CARBOHYDRATES METABOLISM DISORDERS GLUCOSE METABOLISM the cornerstone of

CARBOHYDRATES METABOLISM DISORDERS

GLUCOSE METABOLISM Ø the cornerstone of life Ø neurons are especially dependent on glucose Ø regulatory mechanisms: l l hyperglycemic hormones = glycogenolysis, gluconeogenesis hypoglycemic hormone = insulin


HYPERGLICEMIA (diabetes mellitus) Ø Diabetes - Greek word = to siphon or to pass thru. Ø Mellitus - Latin word = sweet or honey. Ø group of chronic disorders Ø insulin deficiency ABSOLUTE/RELATIVE !!! also affects protein and fat metabolism

CLASSIFICATION Ø type 1 DM - autoimmune pancreatic β-cell destruction = absolute insulin deficiency; Ø type 2 DM - insulin resistance = relative insulin deficiency; Ø “other” specific types of DM (associated with identifiable clinical conditions or syndromes); Ø gestational DM - appears or is first detected during pregnancy.

!!! pre-diabetes Ø impaired glucose tolerance (IGT) Ø impaired fasting glucose (IFG)

ADA diagnosis of DM 1. classic symptoms of diabetes (polyuria, polydipsia, and unexplained weight loss) plus random plasma glucose concentration ≥ 200 mg/d. L (≥ 11. 1 mmol/L); or 2. fasting (≥ 8 -hour) plasma glucose concentration ≥ 126 mg/d. L (≥ 7. 0 mmol/L); or 3. a 2 -hour postload plasma glucose concentration ≥ 200 mg/d. L (≥ 11. 1 mmol/L) during a 75 -g oral glucose tolerance test.

ETIOLOGY Type 1 diabetes Ø Genetic Ø Environmental Ø Autoimmune
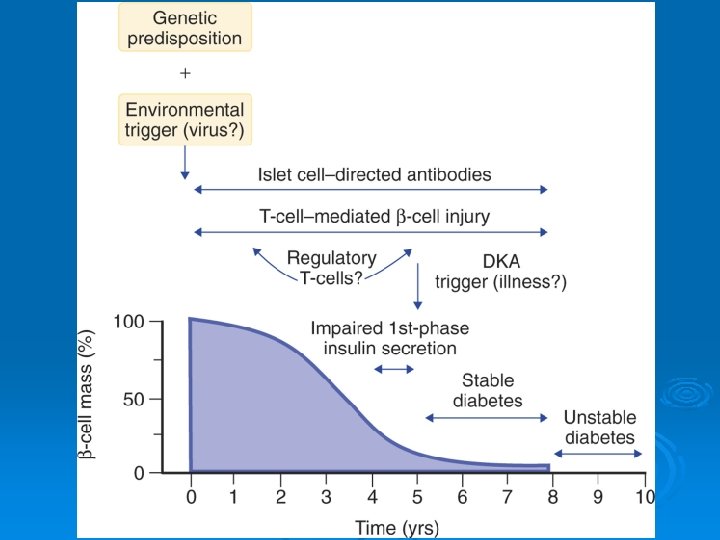

Type 2 diabetes = relative insulin deficiency – insulin resistance / inadequate secretory response Ø complex genetic interactions unrelated to HLA genes Ø environmental factors such as body weight (obesity) and exercise (lack of physical activity).

MODY Ø autosomal dominant inheritance Ø onset in at least 1 family member younger than 25 years Ø absence of autoantibodies Ø correction of fasting hyperglycemia without insulin for at least 2 years Ø absence of ketosis.

Type 2 DM pathogenic mechanisms: progressive loss of insulin secretory capacity. impaired insulin action : Ø Ø l l l impaired mitochondrial function and the resulting accumulation of free fatty acids in insulin-responsive tissues. defects of the insulin receptor. defects in “postreceptor” pathways Adipocyte-Derived Hormones and Cytokines Ø l l Leptin Adiponectin other adipocyte-derived factors (resistin, angiotensinogen, interleukin-6, transforming growth factor-β, plasminogen activator inhibitor 1) TNF-α.
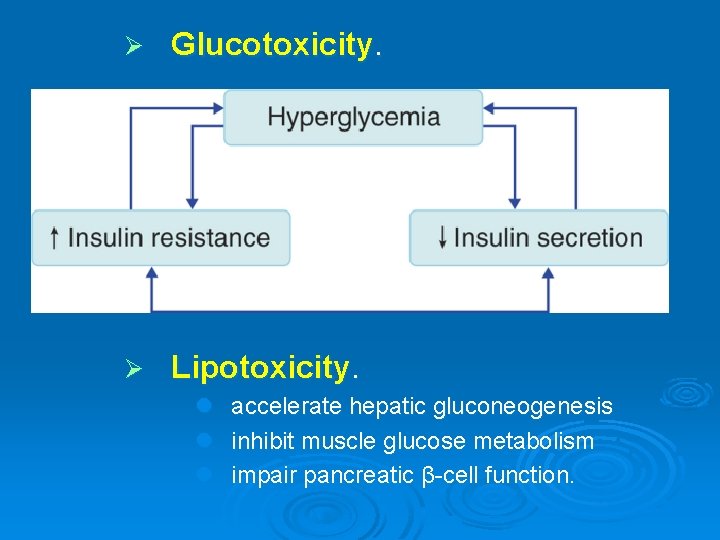
Ø Glucotoxicity. Ø Lipotoxicity. l l l accelerate hepatic gluconeogenesis inhibit muscle glucose metabolism impair pancreatic β-cell function.

Ø Type 1 DM produces profound β-cell failure and insulin deficiency with secondary insulin resistance, Ø Type 2 DM is associated with less severe insulin deficiency but greater insulin resistance.

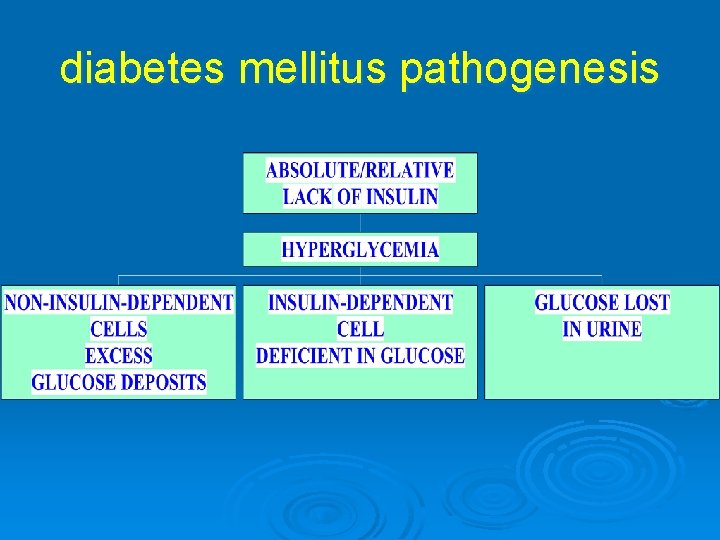
diabetes mellitus pathogenesis
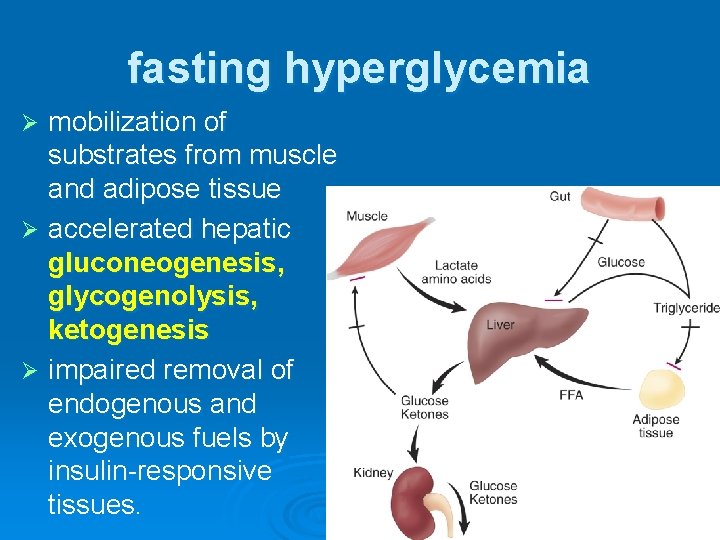
fasting hyperglycemia mobilization of substrates from muscle and adipose tissue Ø accelerated hepatic gluconeogenesis, glycogenolysis, ketogenesis Ø impaired removal of endogenous and exogenous fuels by insulin-responsive tissues. Ø

fasting free fatty acids Ø Ø Ø Insuline deficiency - increase lipolysis Glucagon - accelerating hepatic ketogenesis Catecholamines growth hormone, and cortisol - increase lipolysis. l l type 1 diabetes - converted to ketone bodies type 2 diabetes – insulin suppress the conversion of free fatty acids to ketones !!! The increase in substrate delivery - hepatic steatosis and severe hypertriglyceridemia (endogenous).

Postprandial Hyperglycemia Ø type 1 diabetes – insulin deficiency Ø type 2 diabetes - delayed insulin secretion + hepatic insulin resistance l the liver fails to arrest glucose production l fails to appropriately take up glucose for storage as glycogen l glucose uptake by peripheral tissues is impaired

Hyperglycaemia renal threshold for glucose surpassed (>170 mg/dl) GLUCOSURIA osmotic diuresis POLYURIA dehydration thirst POLYDIPSIA

Ø Type 1 diabetic - defects in the disposal of ingested proteins and fats as well. l Hyperaminoacidemia l Hypertriglyceridemia (exogenous)

ACUTE METABOLIC COMPLICATIONS Ø diabetic ketoacidosis (DKA) Ø hyperosmolar hyperglycemic syndrome (HHS) Ø hypoglycemia

DKA Ø deficient circulating insulin activity Ø excessive secretion of counter- regulatory hormones. Ø hyperglycemia, ketosis, acidosis !!! osmotic diuresis - dehydration and electrolyte loss.

Hyperosmolar Hyperglycemic Syndrome (HHS) Ø patients cannot drink enough liquid to keep pace with a vigorous osmotic diuresis. l l Severe hyperosmolarity (>320 m. Osm/L) Severe hyperglycemia (>600 mg/d. L). Ø severe acidosis and ketosis are generally absent in the HHS!!!

Hypoglycemia the earliest subjective warning signs = autonomic symptoms (sweating, tremor, palpitations) Ø Central nervous system symptoms and signs = neuroglycopenia: Ø l l l Ø nonspecific (e. g. , fatigue or weakness) more clearly neurologic (e. g. , double vision, oral paresthesias, slurring of speech, apraxia, personality change, or behavioral disturbances). irreversible brain damage. Hypoglycemic unawareness syndrome l l l duration of diabetes autonomic neuropathy switched to intensive insulin regimens.

Ø Somogyi phenomenon – 1. 2. 3. normal or increased blood glucose levels at bedtime blood glucose drops in early morning hours (2 to 3 A. M. ) usually because nighttime insulin dose is too high. compensate by producing counterregulatory hormones resulting in hyperglycemia on awakening. Dawn phenomenon = Decrease in the tissue sensitivity to insulin between 5 and 8 A. M. - prebreakfast hyperglycemia ? ? ? release of nocturnal growth hormone Ø

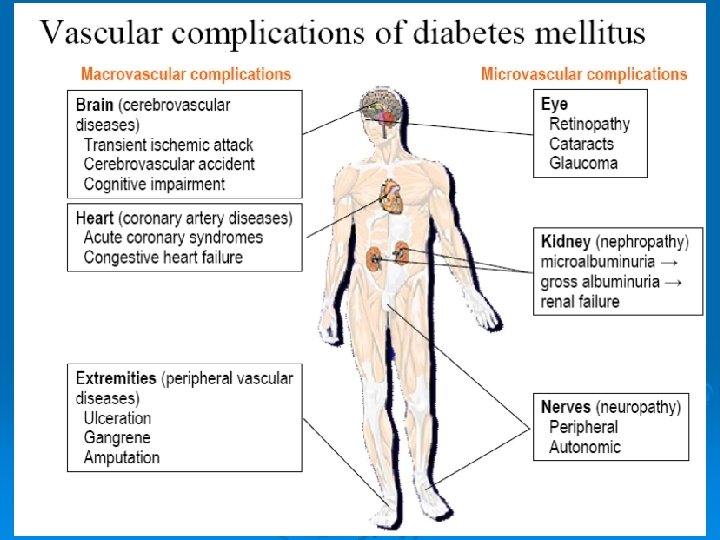
CHRONIC DIABETIC COMPLICATIONS MICROVASCULAR AND NEUROPATHIC COMPLICATIONS Ø Intracellular glucose l l Ø advanced glycation end products (AGEs) accelerated polyol pathway reactive oxygen species Others: cytokines, angiotensin II, endothelin, growth factor stimulation, depletion of basement membrane glycosaminoglycans Hemodynamic changes in the microcirculation

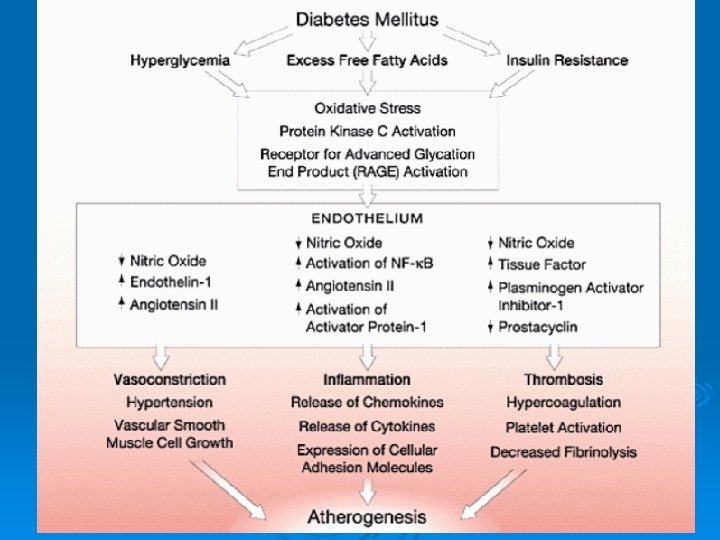
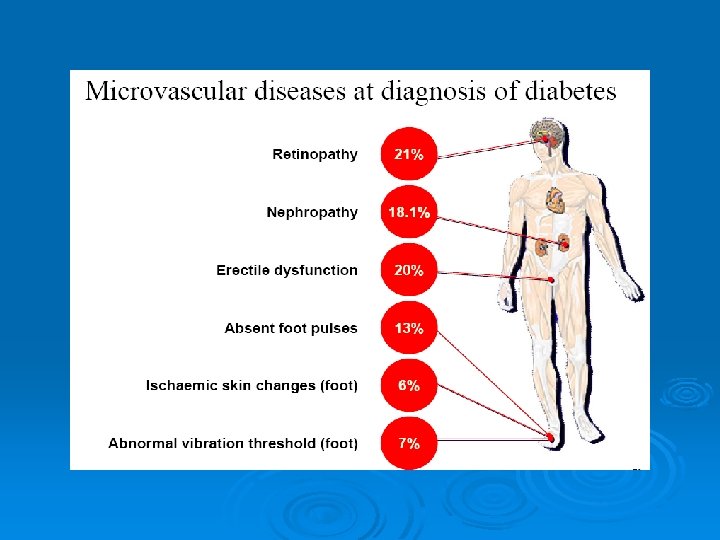

Diabetic retinopathy Ø vascular-neuroinflammatory disease. breakdown of the blood-retinal barrier (BRB) function and loss of retinal neurons. l activated macroglia and neuronal death. l activated microglia exacerbate the damage. l



Diabetic Nephropathy rise in glomerular filtration rate. Ø glomerular lesions Ø increased glomerular permeability. Ø microalbuminuria (30 to 300 mg/day) Ø diffuse glomerulosclerosis Ø massive proteinuria - nephrotic syndrome Ø Systemic hypertension Ø progression to ESRD. Ø

Diabetic Neuropathy Ø metabolic factors Ø vascular Ø Nerve growth factor diminished Ø Autoimmune mechanisms.

Ø Distal symmetrical (sensorimotor) polyneuropathy Ø Acute sensory neuropathy Ø Focal diabetic neuropathies (mononeuropathies) – pain Ø Entrapment syndromes Ø Proximal motor neuropathy amyotrophy) (diabetic

Autonomic neuropathy Ø Cardiovascular abnormalities l l preferential dysfunction of parasympathetic fibers impaired sympathetic vasoconstrictor response and impaired cardiac reflexes. Ø Altered gastrointestinal function l l hypermotility / hypomotility Gastroparesis

Ø Genitourinary alterations l l bladder hypotonia Erectile dysfunction Ø Abnormal sweat production l l l Xerosis. Distal anhidrosis - truncal-facial sweating Generalized anhidrosis


atherosclerosis Ø lipid abnormalities Ø procoagulant state = accentuated platelet aggregation and adhesion, endothelial cell dysfunction. Ø hyperinsulinemia
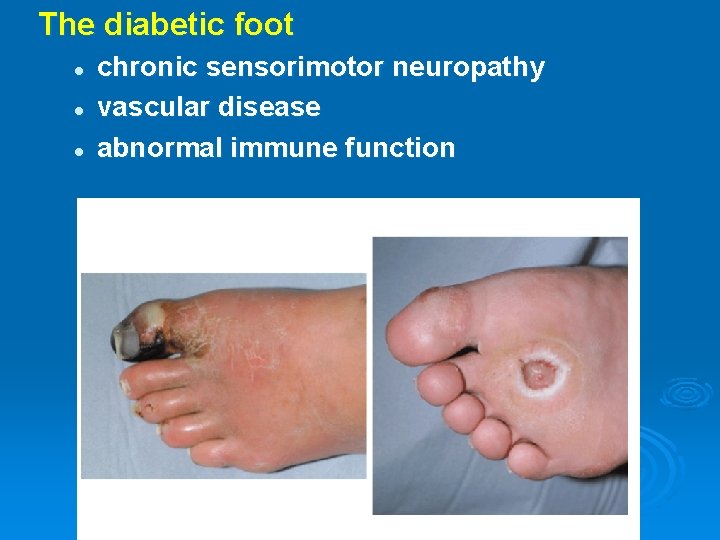
The diabetic foot l l l chronic sensorimotor neuropathy vascular disease abnormal immune function

HYPOGLICEMIA Ø Physiological l hypoglycaemia 3 -5 hours after ingestion of glucose or during prolonged fast Ø Pathological HYPOGLICEMIA Whipple’s triad: l l l LOW BLOOD GLUCOSE below 50 mg/dl symptoms of hypoglycaemia symptoms relieved by glucose


Classification: Ø Fasting l l hypoglycaemia With hyperinsulinemia Without hyperinsulinemia Ø Non-fasting, postprandial or reactive hypoglycaemia

Fasting hypoglycemia with hyperinsulinemia Ø diabetes Ø islet cell tumours Ø factitious hypoglycemia Ø autoimmune hypoglycaemia Ø drugs

Fasting hypoglycemia without hyperinsulinemia Ø Chronic renal impairment l l Decreased renal gluconeogenesis impaired hepatic glycogenolysis and gluconeogenesis !!! l l increased insulin half-life due to decreased renal degradation exaggerated glucose-induces insulin secretion

Øsevere liver disease = hepatogenous hypoglycaemia Ø deficient caloric intake and exerciseinduced hypoglycaemia

septicaemia early phase - hyperglycemia • decrease in insulin-stimulated phosphorylation of insulin receptor • increased clearance of insulin • increased production of corticosteroids. late phase – hypoglycemia • cytokines from macrophages stimulates insulin secretion • direct hypoglycemic effect of endotoxins (inhibit gluconeogenesis) • association of renal failure.

non-islet cell tumours: Ø l l l Increased uptake of glucose to tumors reduced production of glucose reduced gluconeogenesis due to weight loss produce peptides with insulin-like activity cytokines release ? (IGF-2, TNF )

drugs : Ø l l Salicylates non-selective beta-blockers endocrine insufficiency Ø l l l hypopituitarism Addison’s disease isolate GH or ACTH deficiency

Reactive hypoglycaemia Organic causes may lead to rapid emptying of gastric contents Ø Type 2 diabetes mellitus Ø Alcohol Ø l l potentates the hypoglycaemic effect of insulin potentates the insulin-stimulating effect of glucose Idiopathic Ø Inborn errors of metabolism Ø l l l Disorders of carbohydrates metabolism (galactosemia, hereditary fructose intolerance…. ) Disorders of amino acid metabolism (maple syrup urine disease…. ) Disorders of fatty acid metabolism (systemic carnitine deficiency…. )

- Slides: 54