CARBOHYDRATES Made of C O H empirical formula


CARBOHYDRATES • Made of C, O, H – empirical formula: (CH 2 O)n or Cn. H 2 n. On • Functions: – Quick energy source in all organisms – Structural role (i. e. cellulose) – Cell-to-cell recognition • Monomers = monosaccharides • Polymers = polysaccharides

• “simple carbohydrates” = SUGARS! • “complex carbohydrates” = whole grains, potatoes, beans, etc.
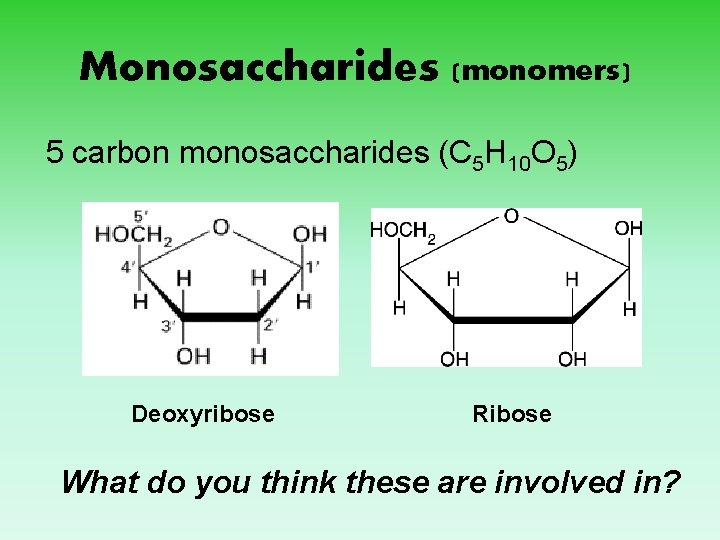
Monosaccharides (monomers) 5 carbon monosaccharides (C 5 H 10 O 5) Deoxyribose Ribose What do you think these are involved in?
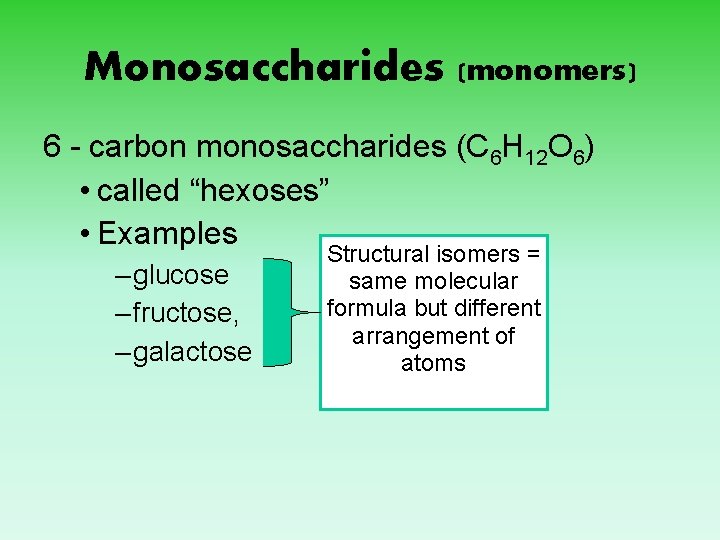
Monosaccharides (monomers) 6 - carbon monosaccharides (C 6 H 12 O 6) • called “hexoses” • Examples – glucose – fructose, – galactose Structural isomers = same molecular formula but different arrangement of atoms
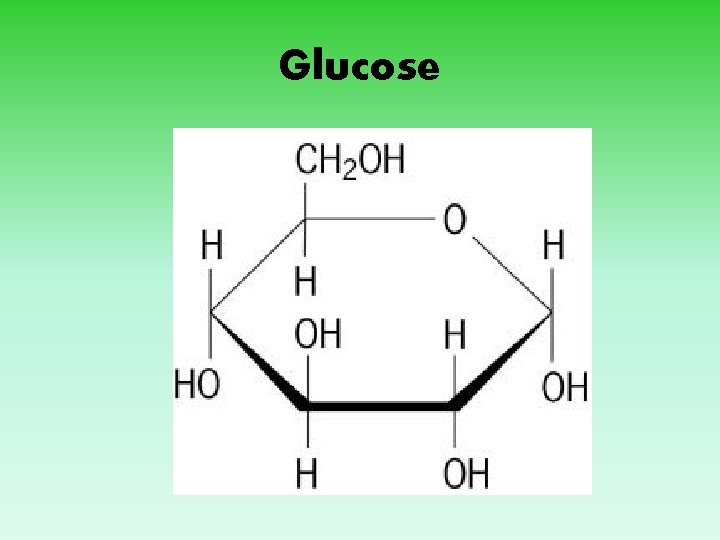
Glucose
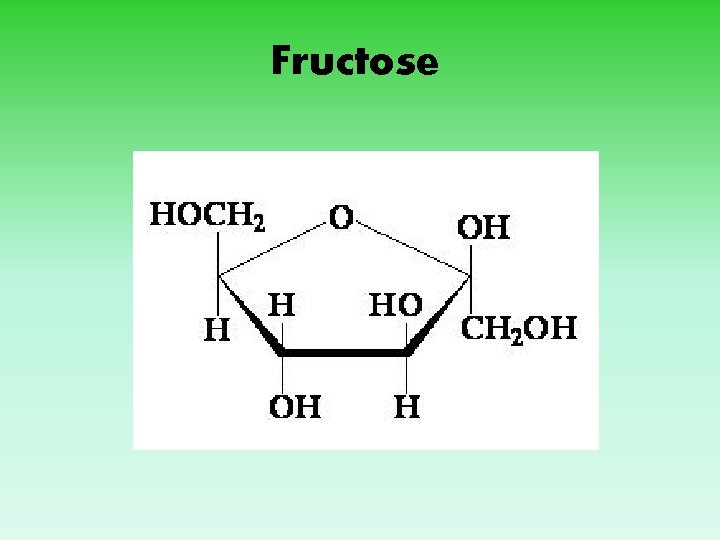
Fructose
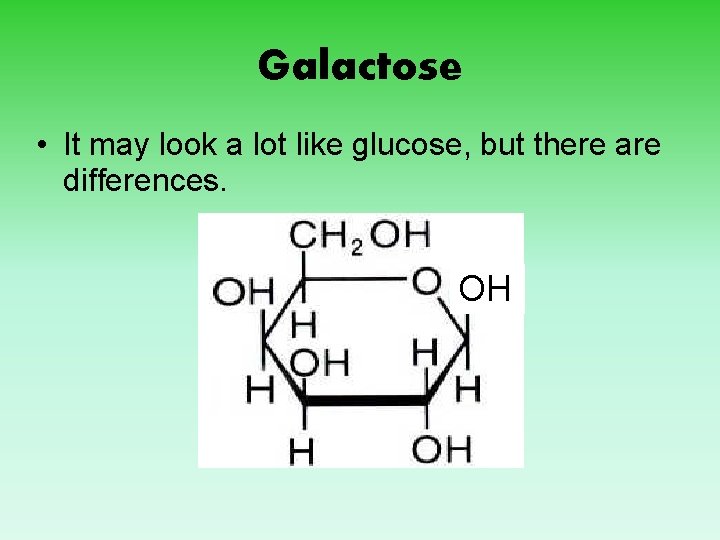
Galactose • It may look a lot like glucose, but there are differences. OH
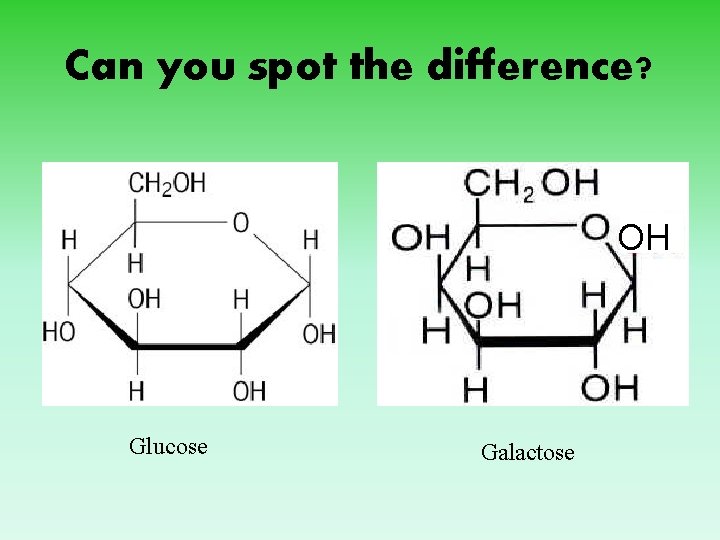
Can you spot the difference? OH Glucose Galactose
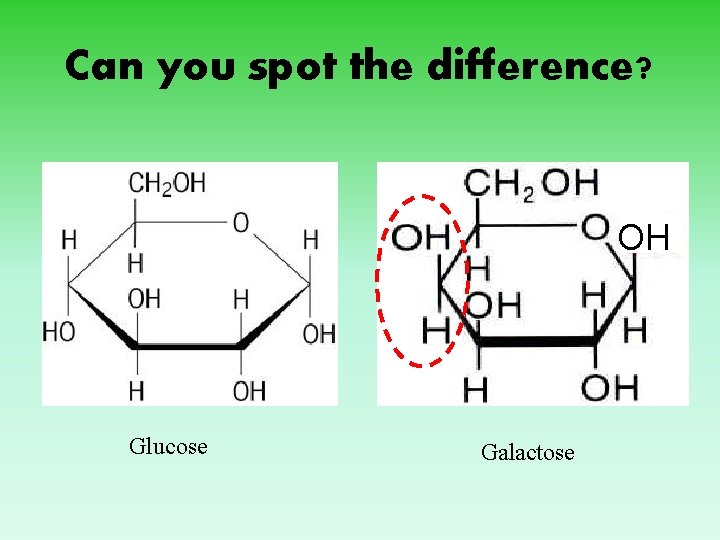
Can you spot the difference? OH Glucose Galactose

Making Disaccharides • 2 monomers join via _______, results in a disaccharide. • Bonds are called glycosidic bonds. • Famous disaccarides include: 1. glucose + glucose = maltose 2. glucose + fructose = sucrose 3. glucose + galactose = lactose
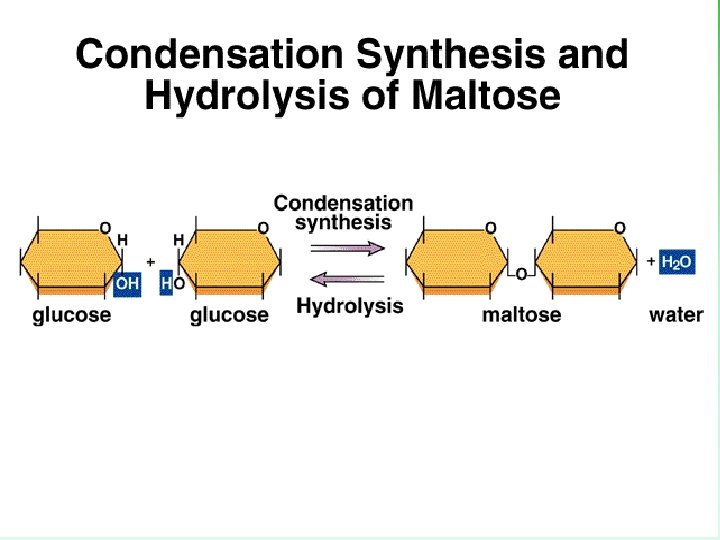
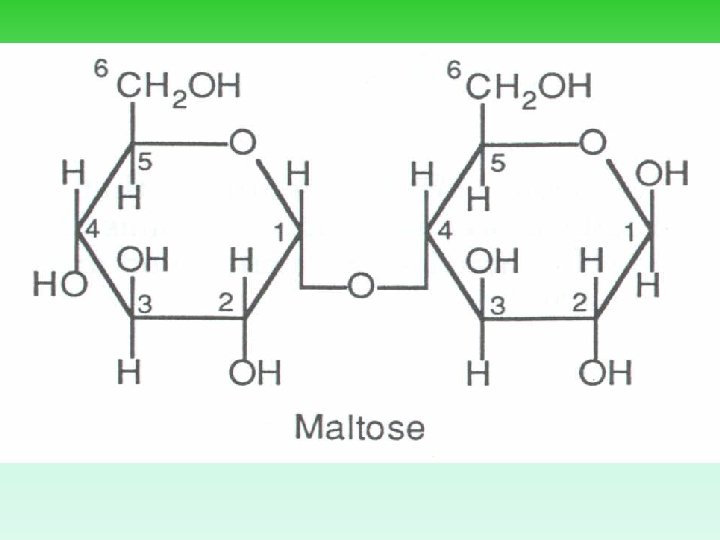
Maltose - A Second Look
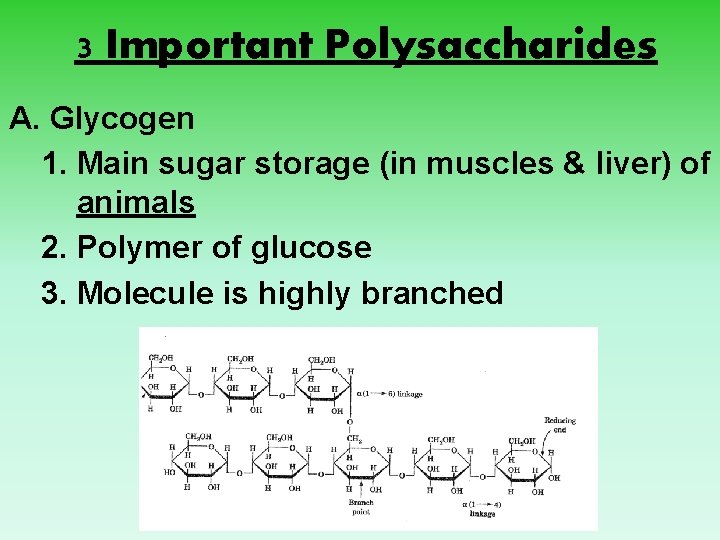
3 Important Polysaccharides A. Glycogen 1. Main sugar storage (in muscles & liver) of animals 2. Polymer of glucose 3. Molecule is highly branched

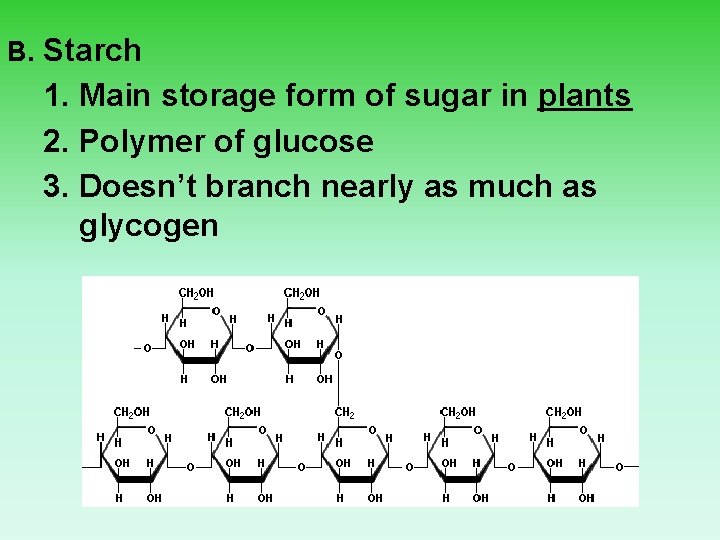
B. Starch 1. Main storage form of sugar in plants 2. Polymer of glucose 3. Doesn’t branch nearly as much as glycogen
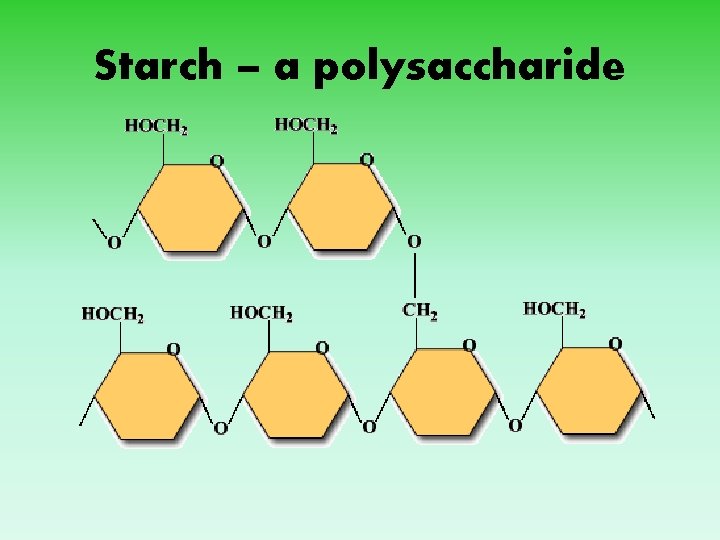
Starch – a polysaccharide
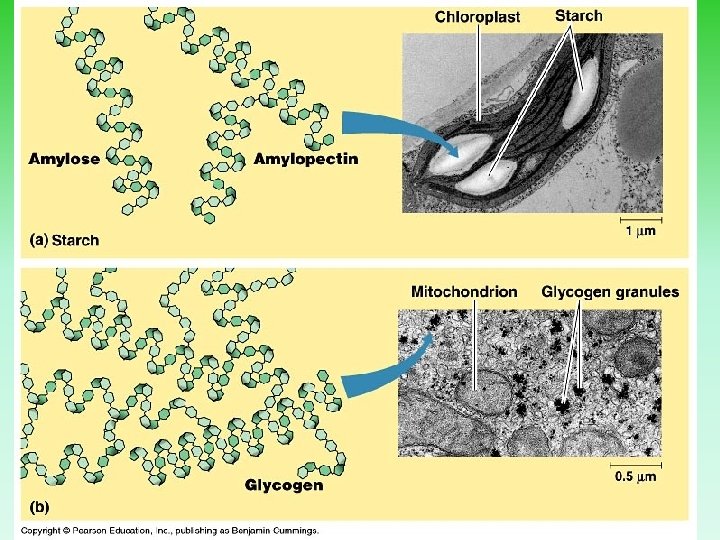

C. Cellulose 1. Structural support in plants 2. Long chains 3. Linkage between carbon atoms of adjacent sugar chains is different 4. No mammal can break this bond so is therefore indigestible 5. Most prevalent polysaccharide on Earth 6. Molecule is unbranched
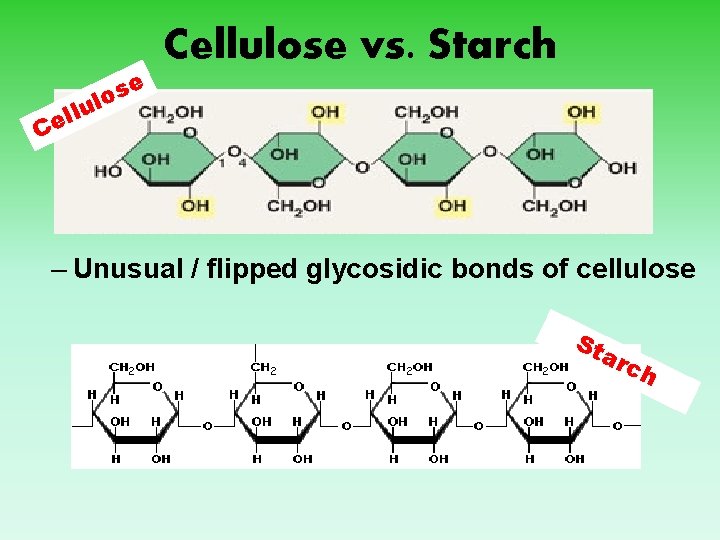
e Cellulose vs. Starch s o l u ll e C – Unusual / flipped glycosidic bonds of cellulose St arc h

Why Fibre?
- Slides: 21