Carbohydrates Monomers Vs Polymers most biological molecules are

Carbohydrates

Monomers Vs. Polymers - most biological molecules are polymers Monomer - one part Dimer – two parts Oligomer – short chain – 3 – 10 parts Polymer - many repeating parts Macromolecules - combination of polymers

Anabolism of Polymers Anabolism: building reactions Polymerization reaction: 2 units form one larger unit Ex: Building starch from glucose Condensation Reaction or Dehydration Synthesis bond is formed by the removal of a water two hydroxyl groups - one molecule loses OH and one loses an H results in a bond based on the remaining O and the H and the OH combine to form water Requires energy and a catalyst

The Synthesis and Breakdown of Polymers Monomers form larger molecules by condensation reactions called dehydration reactions HO 1 3 2 H Unlinked monomer Short polymer Dehydration removes a water molecule, forming a new bond HO Figure 5. 2 A 1 2 H HO 3 H 2 O 4 H Longer polymer (a) Dehydration reaction in the synthesis of a polymer

Catabolism: breaking reactions Hydrolysis Reaction - addition of water to break a polymer chain Also requires energy and enzymes - but generally gives off more energy than it uses

Polymers can disassemble by Hydrolysis HO 1 2 3 4 Hydrolysis adds a water molecule, breaking a bond HO 1 2 3 H Figure 5. 2 B (b) Hydrolysis of a polymer H H 2 O HO H

Dehydration Synthesis and Hydrolysis Build - anabolic - requires energy Break - catabolic - releases energy NOTE: COMBINATION OF MONOMERS IN DIFFERENT QUANTITIES AND PATTERNS RESULTS IN A WIDE VARIETY OF MOLECULES Ex: Alphabet: letters are monomers words are polymers sentences are macromolecules
Four Major Biological Molecules 1. Carbohydrates 2. Lipids 3. Proteins 4. Nucleic Acids
CARBOHYDRATES Elements: CHO and sometimes N FUNCTION: Energy Structure Protection Storage

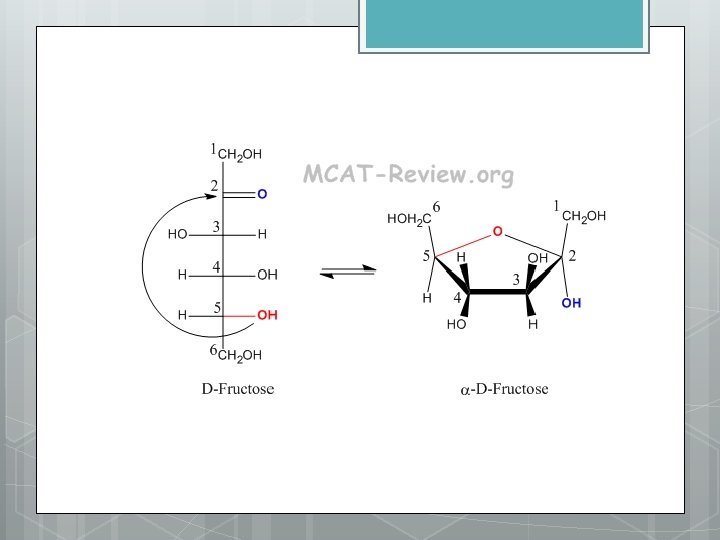
Types of Carbohydrates 1. Sugars: simplest Most common glucose : C 6 H 12 O 6 Classification: Monosaccharides: one sugar unit Ex. Glucose - storage of solar energy via photosynthesis Characteristics: Two types of carbonyls: 1) aldehyde - carbonyl on end = aldose sugar ex. Glucose 2) ketone - carbonyl in middle = ketose sugar ex. fructose - carbonyl position affects ring formation - placement of hydroxyl groups give different properties and results in the formation of isomers Glucose and fructose
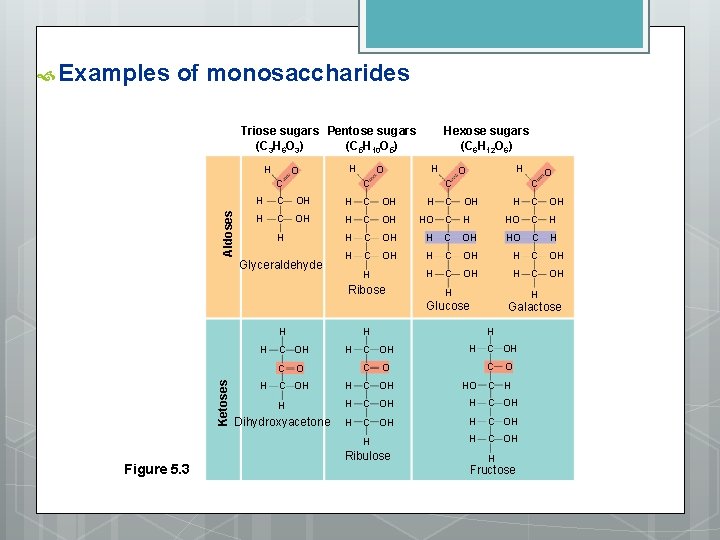
Examples of monosaccharides Triose sugars Pentose sugars (C 3 H 6 O 3) (C 5 H 10 O 5) H O H Aldoses C O Hexose sugars (C 6 H 12 O 6) H C H O C H C OH H C OH HO C H C OH H Glyceraldehyde H Ribose H C OH H HO C H C OH HO C H H C OH H H Glucose H H Ketoses C Figure 5. 3 H C OH H C O Galactose H C OH C O O C OH HO H H C OH Dihydroxyacetone H C OH H Ribulose O C C H H Fructose
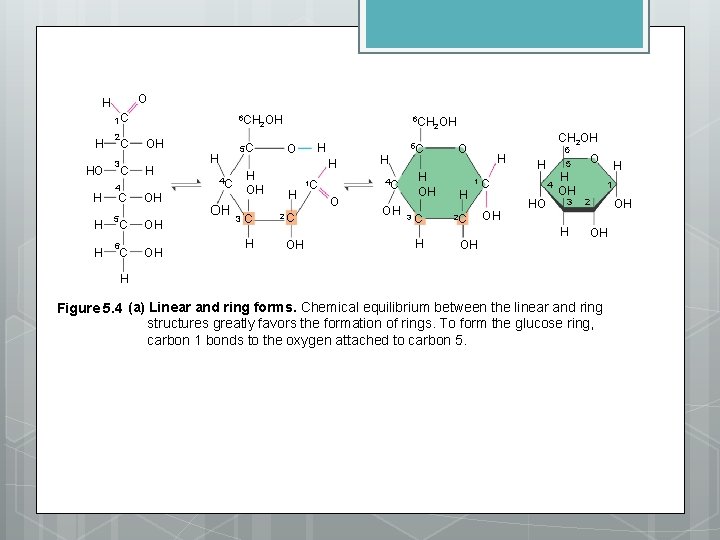
O H 1 C H HO H 2 3 C C 4 C H 5 H 6 C C 6 CH OH 2 OH H OH OH OH 5 C H H OH 4 C OH 3 C H 6 CH OH 2 O H H H 2 C OH H 4 C 1 C O OH 5 C H OH 3 C H CH 2 OH O H H H 1 C 2 C OH OH 6 5 4 HO H OH 3 H O H 1 2 OH OH H Figure 5. 4 (a) Linear and ring forms. Chemical equilibrium between the linear and ring structures greatly favors the formation of rings. To form the glucose ring, carbon 1 bonds to the oxygen attached to carbon 5.

Glucose + Fructose = Sucrose
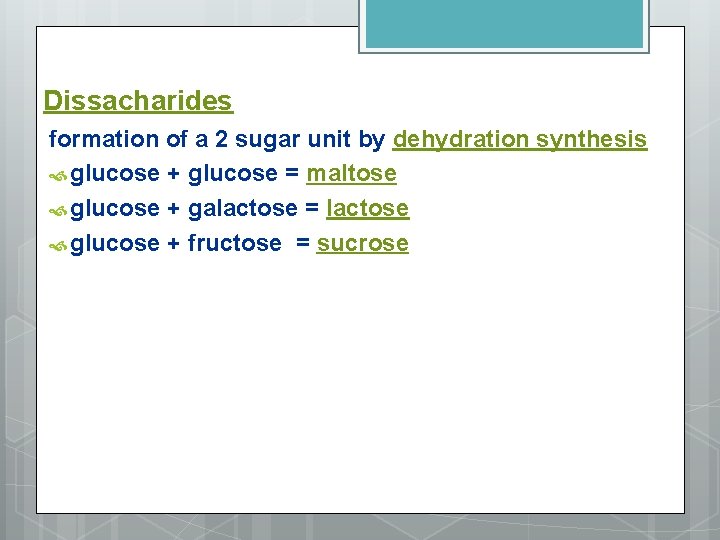
Dissacharides formation of a 2 sugar unit by dehydration synthesis glucose + glucose = maltose glucose + galactose = lactose glucose + fructose = sucrose

Formation of disaccharides Dehydration reaction (a) in the synthesis of maltose. The bonding CH OH CH 2 OH 2 of two glucose units O H forms maltose. The H H glycosidic link joins OH H the number 1 carbon OH OH HO of one glucose to the number 4 carbon of H OH the second glucose. H 2 O Joining the glucose Glucose monomers in a different way would result in a different CH 2 O disaccharide. H H H O H Dehydration reaction O H (b) in the synthesis of H H sucrose. Sucrose is. O O a disaccharide formed H H from glucose and fructose. Notice that fructose, Glucose though a hexose like glucose, forms a five-sided ring. Figure 5. 5 CH 2 OH O O H H HO CH 2 OH OH H CH 2 OH H H O H 1– 4 H H O H 1 glycosidic 4 O O H H linkage H H HO OH O H H OH Maltose CH 2 OH H H O HO H H CH 2 OH O 1– 2 glycosidic 2 1 linkage O H H O Sucrose H H HO CH 2 OH OH H 2 O Fructose OH

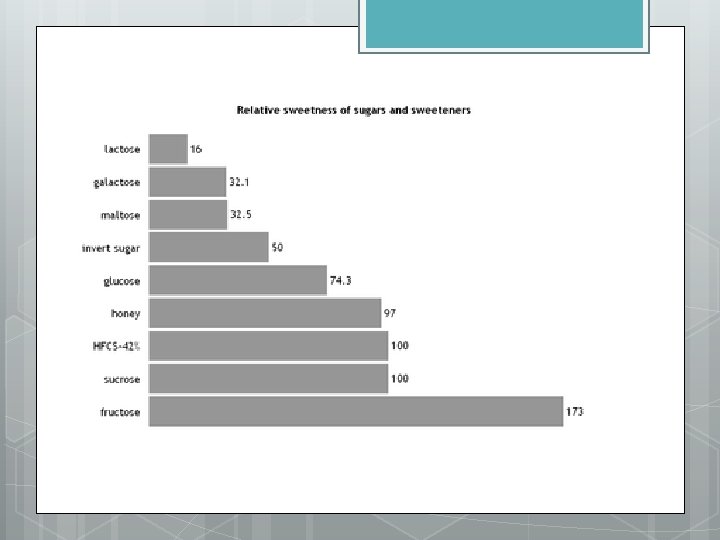
Sweetness of Sugars Why do plants alter glucose to fructose and sucrose?

Oligosaccharides Chains of 3 – 10 sugar units Importance: 1) Cell Surface Structures - Part of the Extracellular Matrix (ECM) - Bind to proteins = Glycoprotein - Bind to lipids = Glycolipids

2) Role of Glycoproteins - part of antibodies - part of mucus and saliva - H antigen (type O), the precursor to the A and B antigens of blood type - Hormones: FSH, LH, TSH, Human Chorionic Gonadotropin, Erythropoietin -Determine Protein Structure: - Pentasaccharides that are added cotranslationally (while a protein is being made) – affect the way a protein folds affecting its overall structure and function - Miraculin - a glycoprotein extracted from Synsepalum dulcificum a berry which alters human tongue receptors to recognize sour foods as sweet

3) Role of Glycolipids Cell to Cell Interactions Immune Response Leukocyte Extravasation Schematische Darstellung der Leukozytenmigration durch das Endothel. Nach einer Grafik von U. H. von Andrian und C. R. Mackay: T-Cell Function and Migration. In: NEJM. October 5 th, 2000, S. 1025. Und K. Ley et al. : Getting to the site of inflammation: the leukocyte adhesion cascade updated. In: Nature Reviews Immunology 7, 2007, S. 679

Polysaccharides: many sugar units Chains of glucose Type of polysaccharide dependent on the type of glucose alpha glucose beta glucose differ in orientation of the hydroxyl group on the number 1 st carbon alpha - down beta - up

Formation of alpha and beta glucose The alpha and beta forms of glucose differ only in the position of the hydroxyl on the first carbon and are NOT mirror images of each other = diastereomer.

Glucose will switch from the ring form to the linear form when in solution. When it reforms a ring it will either be the alpha form or the beta form. In biological systems (depending on what is being made), enzymes will direct the formation of the correct form.

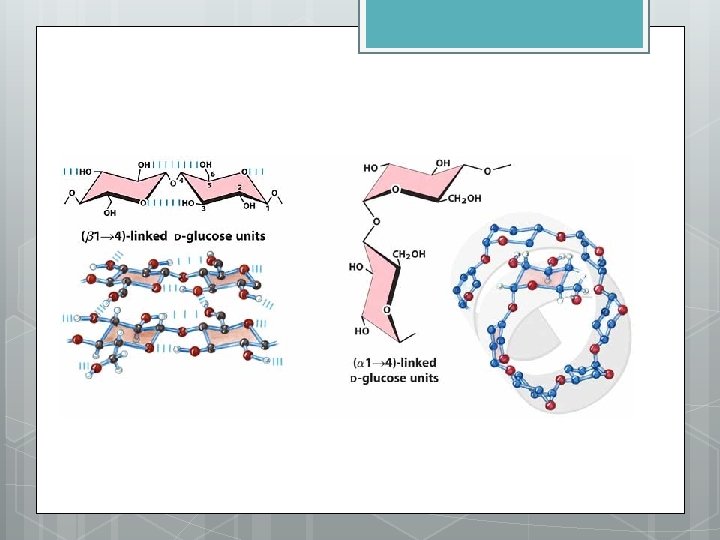
Importance of alpha and beta glucose diastereomers. H CH 2 OH O H H HO 4 H H H O O O H H C H C O H C glucose H C C H C O O H H O H O H CH 2 O H H O 4 1 H H O O H H glucose (a) and glucose ring structures Figure 5. 7 A–C CH 2 OH O O H CH 2 O H H O O 1 4 1 4 H O O O H H (b) Starch: 1– 4 linkage of glucose monomers O O CH 2 H H OH OH O O O O H 1 4 H H O O O CH 2 O O OH OH H H (c) Cellulose: 1– 4 linkage of glucose monomers CH 2 OH O O H 1 O O H


STORAGE POLYSACCHARIDES 1. Starch - storage in plants - as granules in organelles called plastids glucose monomers linked together making an alpha 1 -4 glucosidic linkages two forms of starch amalose - unbranched chains amylopectin - branched 2. Glycogen - storage in animals - storage in liver and muscle cells alpha 1 -4 linkage extensively branched – faster break down
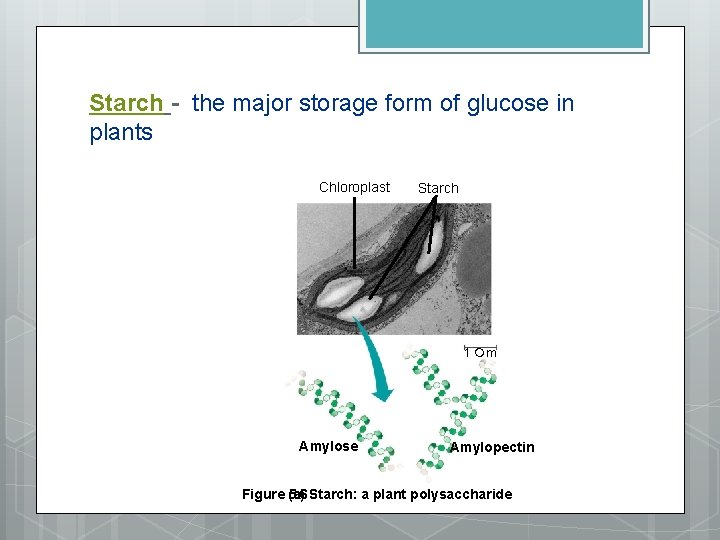
Starch - the major storage form of glucose in plants Chloroplast Starch 1 m Amylose Amylopectin Figure (a) 5. 6 Starch: a plant polysaccharide

Starch molecules in a bean

Glycogen major storage form of glucose in animals Mitochondria Giycogen granules 0. 5 m Glycogen Figure 5. 6 (b) Glycogen: an animal polysaccharide

Structural Polysaccharides provide protection and support 1. Cellulose - long unbranched, straight chains beta 1 -4 linkages makes for alternating bonds makes for a very rigid structure makes up cell walls enzymes that break alpha bonds can't break beta bonds
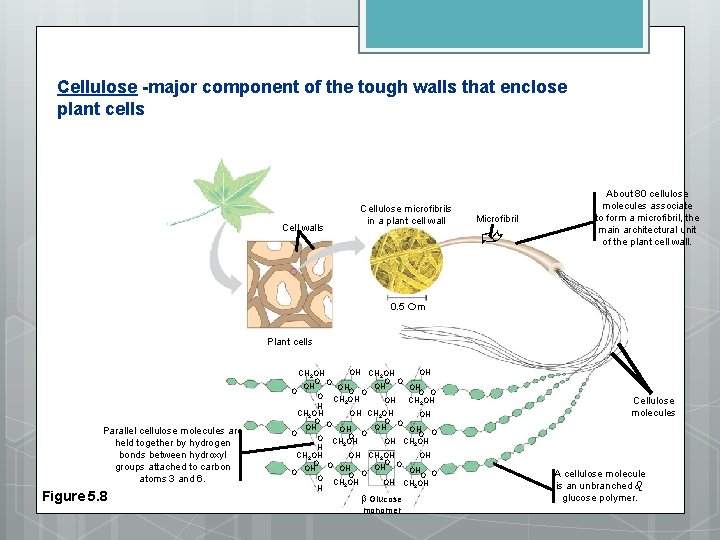
Cellulose -major component of the tough walls that enclose plant cells Microfibril Cell walls Cellulose microfibrils in a plant cell wall About 80 cellulose molecules associate to form a microfibril, the main architectural unit of the plant cell wall. 0. 5 m Plant cells Parallel cellulose molecules are held together by hydrogen bonds between hydroxyl groups attached to carbon atoms 3 and 6. Figure 5. 8 OH CH 2 OH O O OH OH O O O CH OH OH CH 2 2 OH H CH 2 OH OH O O O O CH OH OH CH 2 OH 2 H CH 2 OH OH OH CH 2 OH O OH OH OH O O O CH OH OH CH 2 OH 2 H b Glucose monomer Cellulose molecules A cellulose molecule is an unbranched glucose polymer.

Cellulose Cows is difficult to digest have microbes in their stomachs to facilitate this process – mutualism Figure 5. 9

Structural Polysaccharides 2. Chitin - structure of arthropod exoskeletons and cell walls of fungus differences: glucose with a nitrogen compound attached

Chitin, another important structural polysaccharide Is found in the exoskeleton of arthropods Can be used as surgical thread CH 2 O H O OH H H NH C O CH 3 (a) The structure of the(b) Chitin forms the exoskeleton of arthropods. This cicada chitin monomer. is molting, shedding its old exoskeleton and emerging Figure 5. 10 A–C in adult form. (c) Chitin is used to make a strong and flexible surgical thread that decomposes after the wound or incision heals.
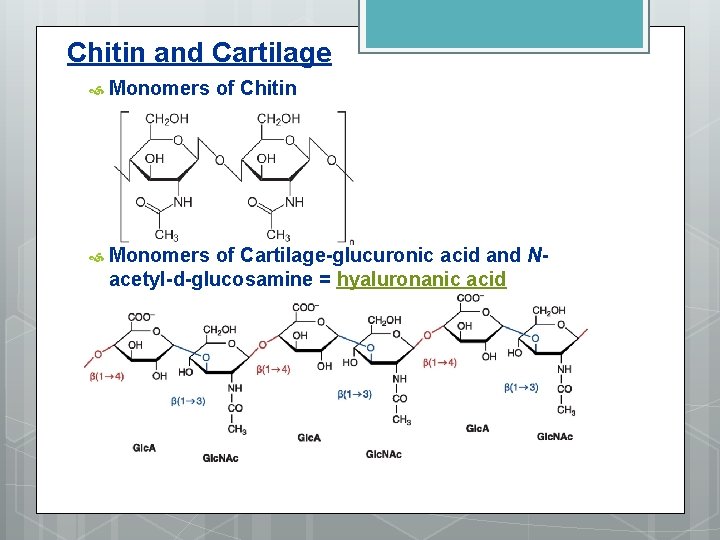
Chitin and Cartilage Monomers of Chitin Monomers of Cartilage-glucuronic acid and Nacetyl-d-glucosamine = hyaluronanic acid

Hyaluronanic Acid In humans, HA is abundant in the vitreous of the eye, the umbilical cord, synovial fluid (joints), heart valves, skin, and skeletal tissues (Dianhua Jiang, Jiurong Liang, Paul W. Noble, Physiological Reviews Published 1 January 2011 Vol. 91 no. 1, 221 -264 DOI: 10. 1152/physrev. 00052. 2009)
- Slides: 40