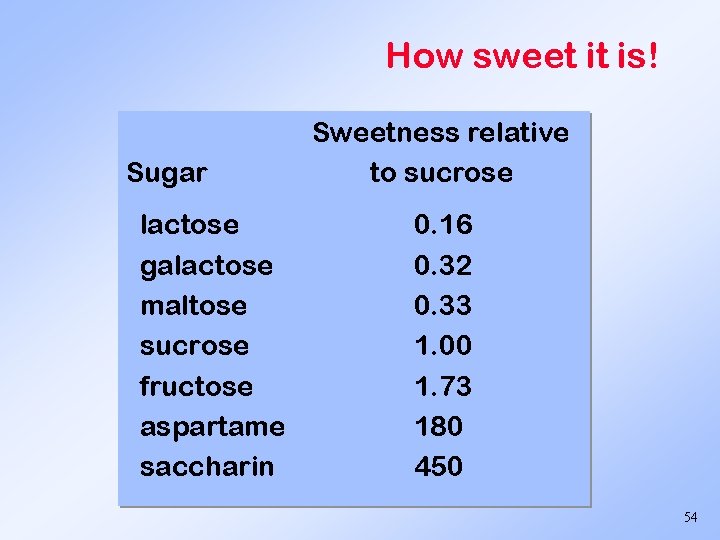
How sweet it is! Sugar lactose galactose maltose sucrose fructose aspartame saccharin Sweetness relative to sucrose 0. 16 0. 32 0. 33 1. 00 1. 73 180 450 54
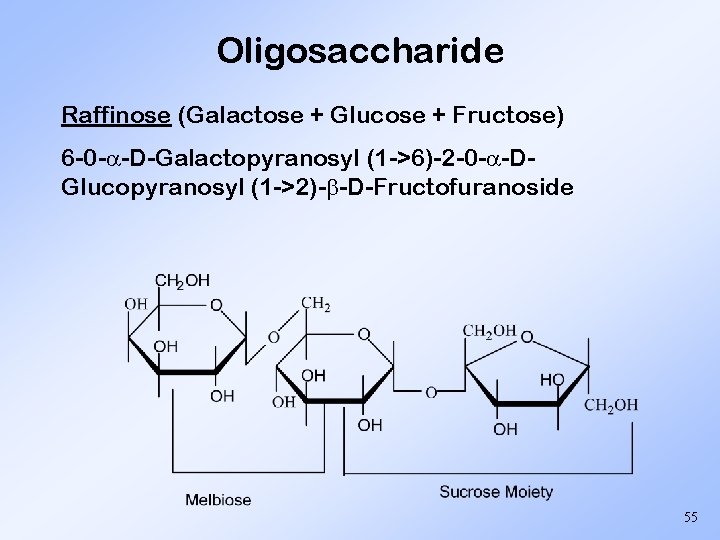
Oligosaccharide Raffinose (Galactose + Glucose + Fructose) 6 -0 - -D-Galactopyranosyl (1 ->6)-2 -0 - -DGlucopyranosyl (1 ->2)- -D-Fructofuranoside 55
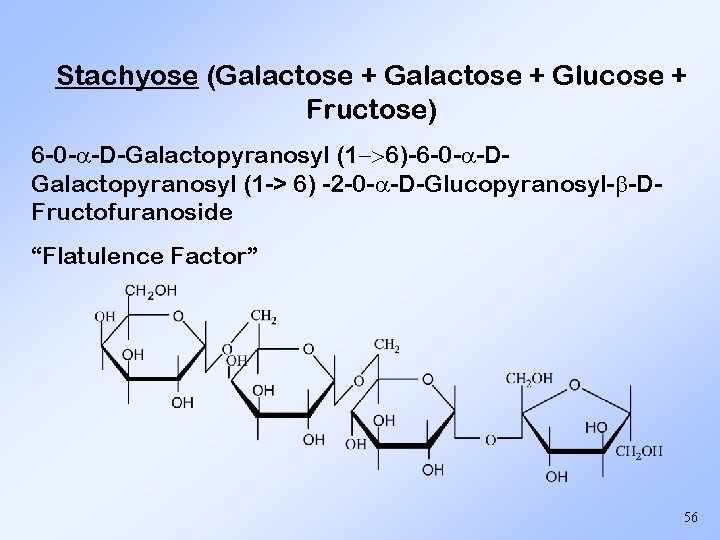
Stachyose (Galactose + Glucose + Fructose) 6 -0 - -D-Galactopyranosyl (1 >6)-6 -0 - -DGalactopyranosyl (1 -> 6) -2 -0 - -D-Glucopyranosyl- -DFructofuranoside “Flatulence Factor” 56

Polysaccharides Carbohydrate polymers Storage Polysaccharides Energy storage - starch and glycogen Structural Polysaccharides Used to provide protective walls or lubricative coating to cells - cellulose and mucopolysaccharides. Structural Peptidoglycans Bacterial cell walls 57

Starch Energy storage used by plants Long repeating chain of -D-glucose Chains up to 4000 units Amylose straight chain major form of starch Amylopectin branched structure 58
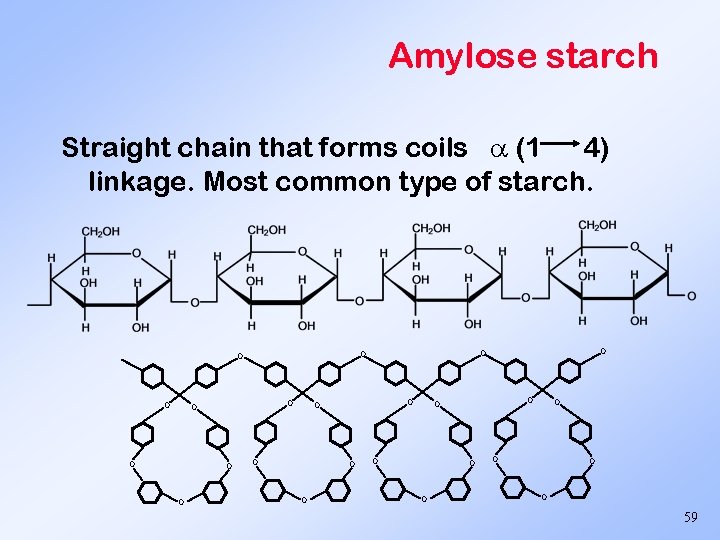
Amylose starch Straight chain that forms coils (1 4) linkage. Most common type of starch. O O O O O O 59
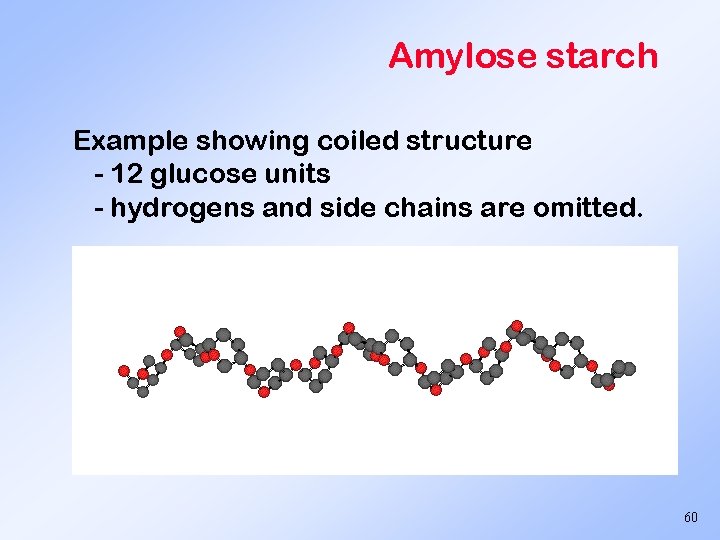
Amylose starch Example showing coiled structure - 12 glucose units - hydrogens and side chains are omitted. 60
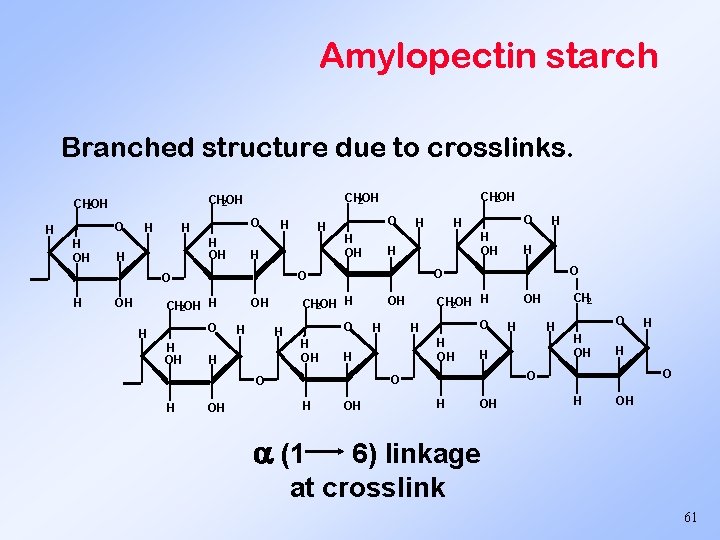
Amylopectin starch Branched structure due to crosslinks. O H H OH H H OH O H OH H CH 2 OH H H H OH OH O H H O CH 2 OH H OH CH 2 OH H O H OH H H O O O H H O CH 2 OH H H O H CH 2 OH H OH (1 6) linkage at crosslink 61

MODIFIED STARCHES 1. Pregelatinized Starch --- simple precooked and roll dried to give product that readily disperses in cold water. 2. Thin-boiling or Acid-modified Starch --suspending granular starch in a very dilute acid under somewhat below its gelatinization temperature (somewhat mild). 3. Oxidized Starches --- oxidation of starch with alkaline hypochloride to get -coo- at C 6. 4. Dextrins – Intermediate in the hydrolysis of starch 62
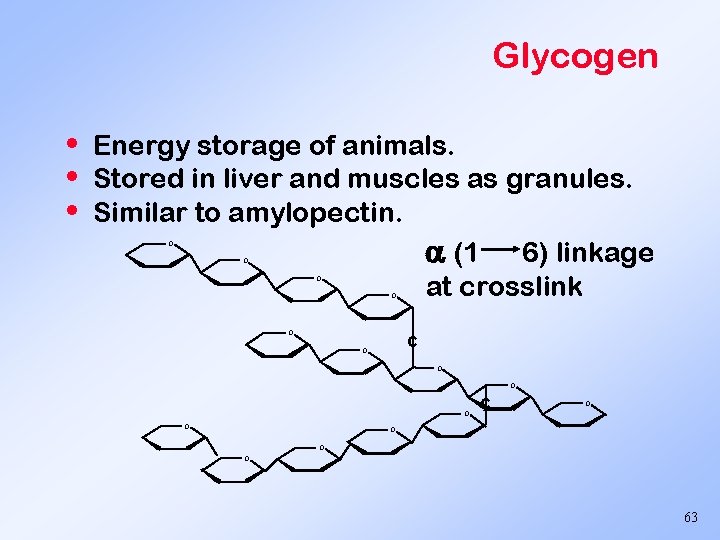
Glycogen • • • Energy storage of animals. Stored in liver and muscles as granules. Similar to amylopectin. (1 6) linkage at crosslink O O O O O c O O 63
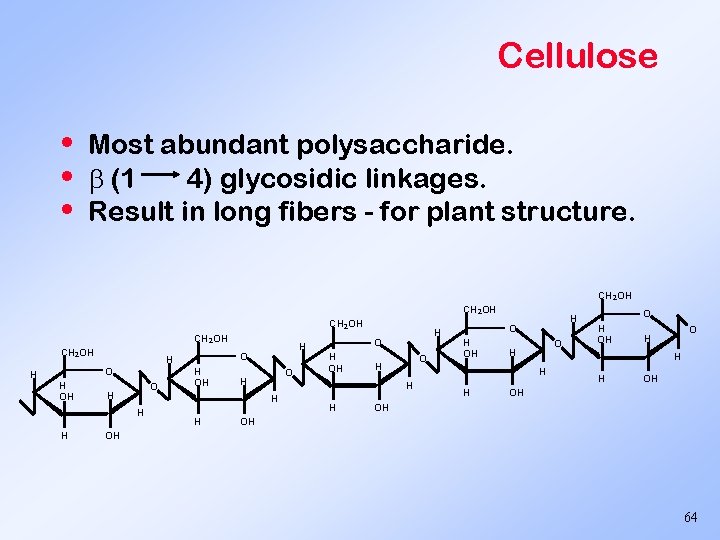
Cellulose • • • Most abundant polysaccharide. (1 4) glycosidic linkages. Result in long fibers - for plant structure. CH 2 OH CH 2 OH O H H H OH O H H H H O H OH OH OH 64
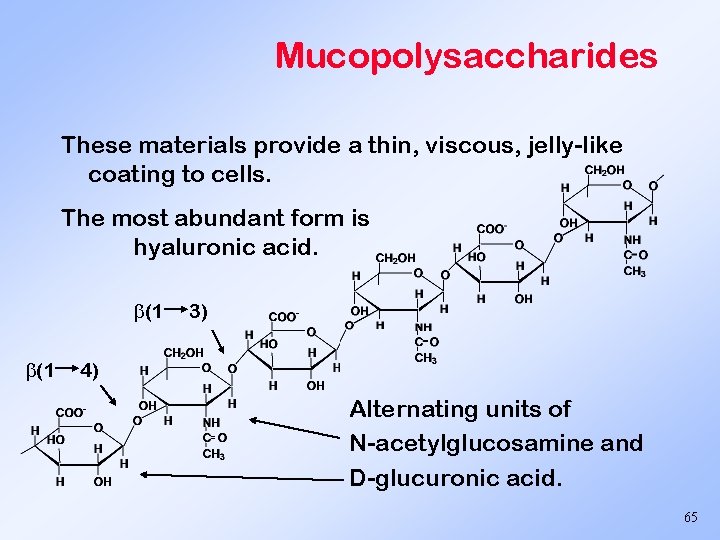
Mucopolysaccharides These materials provide a thin, viscous, jelly-like coating to cells. The most abundant form is hyaluronic acid. (1 (1 3) 4) Alternating units of N-acetylglucosamine and D-glucuronic acid. 65
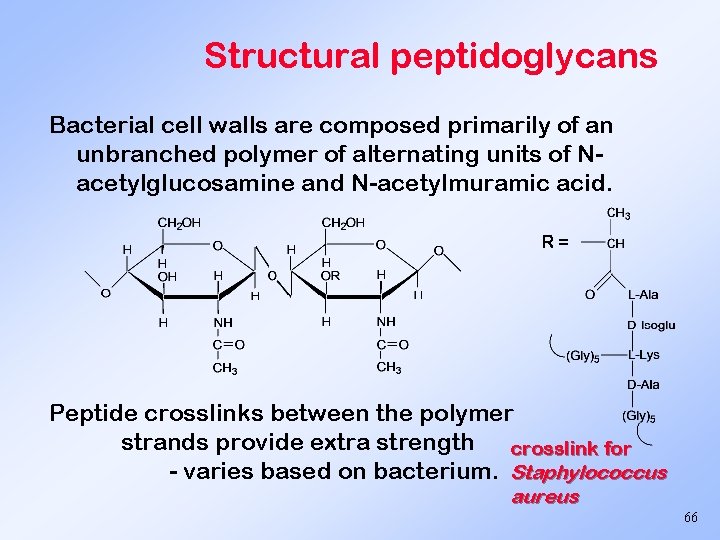
Structural peptidoglycans Bacterial cell walls are composed primarily of an unbranched polymer of alternating units of Nacetylglucosamine and N-acetylmuramic acid. R= Peptide crosslinks between the polymer strands provide extra strength crosslink for - varies based on bacterium. Staphylococcus aureus 66

Glycoproteins / Mucoproteins Proteins that carry covalently bound carbohydrate units. They have many biological functions. • immunological protection • cell-cell recognition • blood clotting • host-pathogen interaction 67

Glycoprotein structure Carbohydrates only account for 1 -30% of the total weight of a glycoprotein. The most common monosaccharides are glucose mannose galactose fucose sialic acid N-acetylgalactosamine N-acetylglucosamine 68
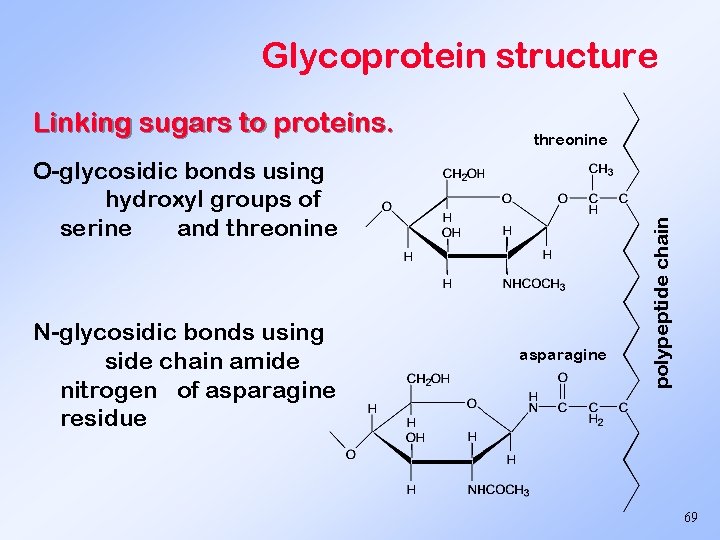
Glycoprotein structure threonine O-glycosidic bonds using hydroxyl groups of serine and threonine N-glycosidic bonds using side chain amide nitrogen of asparagine residue asparagine polypeptide chain Linking sugars to proteins. 69

GLYCOGEN Animal starch. - (1 > 4) linkage and - (1 -> 6) linkage 12 : 1 71
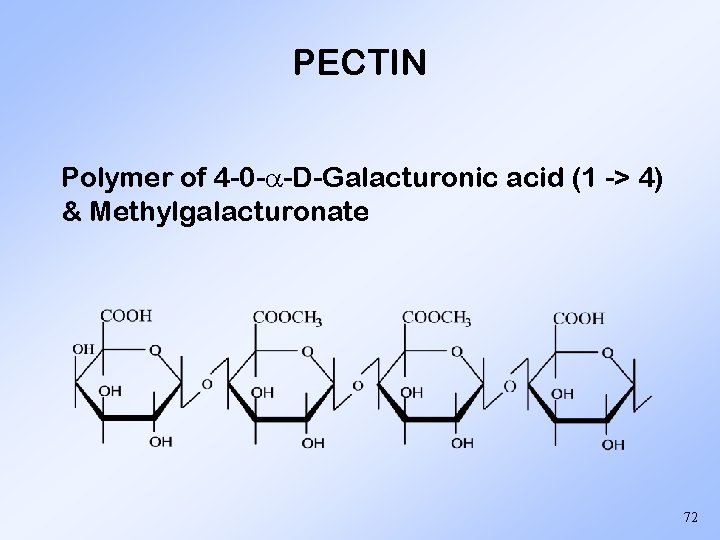
PECTIN Polymer of 4 -0 - -D-Galacturonic acid (1 -> 4) & Methylgalacturonate 72

Inulin / Fructosan: Polysaccharide of fructose Found in tubers and roots Readily soluble in water and is used to determine GFR It is not hydrolyzed by intestinal enzymes 73
- Slides: 72