Carbohydrates Carbohydrates Often called sugars and starches Functions

Carbohydrates

Carbohydrates Often called sugars and starches. Functions – energy, structural support Carbs are made of: 1. 2. 3. ■ ■ ■ C atoms H atoms O atoms NOTE! Usually in a 1: 2: 1 ratio!

What does it mean for the C, H, O atoms to be found in a 1: 2: 1 ratio? (For example if glucose has 6 C, how many H will it have? )
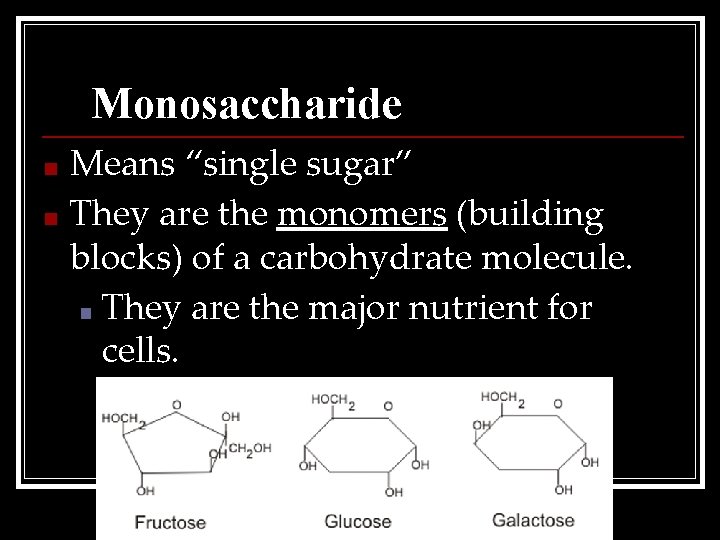
Monosaccharide Means “single sugar” ■ They are the monomers (building blocks) of a carbohydrate molecule. ■ They are the major nutrient for cells. ■

Examples 1. Glucose: Sugar produced by green plants (C 6 H 12 O 6) 2. Galactose: Found in milk 3. Fructose: Sweetest; found in fruit.
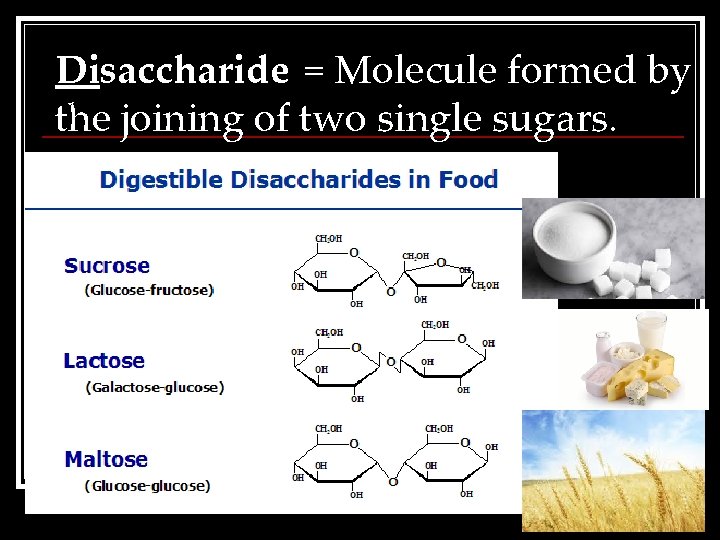
Disaccharide = Molecule formed by the joining of two single sugars.

What do you notice about the appearance of these disaccharides?

Polysaccharide = “Many sugars” Living things store excess sugar in this form. Ex. Cellulose/starch – plants, Glycogen - animals
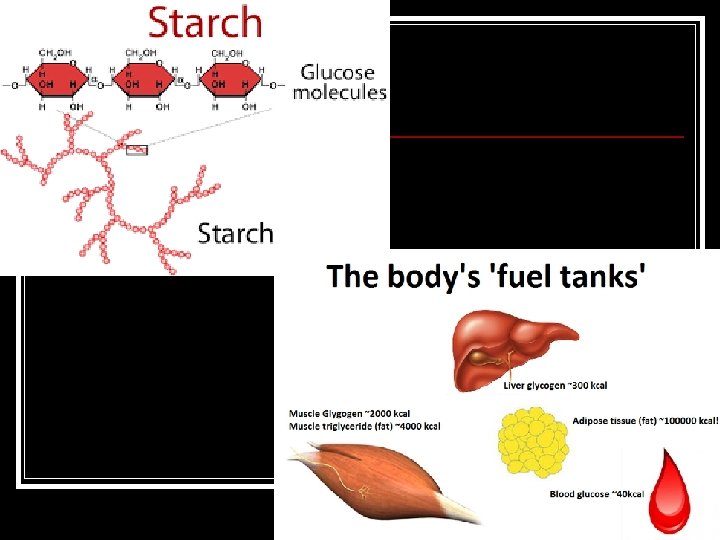

What do you notice about the appearance of these polysaccharides?

Sugars Contain a Great Deal of Energy! ■ Energy is stored in the chemical bonds. ■ Energy is released when the bonds are broken.
Drag your dot to indicate your answer. Do you think monosaccharides, disaccharides, and polysaccharides provide the same amount of energy? Yes No

Dehydration Synthesis ■ The chemical reaction that joins monomers together by removing a molecule of water. ■ Dehydration = “loss of water” Synthesis = “putting together” ■
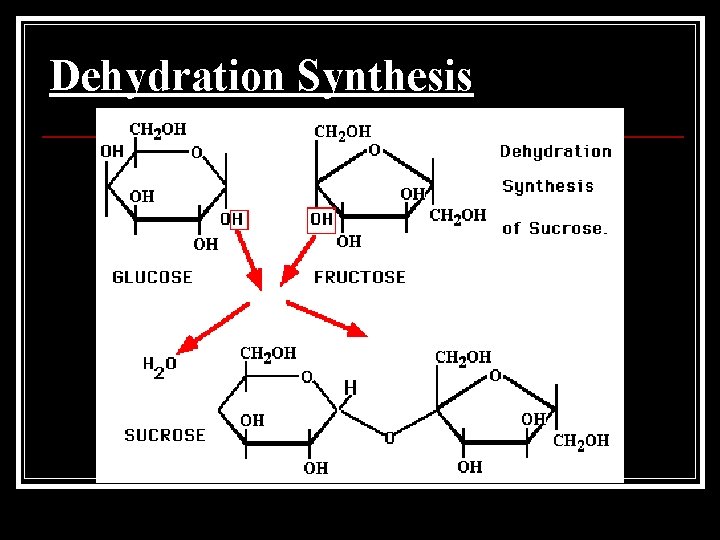
Dehydration Synthesis
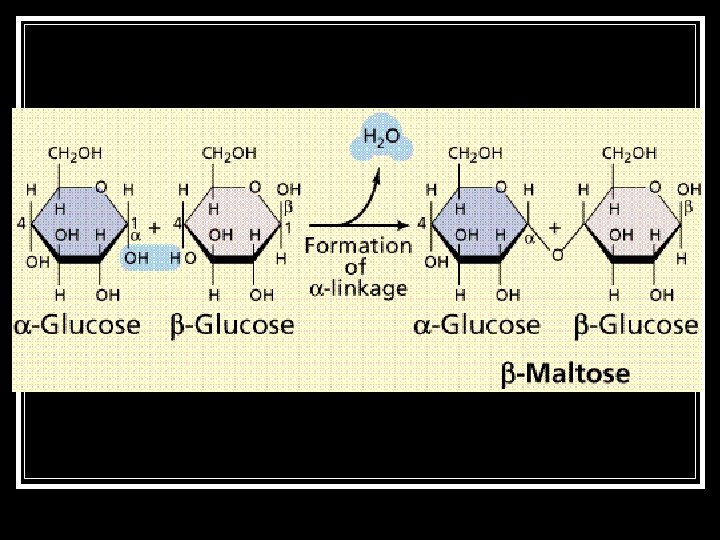

Hydrolysis (Condensation) ■ The exact opposite of Dehydration Synthesis. ■ A molecule of water is added to break a bond.
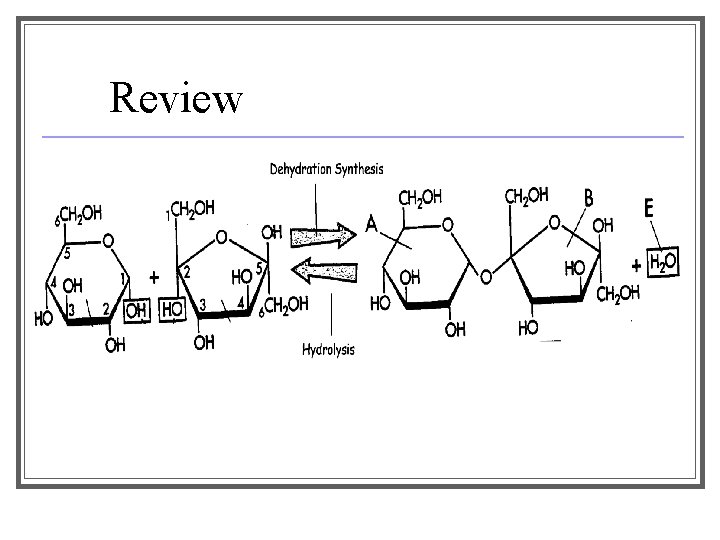
Review

Recap! ■ D. S. → starts with two separate things, ends with one, removes water ■ Hydrolysis → starts with one thing, ends with two, adds water
- Slides: 18