Carbohydrates Contain the Elements n Carbon n Hydrogen

Carbohydrates Contain the Elements: n. Carbon n. Hydrogen n. Oxygen They Are Split Into Three Groups Known As: n. Monosaccharides(Monomers) n. Disaccharides(Dimers) n. Polsaccharides(Polymers)

Carbohydrates • Many carbohydrates are soluble in water. • The usual chemical test for the simpler carbohydrates is heating with Benedicts solution. • The formula for a carbohydrate is (CH 2 O)n • The n represents the number of times the CH 2 O unite is repeated.

Monosaccharides (monos) The Most Important Monosaccharide Is Glucose. n n n A Monosaccharide is made up of 1 sugar unit. Monos are reducing sugars. Fructose and Galactose are all so Monosaccharides, They all have the same chemical formula but different structures.
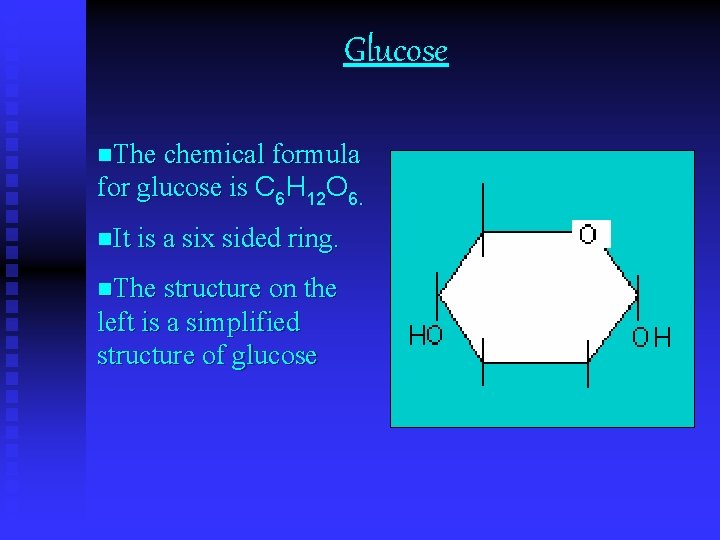
Glucose n. The chemical formula for glucose is C 6 H 12 O 6. n. It is a six sided ring. n. The structure on the left is a simplified structure of glucose

Glycosidic Bond n This is when two monosaccharides join to form a Disaccharide. n The reaction is similar to condensation. n The reaction involves the water been given off. There are three types of Disaccharides: 1. Sucrose 2. Lactose 3. Maltose
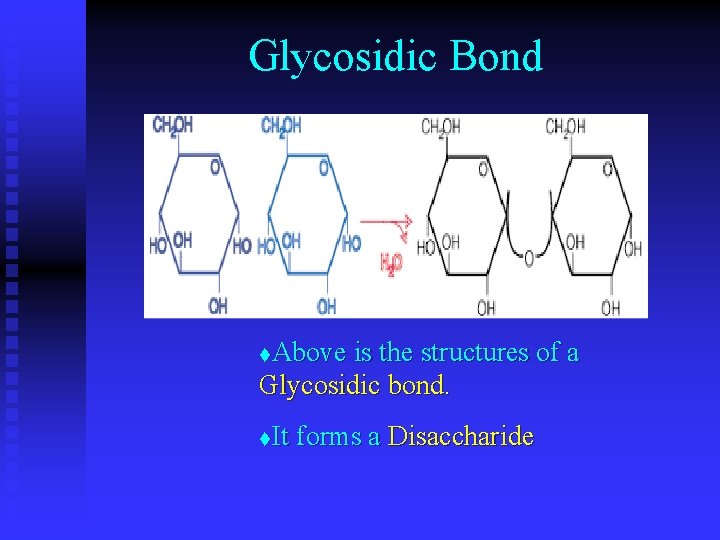
Glycosidic Bond Above is the structures of a Glycosidic bond. t It forms a Disaccharide t
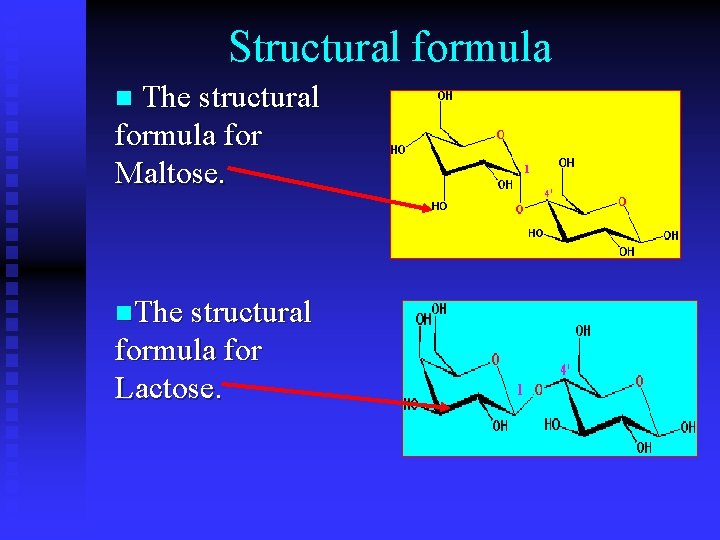
Structural formula n The structural formula for Maltose. n. The structural formula for Lactose.

Hydrolysis n This is the breaking down of a glycosidic bond. n Instead of water been taken away water is added. n Lysis means splitting.

By Sarah Kennedy Tutor Mr Rothery Data 14. 11. 02
- Slides: 9