CARBOHYDRATES Living things use carbohydrates as a key

CARBOHYDRATES • Living things use carbohydrates as a key source of ENERGY! • Plants use carbohydrates for structure (CELLULOSE) – include sugars and complex carbohydrates (starches) – contain the elements carbon, hydrogen, and oxygen (the hydrogen is in a 2: 1 ratio to oxygen)
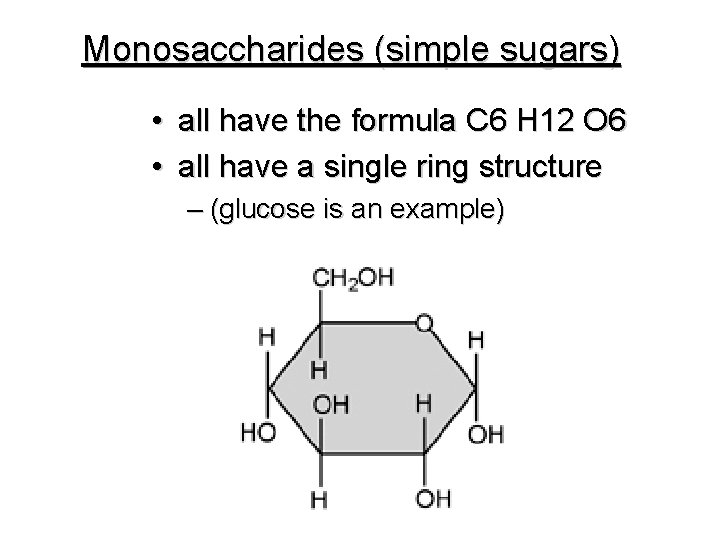
Monosaccharides (simple sugars) • all have the formula C 6 H 12 O 6 • all have a single ring structure – (glucose is an example)
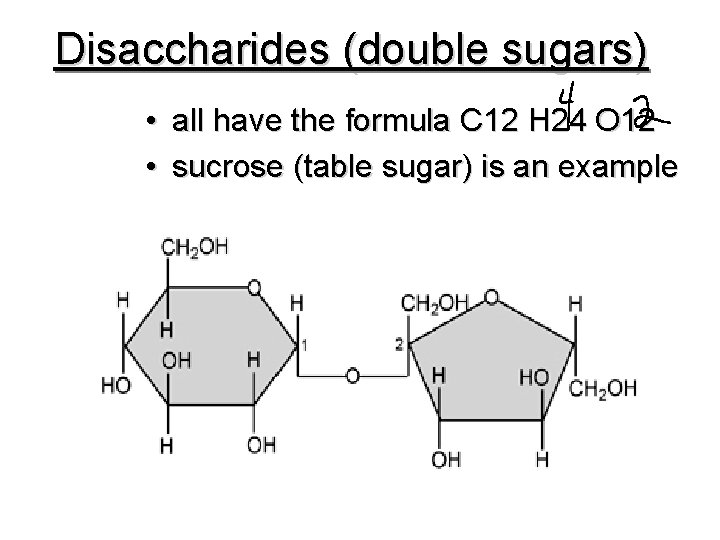
Disaccharides (double sugars) • all have the formula C 12 H 24 O 12 • sucrose (table sugar) is an example
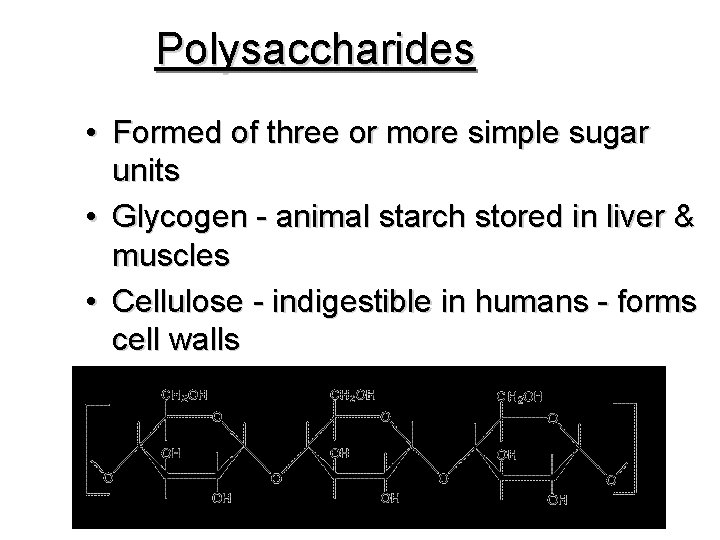
Polysaccharides • Formed of three or more simple sugar units • Glycogen - animal starch stored in liver & muscles • Cellulose - indigestible in humans - forms cell walls • Starches - used as energy storage

Lipids (Fats) • Fats, oils, waxes, steroids • Chiefly function in energy storage, protection, and insulation • Tend to be large molecules -- an example of a neutral lipid is below

• Neutral lipids are formed from the union of one glycerol molecule and 3 fatty acids • 3 fatty acids + glycerol ----> neutral fat (lipid) • Fats -- found chiefly in animals • Oils and waxes -- found chiefly in plants • Oils are liquid at room temperature, waxes are solids • Lipids along with proteins are key components of cell membranes • Steroids are special lipids used to build many reproductive hormones and cholesterol

Major Protein Functions • contain the elements carbon, hydrogen, oxygen, and nitrogen • composed of MANY amino acid subunits • Growth and repair • Support, movement, structure • Buffer -- helps keep body p. H constant • Cell communication • Carrying out chemical reactions

Polypeptide (protein) • composed of three or more amino acids linked by synthesis reactions • Examples of proteins include insulin, hemoglobin, and enzymes. • ** There an extremely large number of different proteins. • The bases for variability include differences in the number, kinds and sequences of amino acids in the proteins

NUCLEIC ACIDS • • in all cells composed of NUCLEOTIDES store & transmit heredity/genetic information Nucleotides consist of 3 parts: 1. 5 -Carbon Sugar 2. Phosphate Group 3. Nitrogenous Base
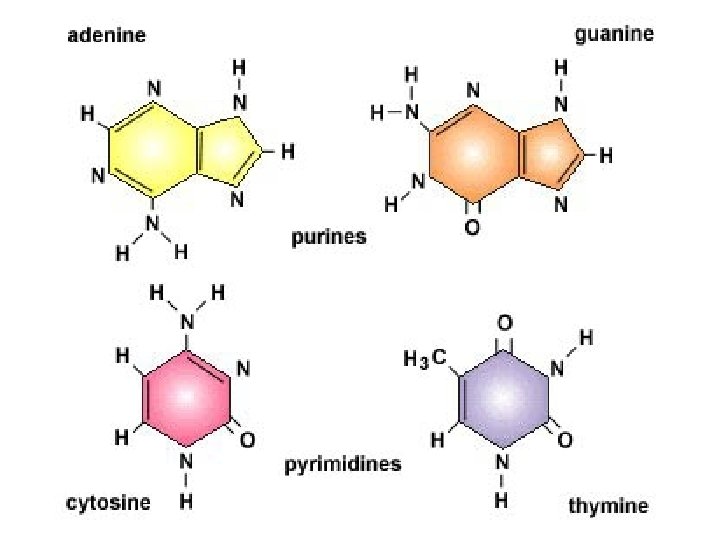
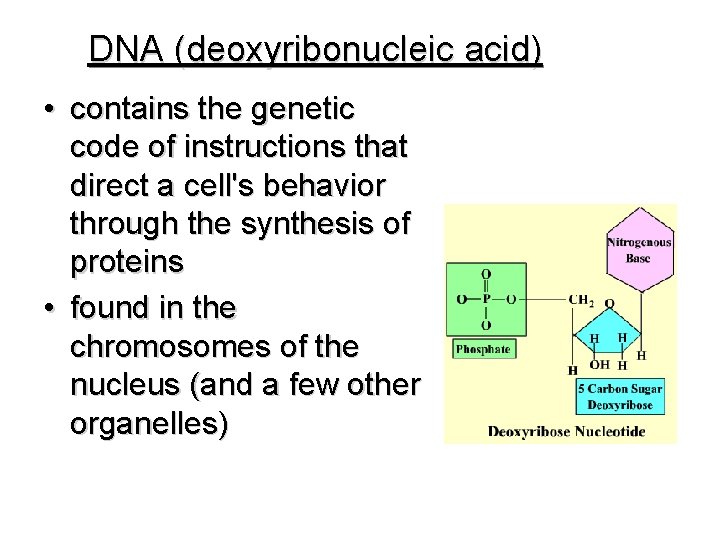
DNA (deoxyribonucleic acid) • contains the genetic code of instructions that direct a cell's behavior through the synthesis of proteins • found in the chromosomes of the nucleus (and a few other organelles)
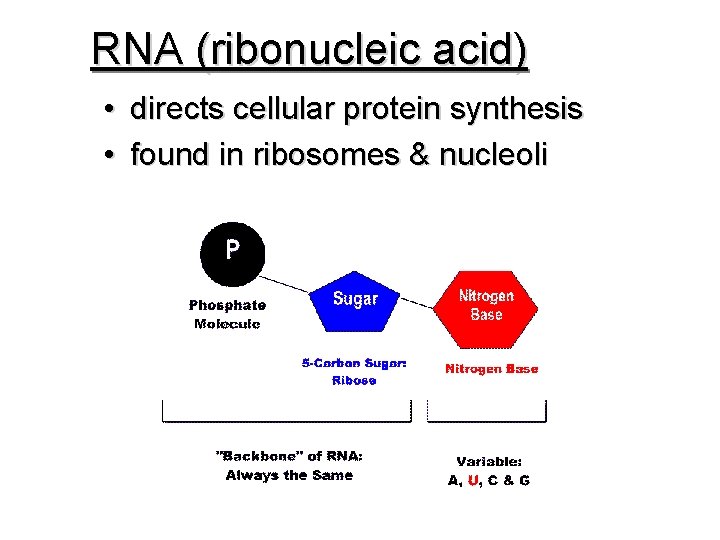
RNA (ribonucleic acid) • directs cellular protein synthesis • found in ribosomes & nucleoli

CHEMICAL REACTIONS • a process that changes one set of chemicals into another set of chemicals • REACTANTS – elements or compounds that enter into a chemical reaction • PRODUCTS – elements or compounds that are produced in a chemical reaction • Chemical reactions always involve the breaking of bonds in reactants and the formation of new bonds in products.

• In a reaction, energy is either TAKEN IN (ENDOTHERMIC) or GIVEN OFF (EXOTHERMIC) • Can you think of an everyday example of each type of reaction?

Enzymes

Facts about Enzymes • • • All enzymes are proteins Enzymes are specific Enzymes are organic compounds Organic Catalysts control the rate of chemical reactions

Enzyme Names • end in –ase • named for the molecules they act upon – Examples: maltase acts upon maltose lipase acts upon lipids protease acts upon proteins
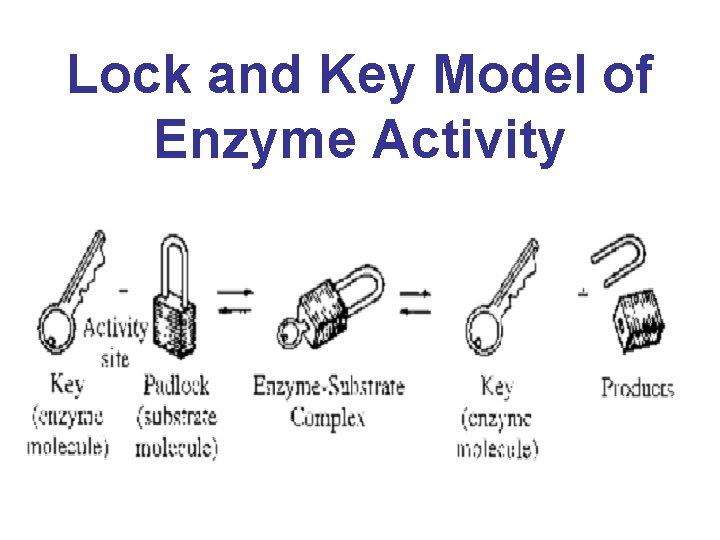
Lock and Key Model of Enzyme Activity
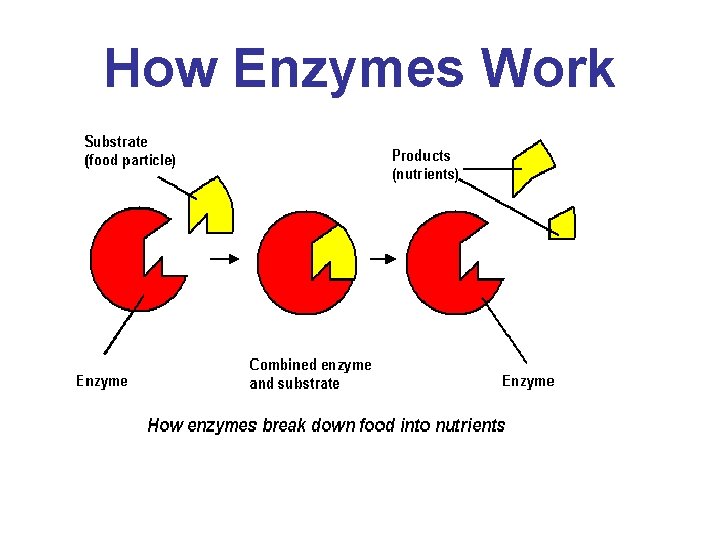
How Enzymes Work

• The enzyme has a unique 3 -D shape that only allows molecules that are the correct size and shape to make close contact • The region within the enzyme where the substrate makes close contact is called the active site
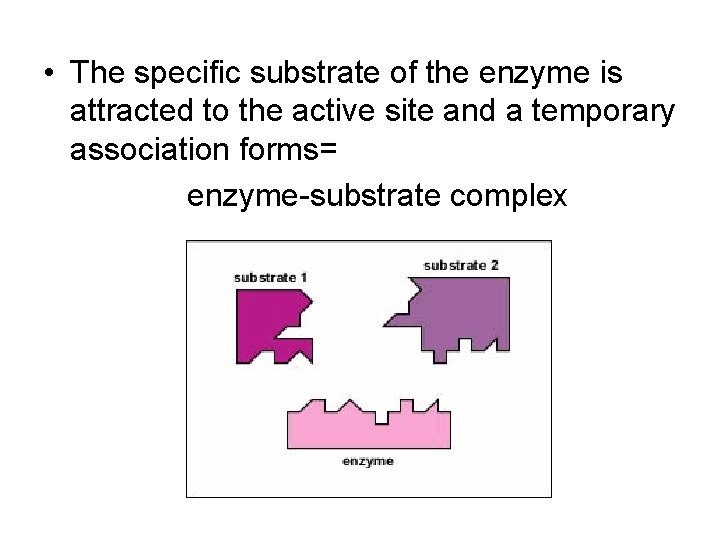
• The specific substrate of the enzyme is attracted to the active site and a temporary association forms= enzyme-substrate complex

Factors Affecting Enzyme Activity

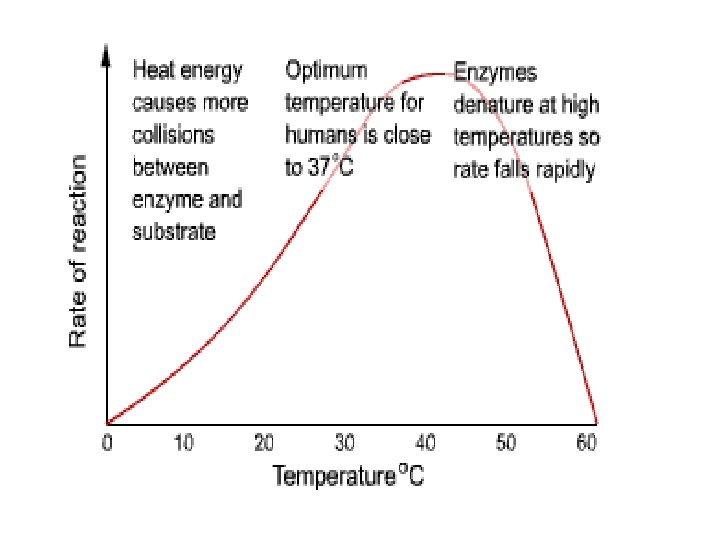
1. Temperature • As temperature increases, enzyme activity increases • Molecules move faster and enzymes come into contact more often with substrate molecules • At very high temperatures, the enzymes unfold and the active sites no longer fit substrate molecules • For most human enzymes, denaturation (breakdown) occurs at 40 o. C • Body temperature = 37 o. C (optimum temperature for enzyme activity)

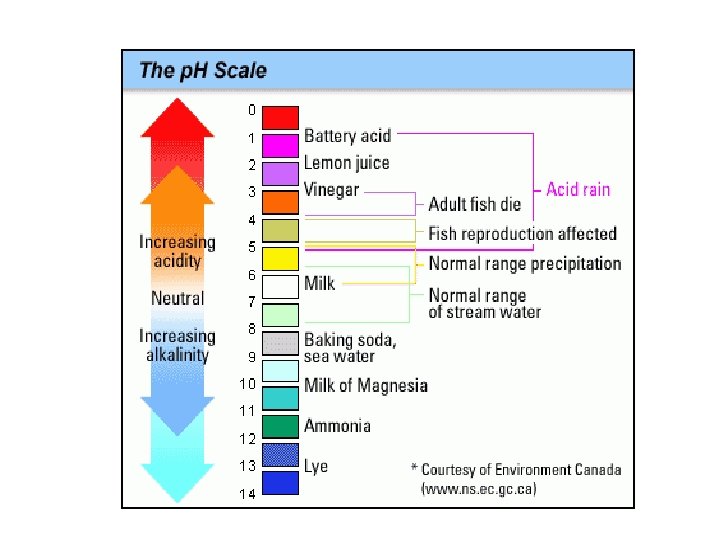
4. p. H • Each enzyme has an optimal p. H in which it can function. • The p. H scale measures how acidic or basic a substance is. • The p. H scale ranges from 0 -14. • A p. H of 7 is neutral, les than 7 is acidic, more than 7 is basic. • Enzymes have different shapes at different p. H values • Each enzyme works best within a narrow p. H range
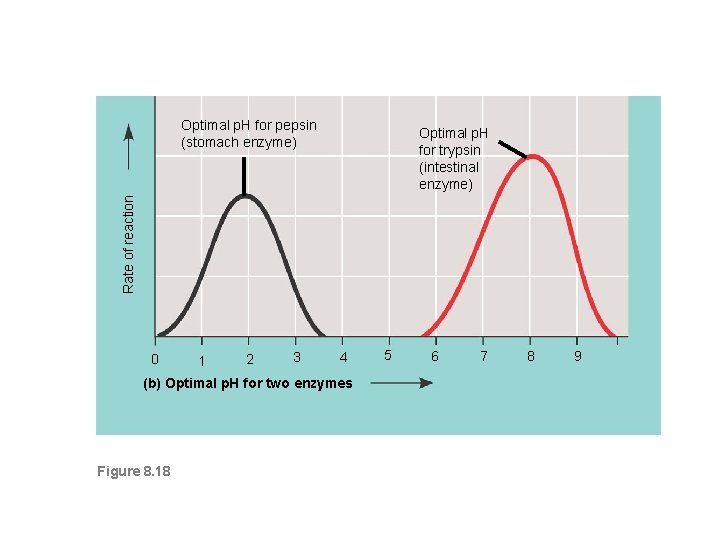
Optimal p. H for pepsin (stomach enzyme) Rate of reaction Optimal p. H for trypsin (intestinal enzyme) 0 1 2 3 4 (b) Optimal p. H for two enzymes Figure 8. 18 5 6 7 8 9
- Slides: 28