Carbohydrates Monosaccharide Sugars Disaccharide Carbohydrates Starch Polysaccharide Glycogen

Carbohydrates
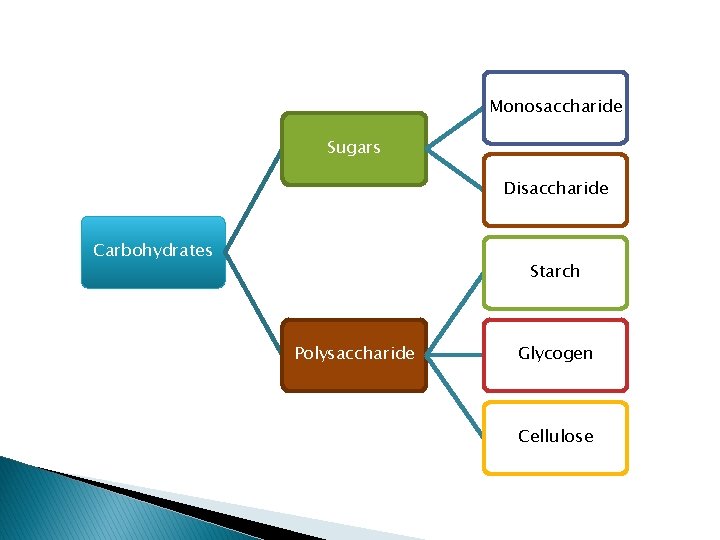
Monosaccharide Sugars Disaccharide Carbohydrates Starch Polysaccharide Glycogen Cellulose

FACTS ABOUT POLYSACCHARDIDES r Mac le u c e ol No om t Sw Insolu ble eet

FACTS ABOUT MONOSACCHARIDES AND DISACCHARIDES all m S les u c le Mo Its Cry sta liz ily d a e Re lubl so e e m a d N n e ays e” w l a os “ n i Sw e et

GLUCOSE Glucose is the main source of energy in respiration due to the large number of CH bonds. When These Bonds are broken large amounts of energy are released. Glucose has a small size and is therefore able to pass through the cell membrane of cells to engage in respiratory reactions. They are building blocks of larger molecules as it can easily undergo condensation reactions with other monomers.
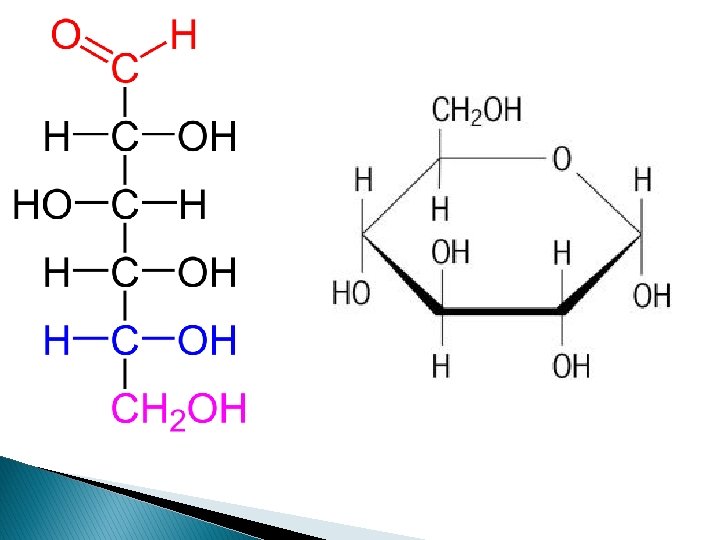
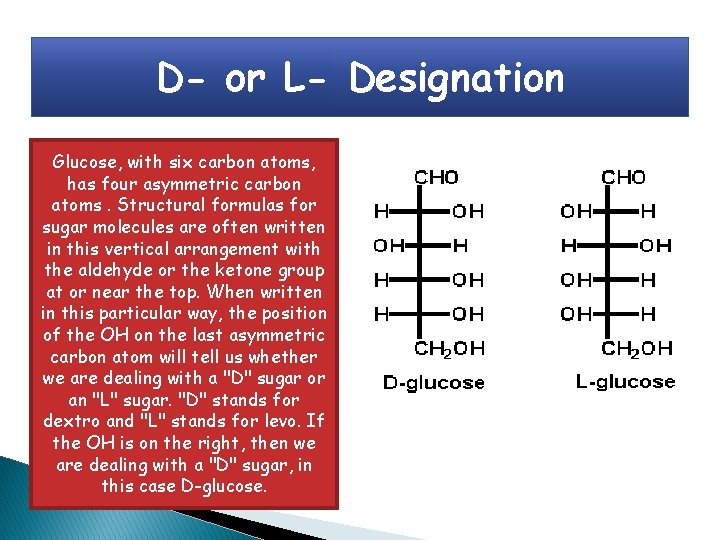
D- or L- Designation Glucose, with six carbon atoms, has four asymmetric carbon atoms. Structural formulas for sugar molecules are often written in this vertical arrangement with the aldehyde or the ketone group at or near the top. When written in this particular way, the position of the OH on the last asymmetric carbon atom will tell us whether we are dealing with a "D" sugar or an "L" sugar. "D" stands for dextro and "L" stands for levo. If the OH is on the right, then we are dealing with a "D" sugar, in this case D-glucose.
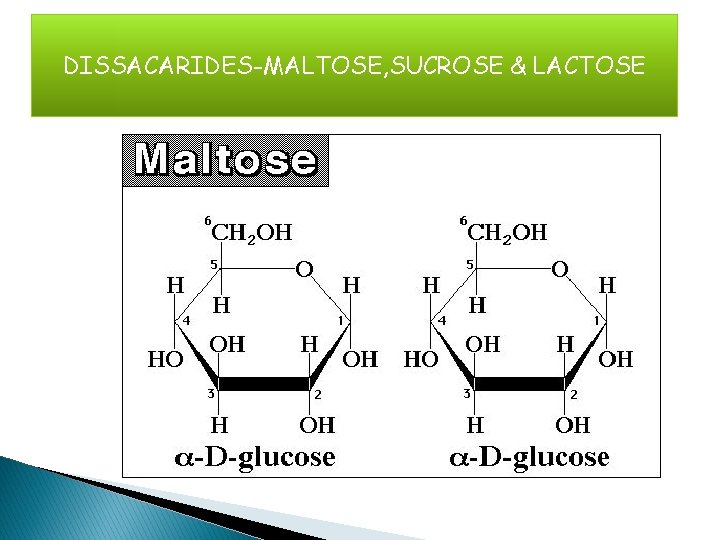
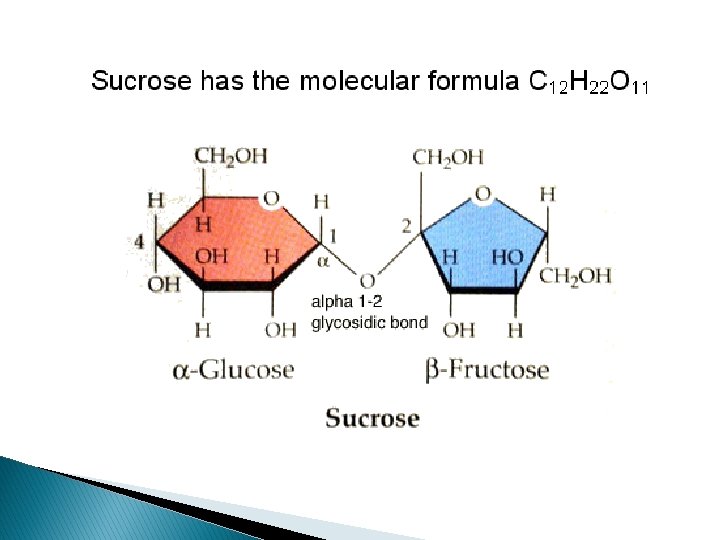
DISSACARIDES-MALTOSE, SUCROSE & LACTOSE
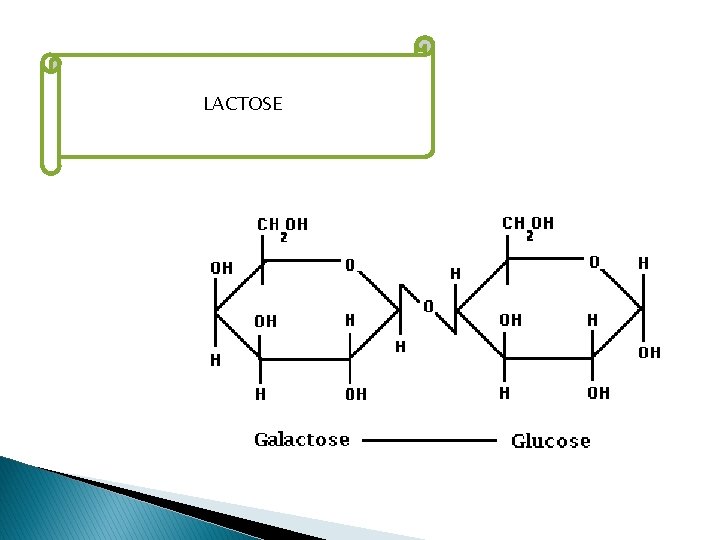
LACTOSE
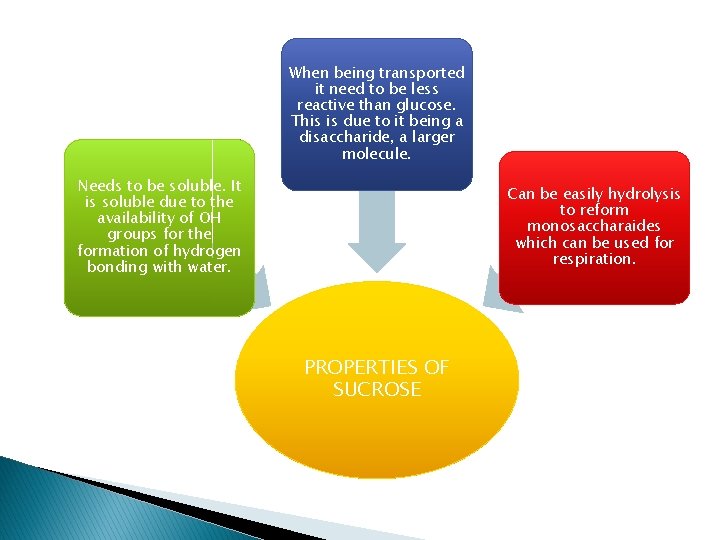
When being transported it need to be less reactive than glucose. This is due to it being a disaccharide, a larger molecule. Needs to be soluble. It is soluble due to the availability of OH groups for the formation of hydrogen bonding with water. Can be easily hydrolysis to reform monosaccharaides which can be used for respiration. PROPERTIES OF SUCROSE
POLYSACCHARIDES- STARCH, GLYCOGEN & CELLULOSE

STARCH Starch is made up of amylose and amylopectin, consisting of alpha glucose molecules. In amylose because the side chains of C 6 are all found above the plane of the ring it is accomodated by having the molecule bend to form a hexical structure. Amylopectin forms branching structures(1 -6 glycosidic bond) making the molecule blunky and compact. A susupension of amylose in water gives a blue-black colour with iodine-potassium iodide solution, whereas amylopectin gives a red-violet colour. This forms the basis of the test for starch.
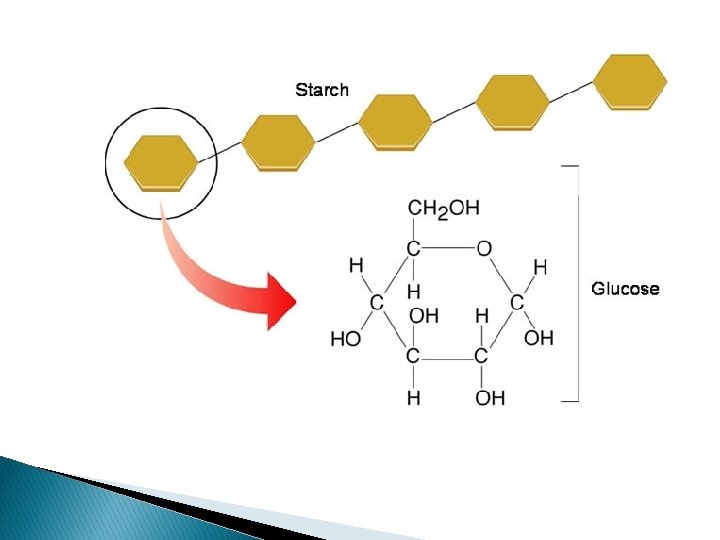

Glycogen is similar in structure to that of amylopectin of starch, however there chains are shorter and more branched.
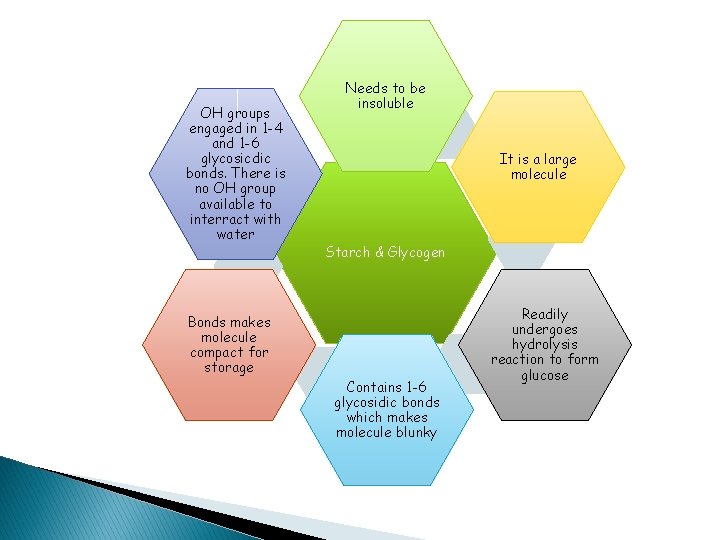
OH groups engaged in 1 -4 and 1 -6 glycosicdic bonds. There is no OH group available to interract with water Needs to be insoluble It is a large molecule Starch & Glycogen Bonds makes molecule compact for storage Contains 1 -6 glycosidic bonds which makes molecule blunky Readily undergoes hydrolysis reaction to form glucose
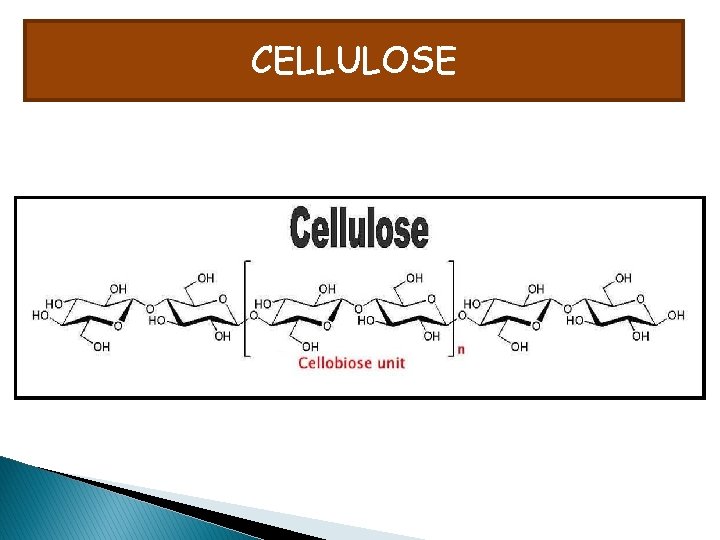
CELLULOSE

PROPERTIES OF CELLULOSE Adjacent monomers need to be rotated at 180 degrees to allow condensation and formation of glycosicdic bond Results in strong molecules because H atoms of OH groups are weakly attracted to O atom of cellulose molecule. Hydrogen bonds are weak but collectively they have enormous strength. Needs to be insoluble in water. The OH groups which would normally engage in hydrogen bonding with water are involved in hydrogen bonding with parallel fibres that run adjacent to each other in addiction OH groups project inward inside the molecule.

- Slides: 19