Carbohydrates Dr Mamoun Ahram Nursing Summer semester 2016

Carbohydrates Dr. Mamoun Ahram Nursing Summer semester, 2016

What are they? Carbohydrates are polyhydroxy aldehydes or ketones The term “carbohydrate” comes from the fact that when you heat sugars, you get carbon and water Saccharide is another name for a carbohydrate

Classifications Monosaccharides contain a single unit Glucose, fructose Disaccharides consist of two monosaccharide units Sucrose Oligosaccharides contain from 3 to 10 units Raffinose Polysaccharides contain very long chains of hundreds or thousands of monosaccharide units Cellulose, glycogen, starch
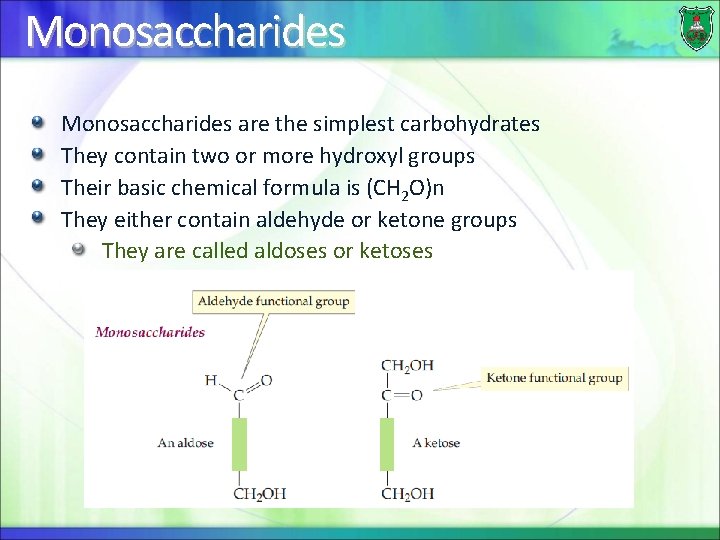
Monosaccharides are the simplest carbohydrates They contain two or more hydroxyl groups Their basic chemical formula is (CH 2 O)n They either contain aldehyde or ketone groups They are called aldoses or ketoses
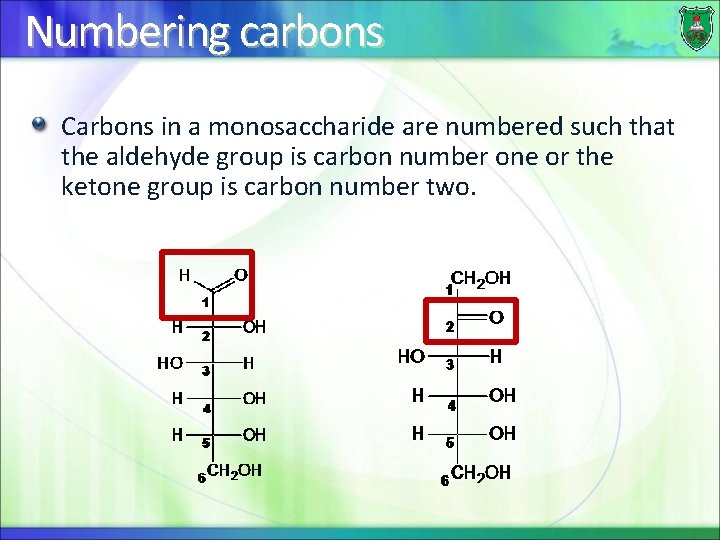
Numbering carbons Carbons in a monosaccharide are numbered such that the aldehyde group is carbon number one or the ketone group is carbon number two.
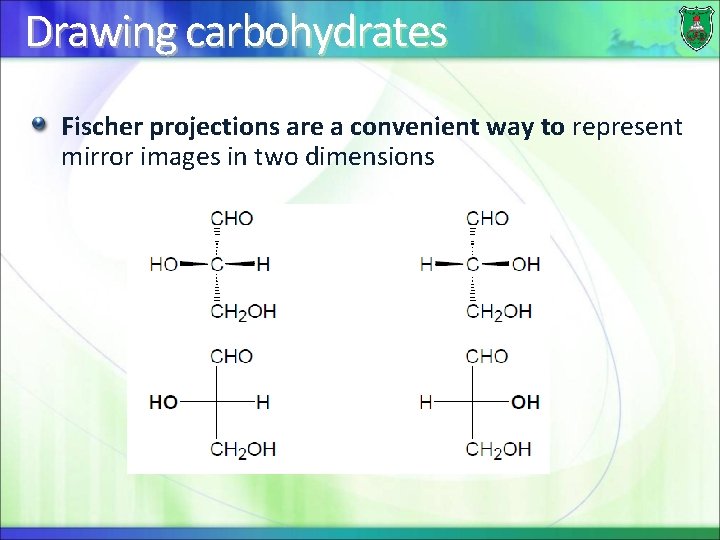
Drawing carbohydrates Fischer projections are a convenient way to represent mirror images in two dimensions

Another classification They can be classified by the number of carbon atoms they contain. The smallest monosaccharides contain three carbons (n = 3). They are referred to as trioses Four, five, six, and seven carbon atoms are called tetroses, pentoses, hexoses, and heptoses, respectively.
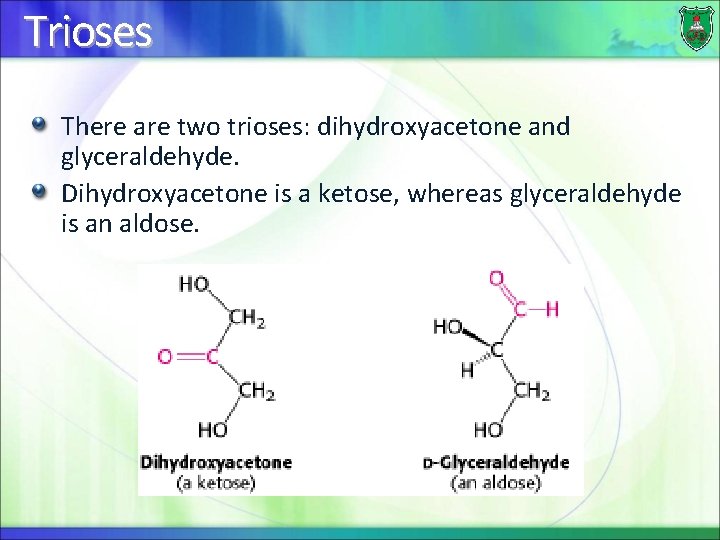
Trioses There are two trioses: dihydroxyacetone and glyceraldehyde. Dihydroxyacetone is a ketose, whereas glyceraldehyde is an aldose.
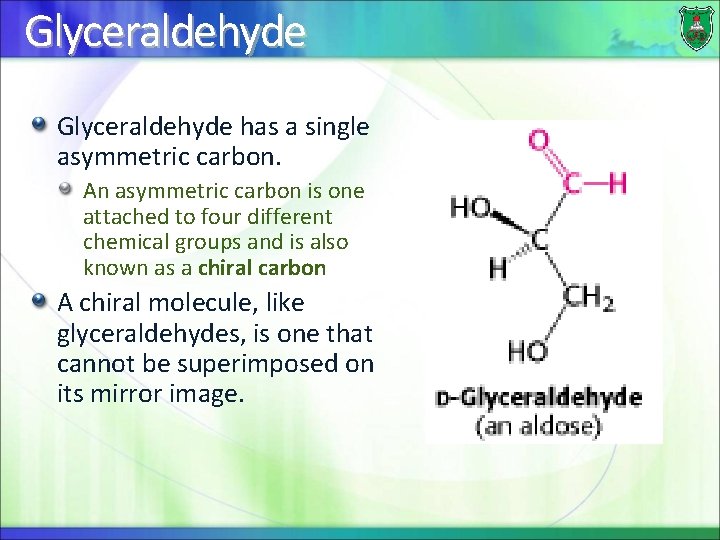
Glyceraldehyde has a single asymmetric carbon. An asymmetric carbon is one attached to four different chemical groups and is also known as a chiral carbon A chiral molecule, like glyceraldehydes, is one that cannot be superimposed on its mirror image.

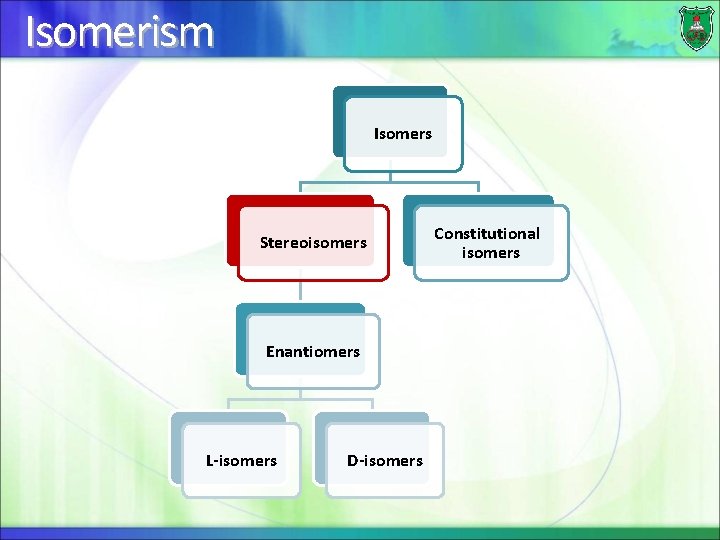
Isomerism Isomers Stereoisomers Enantiomers L-isomers D-isomers Constitutional isomers

Stereoisomers The two forms of glyceraldehydes are known as stereoisomers. Stereoisomers are molecules that have the same molecular formula and atomic connectivity, but have a different arrangement of the atoms in space.
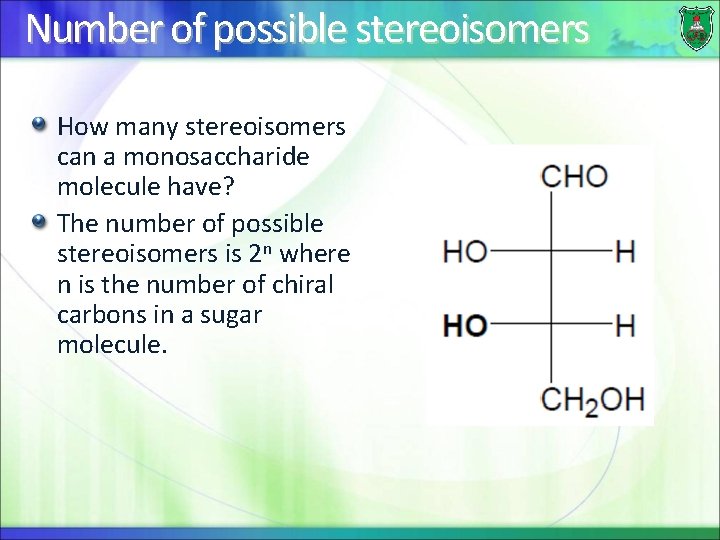
Number of possible stereoisomers How many stereoisomers can a monosaccharide molecule have? The number of possible stereoisomers is 2 n where n is the number of chiral carbons in a sugar molecule.
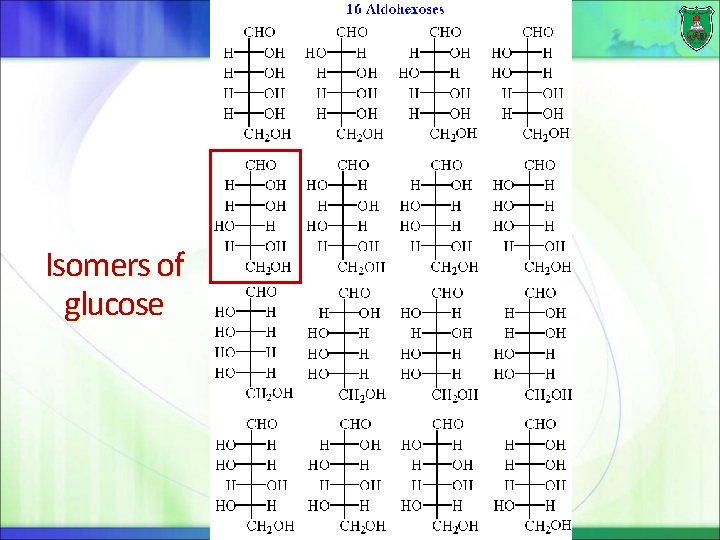
Isomers of glucose
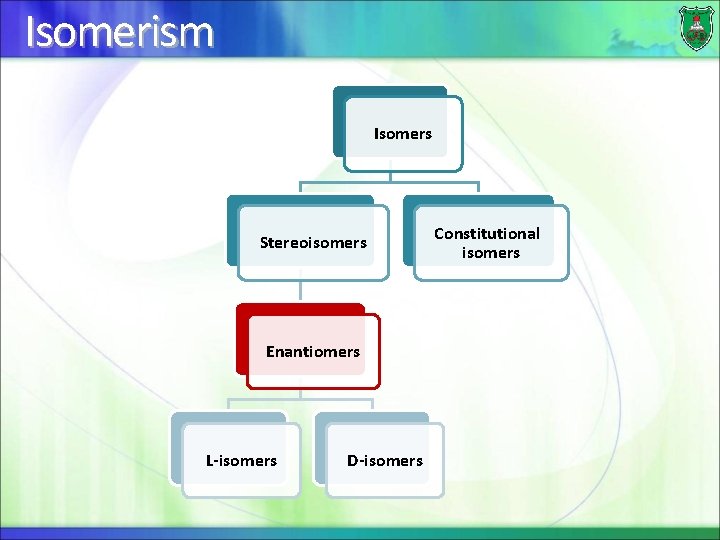
Isomerism Isomers Stereoisomers Enantiomers L-isomers D-isomers Constitutional isomers

Enantiomers When two stereoisomers are mirror images of each other, they are known as enantiomers.
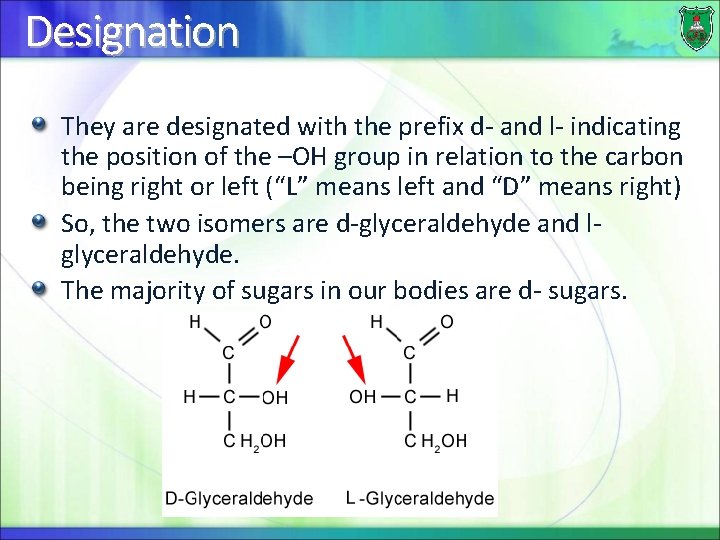
Designation They are designated with the prefix d- and l- indicating the position of the –OH group in relation to the carbon being right or left (“L” means left and “D” means right) So, the two isomers are d-glyceraldehyde and lglyceraldehyde. The majority of sugars in our bodies are d- sugars.

Naming D- or L When there is more than one chiral center in a carbohydrate, look at the chiral carbon farthest from the carbonyl group: If the hydroxy group points to right when the carbonyl is “up” it is the D-isomer, and when the hydroxy group points to the left, it is the L-isomer.

Dextrose The form most commonly found in living organisms, Dglucose, medically known as dextrose, has only one mirror image.

Cyclic sugars A monosaccharide that has 5 or 6 C atoms can form very stable ring structures. In the cell 99% of pentoses and hexoses are in the ring form. The ring forms when the aldehyde or ketone can react with one of the hydroxyl groups.
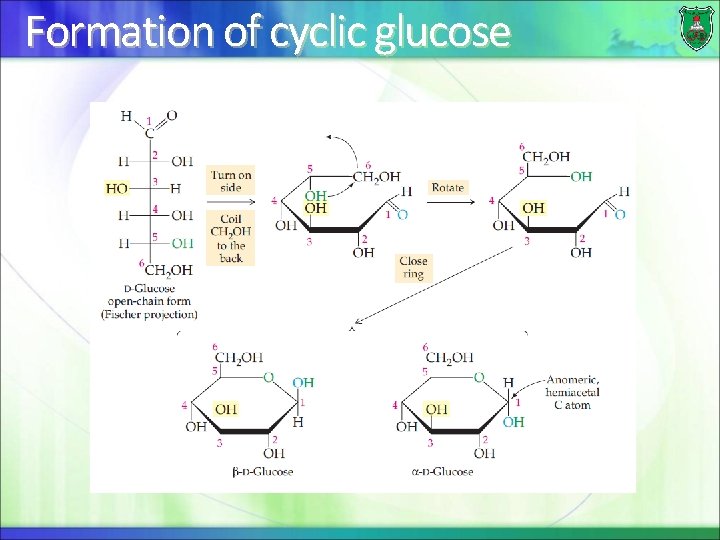
Formation of cyclic glucose
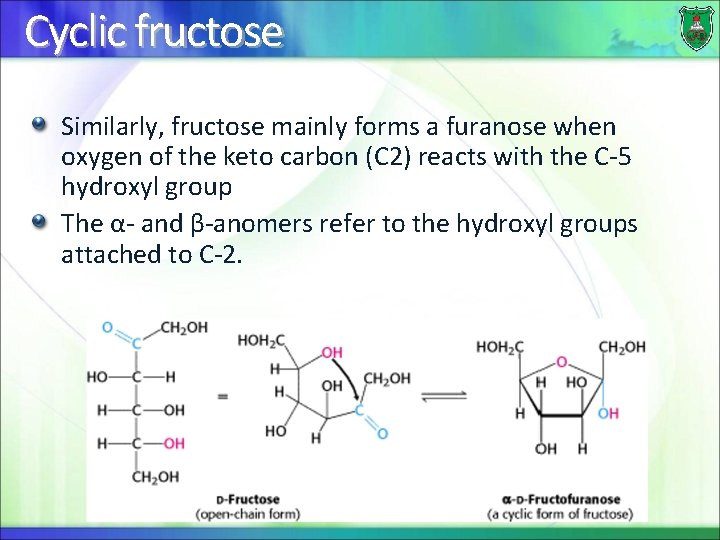
Cyclic fructose Similarly, fructose mainly forms a furanose when oxygen of the keto carbon (C 2) reacts with the C-5 hydroxyl group The α- and β-anomers refer to the hydroxyl groups attached to C-2.
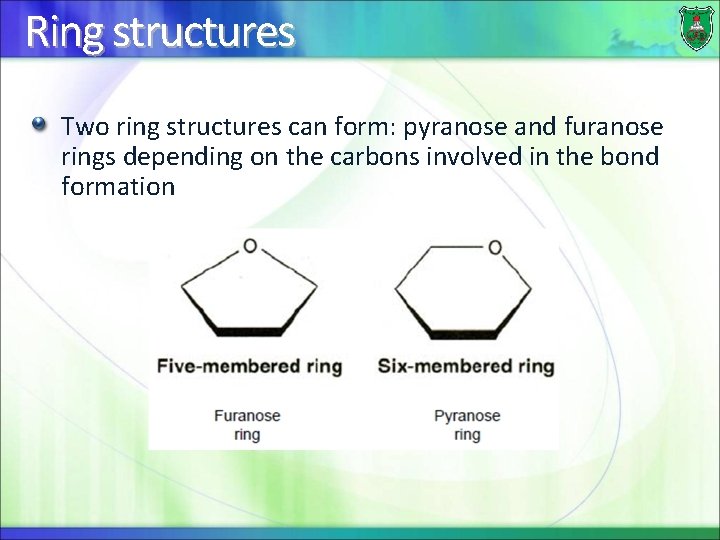
Ring structures Two ring structures can form: pyranose and furanose rings depending on the carbons involved in the bond formation
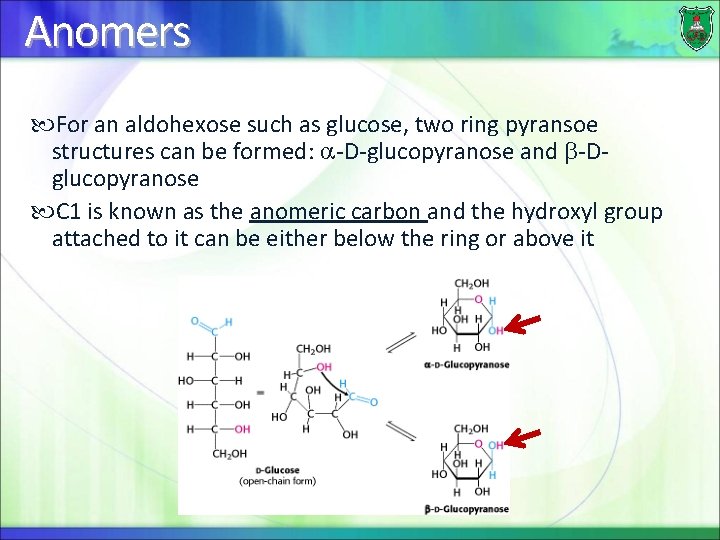
Anomers For an aldohexose such as glucose, two ring pyransoe structures can be formed: -D-glucopyranose and -Dglucopyranose C 1 is known as the anomeric carbon and the hydroxyl group attached to it can be either below the ring or above it
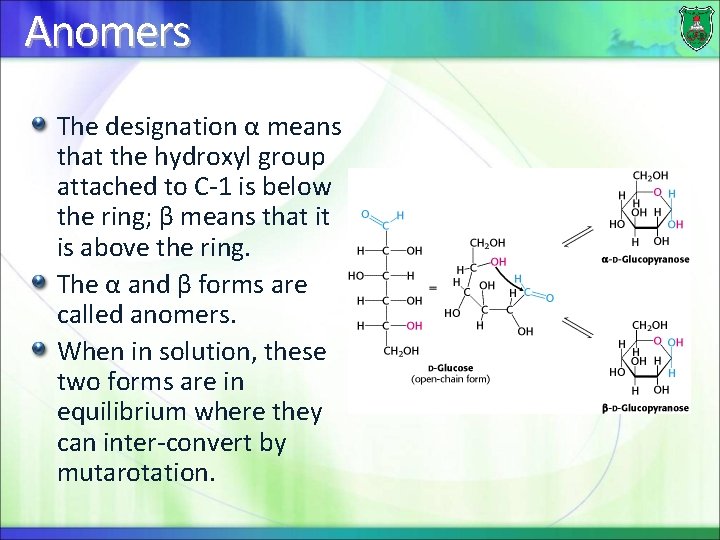
Anomers The designation α means that the hydroxyl group attached to C-1 is below the ring; β means that it is above the ring. The α and β forms are called anomers. When in solution, these two forms are in equilibrium where they can inter-convert by mutarotation.
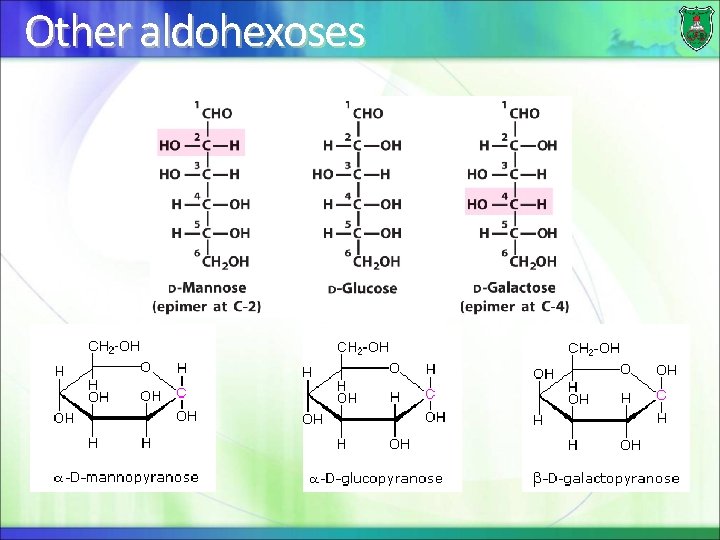
Other aldohexoses
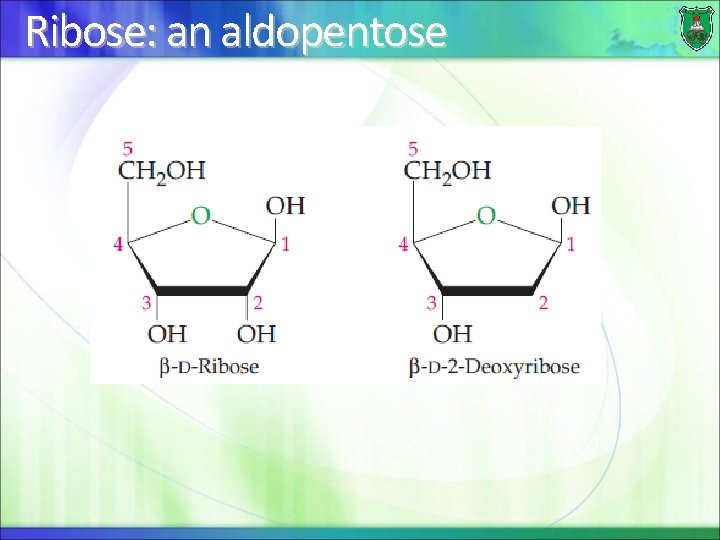
Ribose: an aldopentose
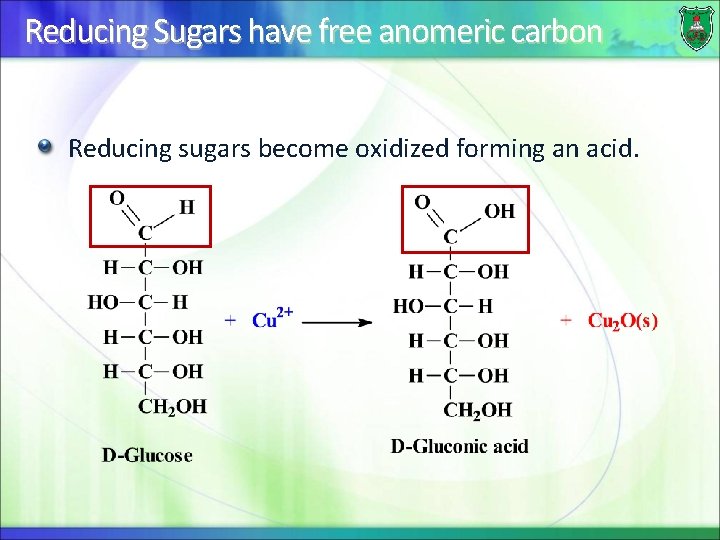
Reducing Sugars have free anomeric carbon Reducing sugars become oxidized forming an acid.
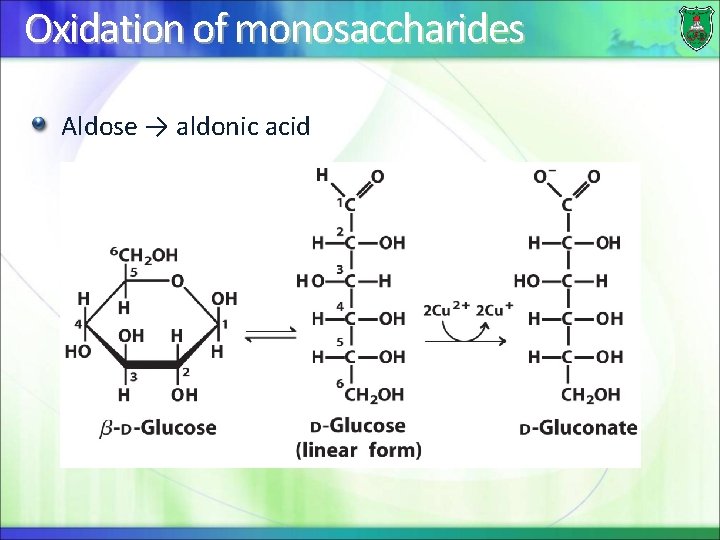
Oxidation of monosaccharides Aldose → aldonic acid
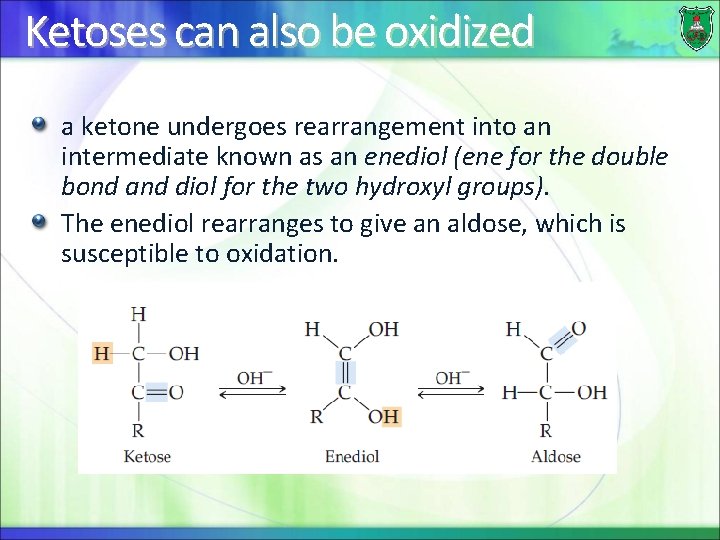
Ketoses can also be oxidized a ketone undergoes rearrangement into an intermediate known as an enediol (ene for the double bond and diol for the two hydroxyl groups). The enediol rearranges to give an aldose, which is susceptible to oxidation.
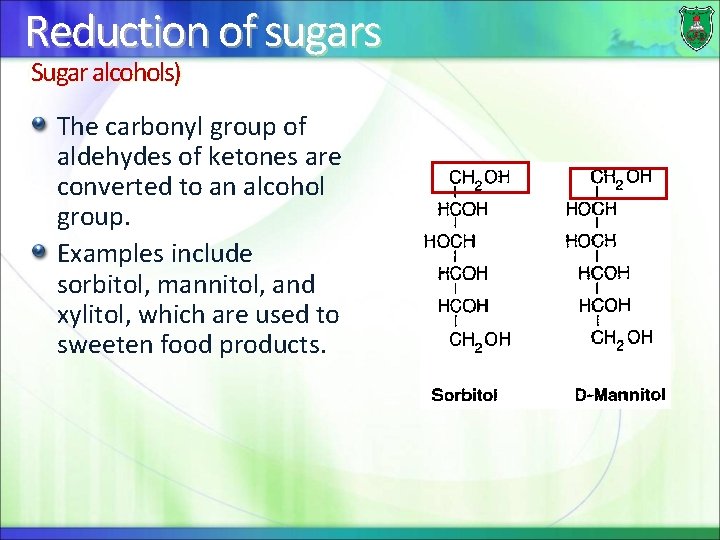
Reduction of sugars (Sugar alcohols) The carbonyl group of aldehydes of ketones are converted to an alcohol group. Examples include sorbitol, mannitol, and xylitol, which are used to sweeten food products.
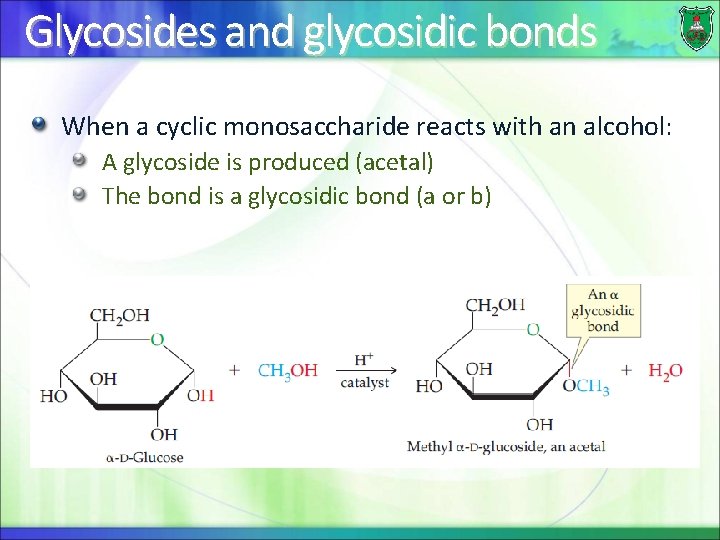
Glycosides and glycosidic bonds When a cyclic monosaccharide reacts with an alcohol: A glycoside is produced (acetal) The bond is a glycosidic bond (a or b)
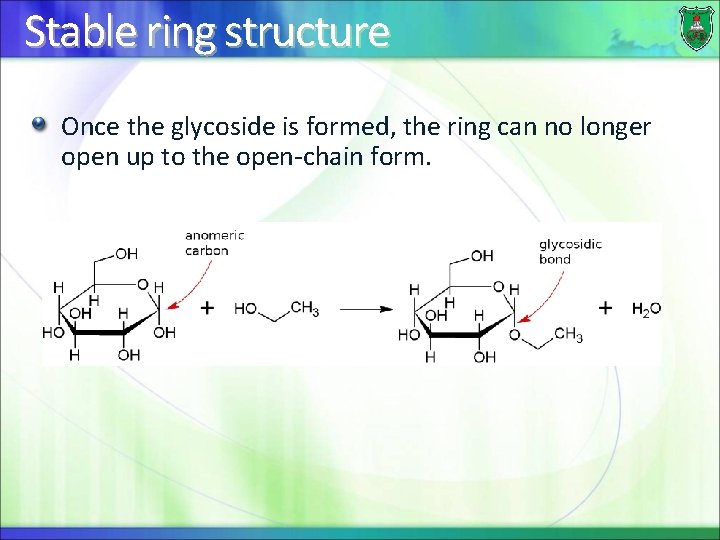
Stable ring structure Once the glycoside is formed, the ring can no longer open up to the open-chain form.

Common derivatives Oxidation of aldehyde of aldose Aldonic acids Reduction of carbonyl of aldose or ketose Alditols Condensation reactions between anomeric –OH and alcohols to form acetals or ketals: Glycosides
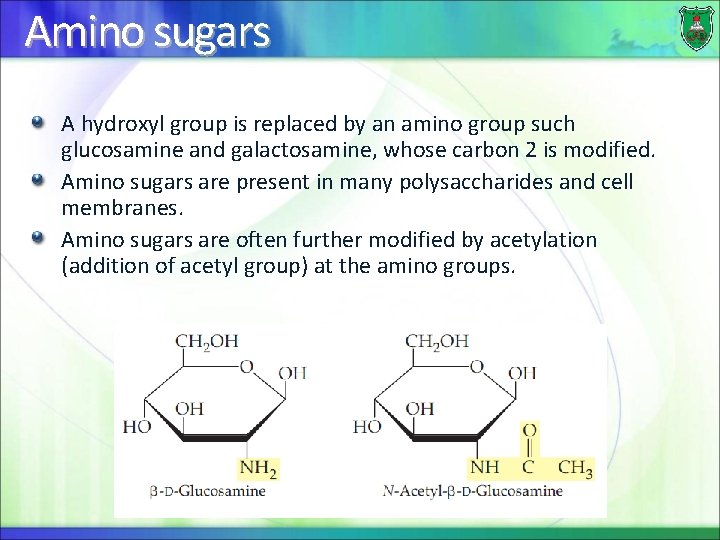
Amino sugars A hydroxyl group is replaced by an amino group such glucosamine and galactosamine, whose carbon 2 is modified. Amino sugars are present in many polysaccharides and cell membranes. Amino sugars are often further modified by acetylation (addition of acetyl group) at the amino groups.
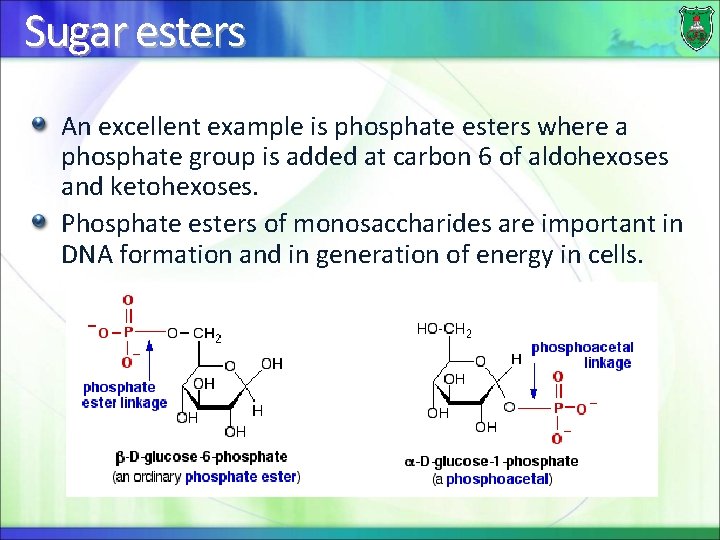
Sugar esters An excellent example is phosphate esters where a phosphate group is added at carbon 6 of aldohexoses and ketohexoses. Phosphate esters of monosaccharides are important in DNA formation and in generation of energy in cells.
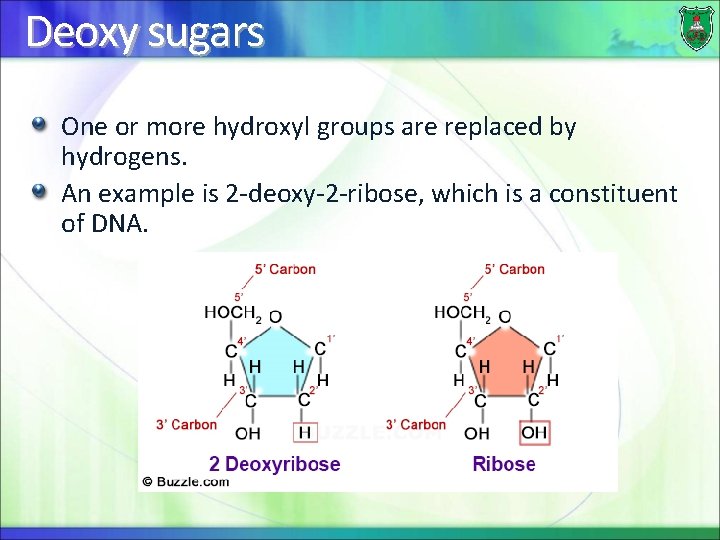
Deoxy sugars One or more hydroxyl groups are replaced by hydrogens. An example is 2 -deoxy-2 -ribose, which is a constituent of DNA.
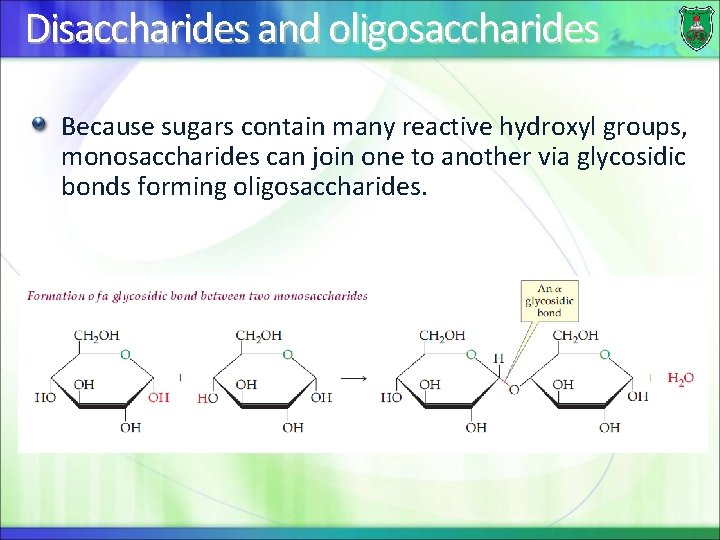
Disaccharides and oligosaccharides Because sugars contain many reactive hydroxyl groups, monosaccharides can join one to another via glycosidic bonds forming oligosaccharides.
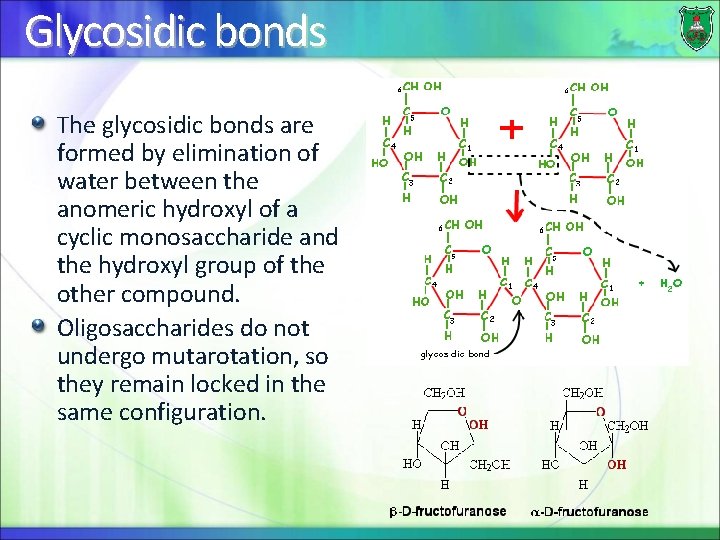
Glycosidic bonds The glycosidic bonds are formed by elimination of water between the anomeric hydroxyl of a cyclic monosaccharide and the hydroxyl group of the other compound. Oligosaccharides do not undergo mutarotation, so they remain locked in the same configuration.

Disaccharide A disaccharide consists of two sugars joined by a glycosidic bond They can be of the same sugar, thus termed homodisaccharides If they are made of two different sugars, they are known as heterodisaccharides

Distinctions of disaccharides The 2 specific sugar monomers involved and their stereoconfigurations (D- or L-) The carbons involved in the linkage (C-1, C-2, C-4, or C 6) The order of the two monomer units, if different (example: galactose followed by glucose) The anomeric configuration of the OH group on carbon 1 of each residue (α or β)
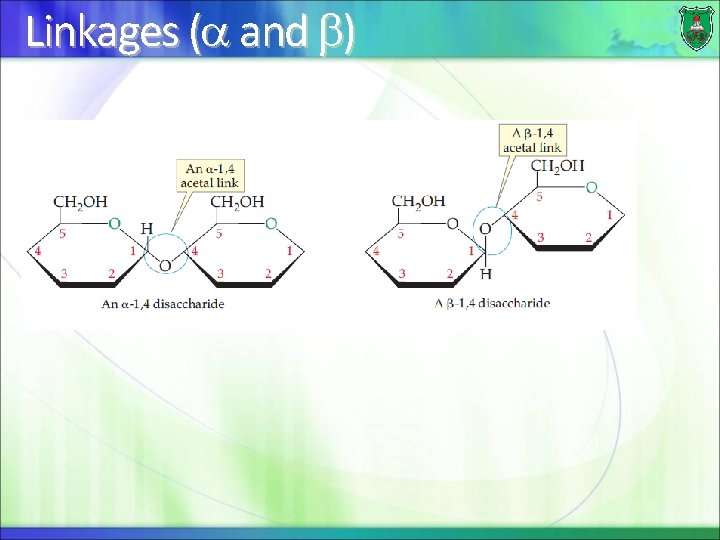
Linkages ( and )
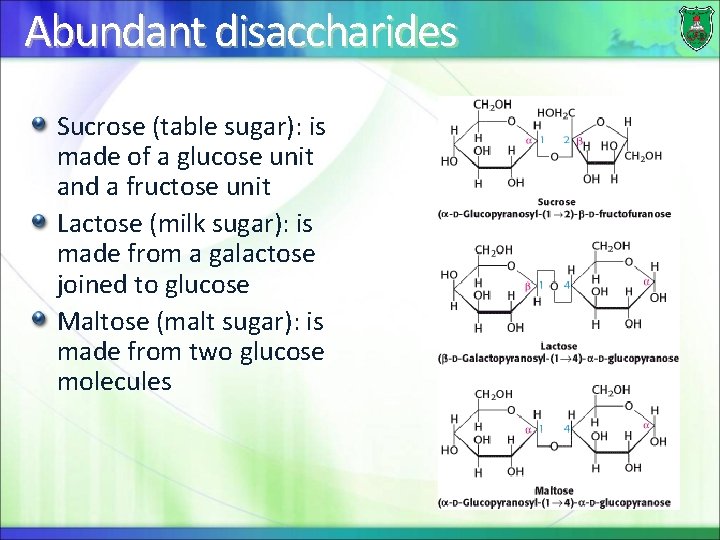
Abundant disaccharides Sucrose (table sugar): is made of a glucose unit and a fructose unit Lactose (milk sugar): is made from a galactose joined to glucose Maltose (malt sugar): is made from two glucose molecules
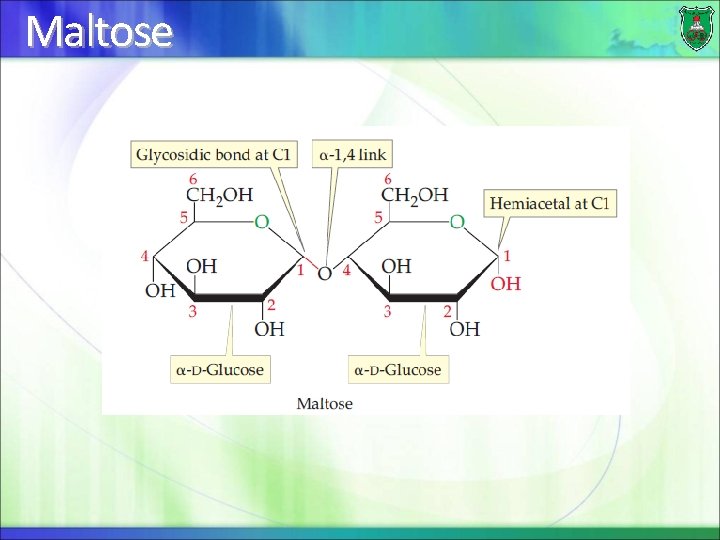
Maltose
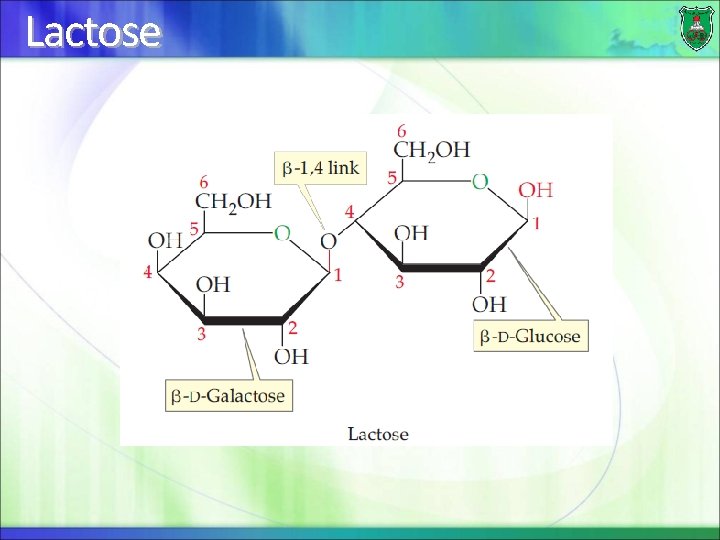
Lactose

Lactose intolerance It is a condition caused by defective lactase, which digests lactose. Because lactose remains in the intestines, it draws in excess water. Bacteria also ferment the lactose to produce carbon dioxide and methane. Symptoms: bloating, cramps, flatulence, and diarrhea. Treatment: a lactose-free diet, artificial sweeteners lactose-low milk products.
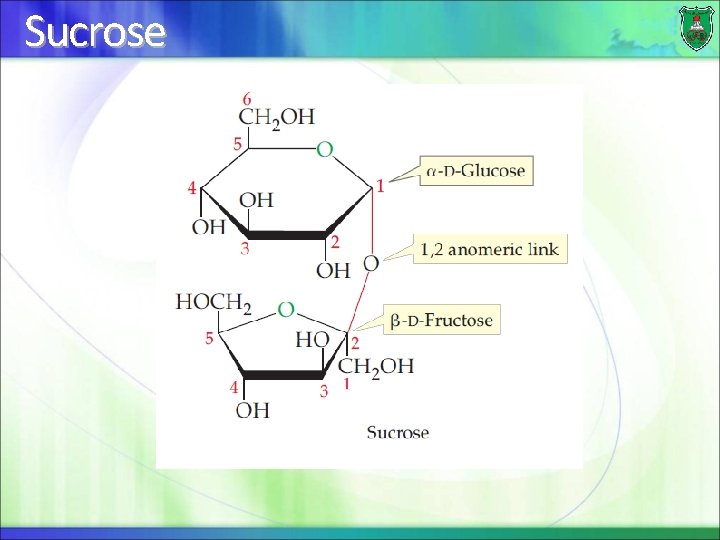
Sucrose
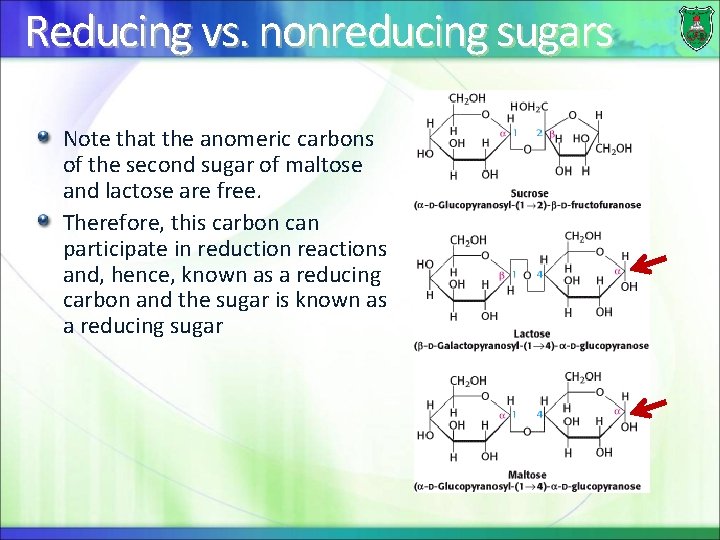
Reducing vs. nonreducing sugars Note that the anomeric carbons of the second sugar of maltose and lactose are free. Therefore, this carbon can participate in reduction reactions and, hence, known as a reducing carbon and the sugar is known as a reducing sugar
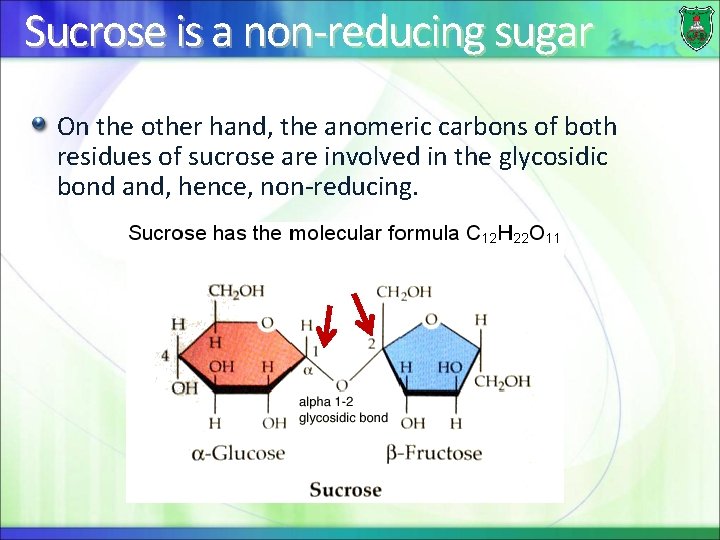
Sucrose is a non-reducing sugar On the other hand, the anomeric carbons of both residues of sucrose are involved in the glycosidic bond and, hence, non-reducing.

Polysaccharides, also called glycans, consist of more than 10 monosaccharides and their derivatives. If a polysaccharide contains only one kind of monosaccharide molecule, it is a homopolysaccharide, or homoglycan, whereas those containing more than one kind of monosaccharide are heteropolysaccharides. Polysaccharides differ not only in the nature of their component monosaccharides but also in the length of their chains and in the amount of chain branching that occurs.

Storage polysaccharides are an important carbohydrate form in plants and animals. There are three important forms of storage carbohydrates: glycogen, starch, and dextran.

Glycogen The major form of storage polysaccharide in animals is glycogen. Glycogen is found mainly in the liver (where it may amount to as much as 10% of liver mass) and skeletal muscle (where it accounts for 1 to 2% of muscle mass).
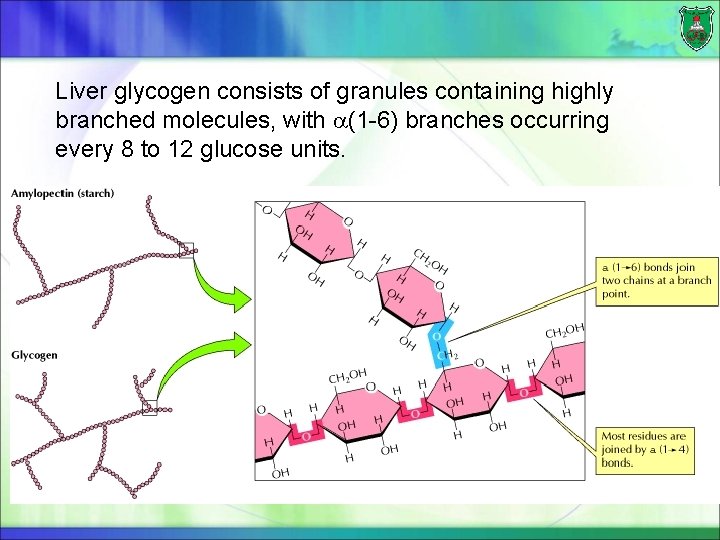
Liver glycogen consists of granules containing highly branched molecules, with (1 -6) branches occurring every 8 to 12 glucose units.
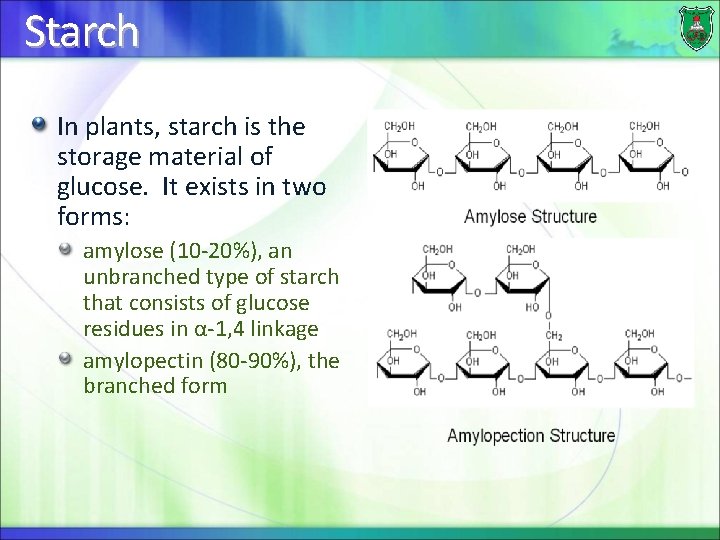
Starch In plants, starch is the storage material of glucose. It exists in two forms: amylose (10 -20%), an unbranched type of starch that consists of glucose residues in α-1, 4 linkage amylopectin (80 -90%), the branched form

Structuralpolysaccharides

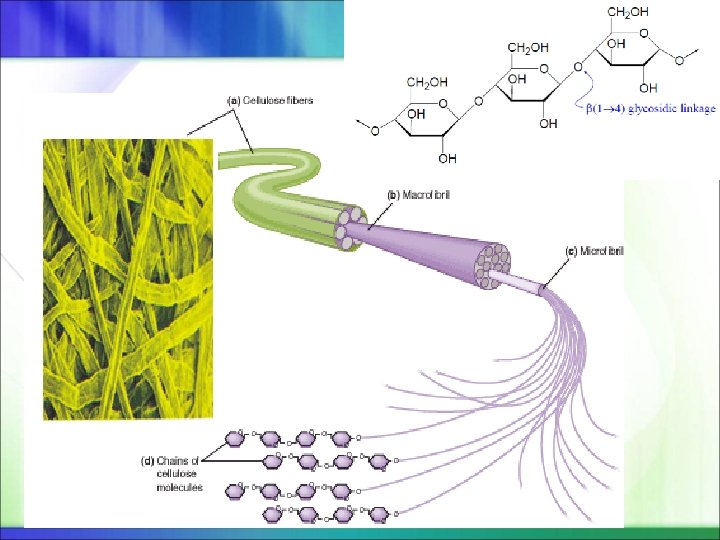
Cellulose It is the other major polysaccharide of glucose found in plants It is an unbranched polymer of glucose residues joined by β-1, 4 linkages The β configuration allows cellulose to form very long, straight chains Fibrils are formed by parallel chains that interact with one another through hydrogen bonds The straight chain formed by β linkages is optimal for the construction of fibers having a high tensile strength In contrast, the open helix formed by α linkages of glycogen and starch is well suited to forming an accessible store of sugar Mammals lack cellulases and therefore cannot digest wood and vegetable fibers
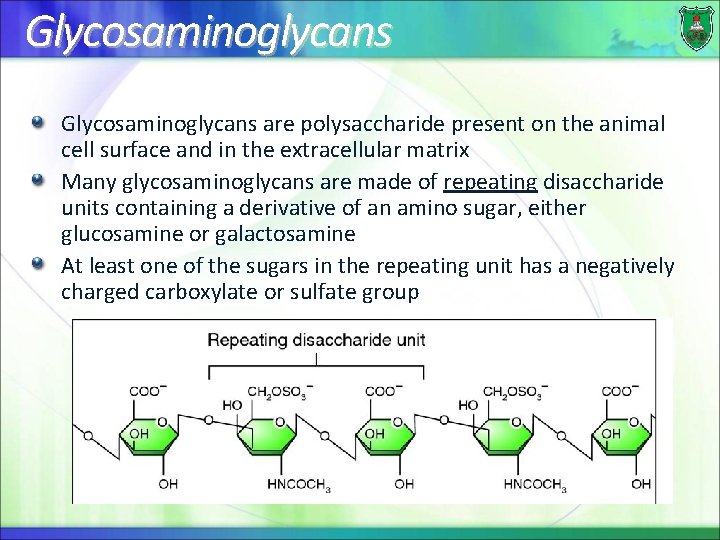
Glycosaminoglycans are polysaccharide present on the animal cell surface and in the extracellular matrix Many glycosaminoglycans are made of repeating disaccharide units containing a derivative of an amino sugar, either glucosamine or galactosamine At least one of the sugars in the repeating unit has a negatively charged carboxylate or sulfate group
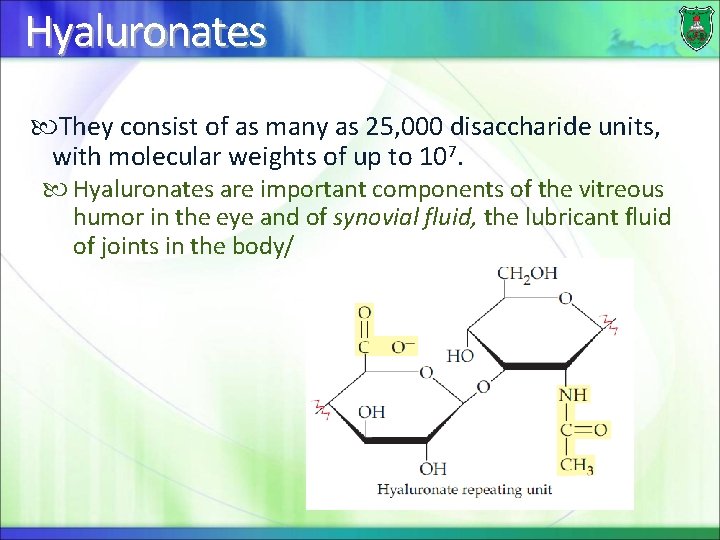
Hyaluronates They consist of as many as 25, 000 disaccharide units, with molecular weights of up to 107. Hyaluronates are important components of the vitreous humor in the eye and of synovial fluid, the lubricant fluid of joints in the body/
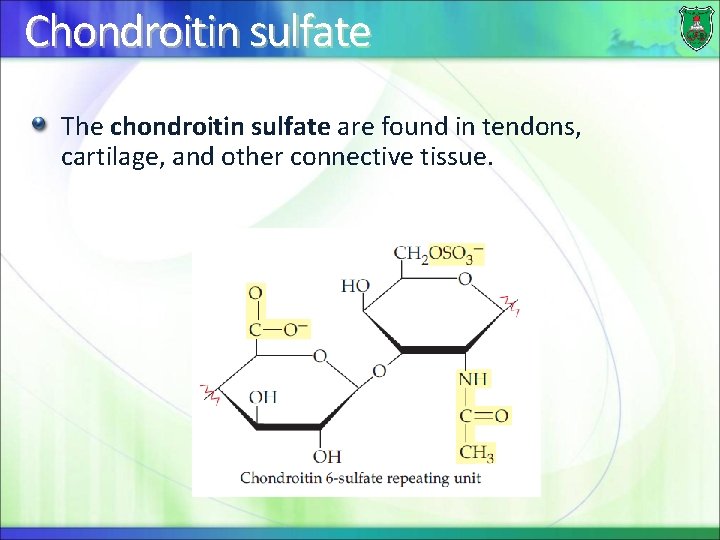
Chondroitin sulfate The chondroitin sulfate are found in tendons, cartilage, and other connective tissue.
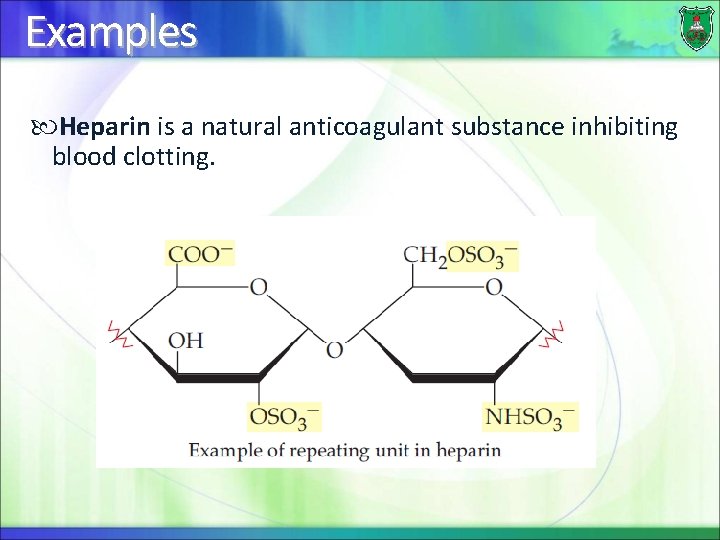
Examples Heparin is a natural anticoagulant substance inhibiting blood clotting.

Proteoglycans Glycosaminoglycans are usually attached to proteins to form proteoglycans, which contain more carbohydrates than proteins in total weight. Proteoglycans function as lubricants and structural components in connective tissue, mediate adhesion of cells to the extracellular matrix, and bind factors that stimulate cell proliferation.
Glycoproteins Carbohydrate groups are covalently attached to many different proteins to form glycoproteins. Carbohydrates are a much smaller percentage of the weight of glycoproteins. Carbohydrates are linked to some soluble proteins as well as membrane proteins. In particular, many of the proteins secreted from cells are glycosylated. The oligosaccharide precursors added to proteins may play a role in protein folding as well as in protein targeting.
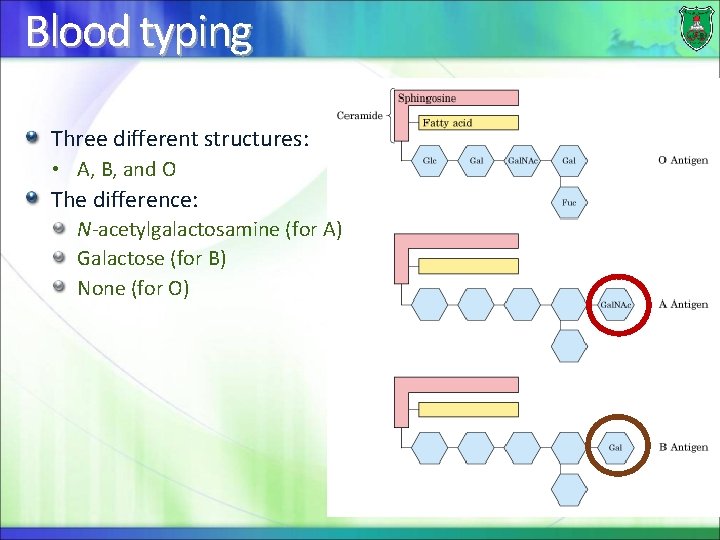
Blood typing Three different structures: • A, B, and O The difference: N-acetylgalactosamine (for A) Galactose (for B) None (for O)
- Slides: 64