Carbohydrates Simple Sugars Learning Objectives Distinguish between monosaccharides

Carbohydrates Simple Sugars

Learning Objectives • . Distinguish between monosaccharides, disaccharides and polysaccharides (glycogen and starch – amylose and amylopectin) and relate their structures to their roles in providing and storing energy (β-glucose and cellulose are not required in this topic).

Learning objectives • Describe how monosaccharides join to form disaccharides (sucrose, lactose and maltose) and polysaccharides (glycogen and amylose) through condensation reactions forming glycosidic bonds, and how these can be split through hydrolysis reactions.

Carbohydrates • • • Functions: Energy source Energy store e. g. starch or glycogen Structural material e. g. cellulose Elements made of: Carbon, hydrogen, oxygen General formula Cn(H 2 O)n
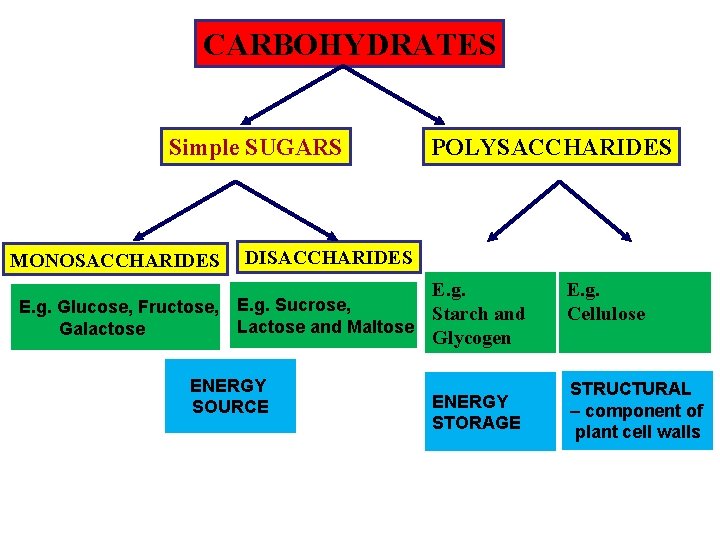
CARBOHYDRATES Simple SUGARS MONOSACCHARIDES POLYSACCHARIDES DISACCHARIDES E. g. Glucose, Fructose, E. g. Sucrose, Starch and Lactose and Maltose Galactose Glycogen ENERGY SOURCE ENERGY STORAGE E. g. Cellulose STRUCTURAL – component of plant cell walls

Monosaccharides • The simplest carbohydrates ‘single sugars’ • Contain between 3 and 6 carbon atoms 3 C’s = Triose sugar e. g. glyceraldehyde 5 – pentose sugar e. g. ribose & deoxyribse 6 – hexose sugar e. g. glucose & fructose, Properties • Water soluble • Sweet • Form crystals
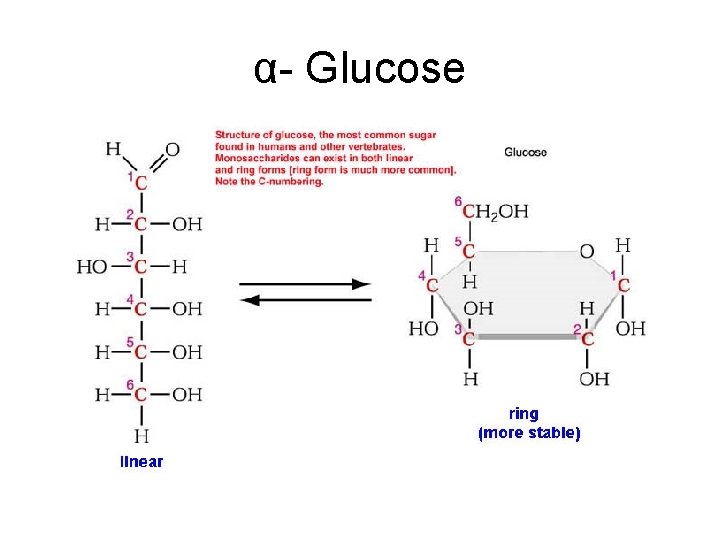
α- Glucose
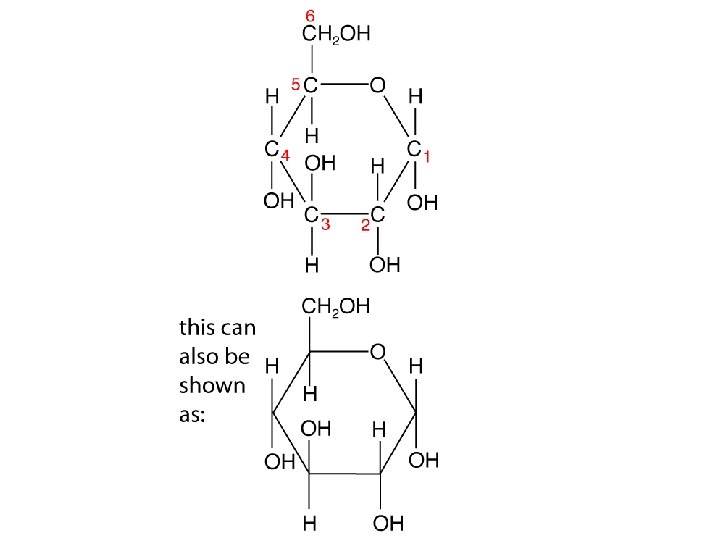
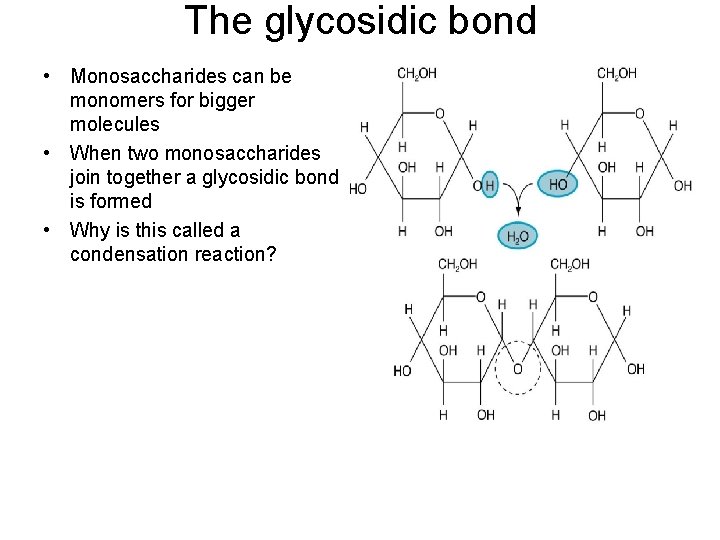
The glycosidic bond • Monosaccharides can be monomers for bigger molecules • When two monosaccharides join together a glycosidic bond is formed • Why is this called a condensation reaction?

Disaccharides • Double sugars made from 2 monosaccharides • Formed by CONDENSATION reaction • Each mono joined by a GLYCOSIDIC bond Examples Maltose = 2 x α-glucose Sucrose = glucose + fructose Lactose = glucose + galactose
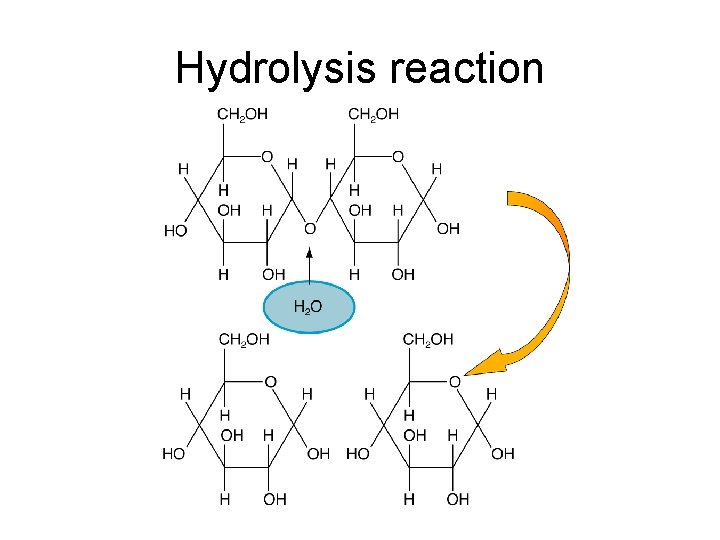
Hydrolysis reaction
- Slides: 11