Carbohydrates Carbohydrates are composed of C H O
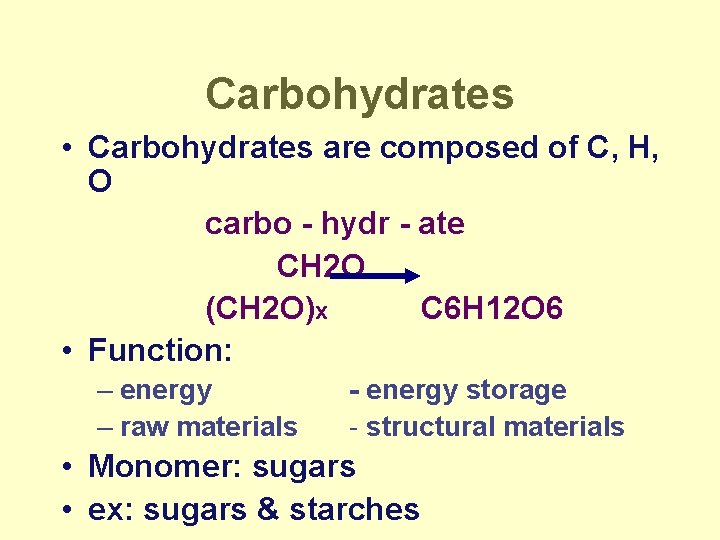
Carbohydrates • Carbohydrates are composed of C, H, O carbo - hydr - ate CH 2 O (CH 2 O)x C 6 H 12 O 6 • Function: – energy – raw materials - energy storage - structural materials • Monomer: sugars • ex: sugars & starches
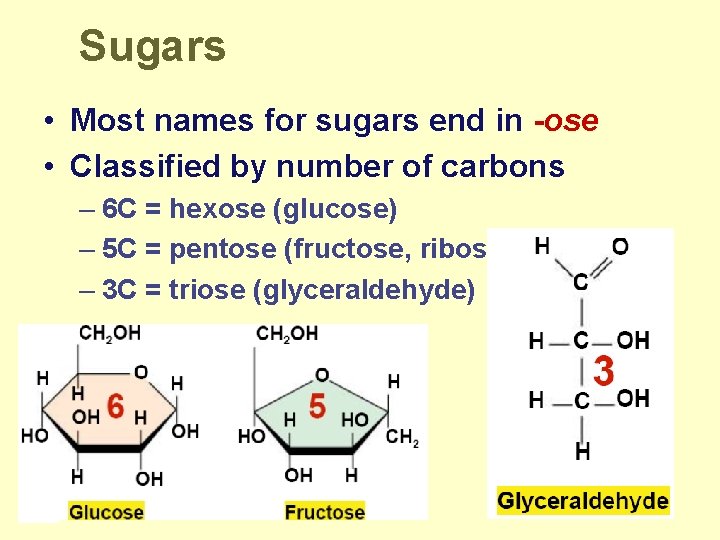
Sugars • Most names for sugars end in -ose • Classified by number of carbons – 6 C = hexose (glucose) – 5 C = pentose (fructose, ribose) – 3 C = triose (glyceraldehyde)
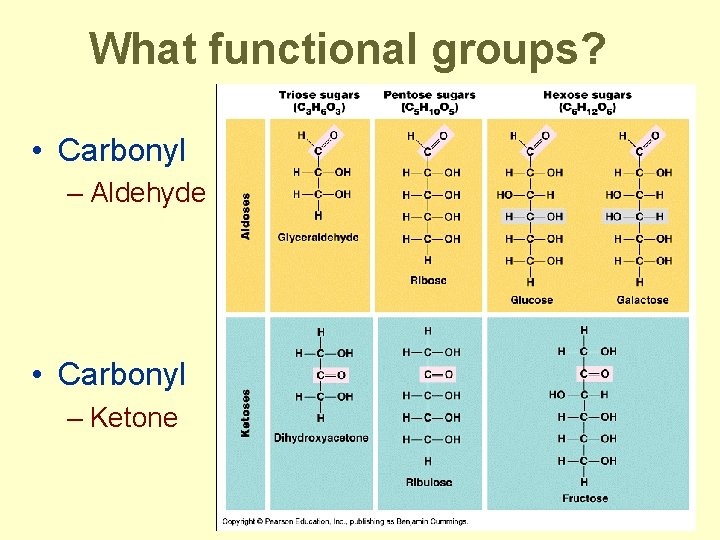
What functional groups? • Carbonyl – Aldehyde • Carbonyl – Ketone
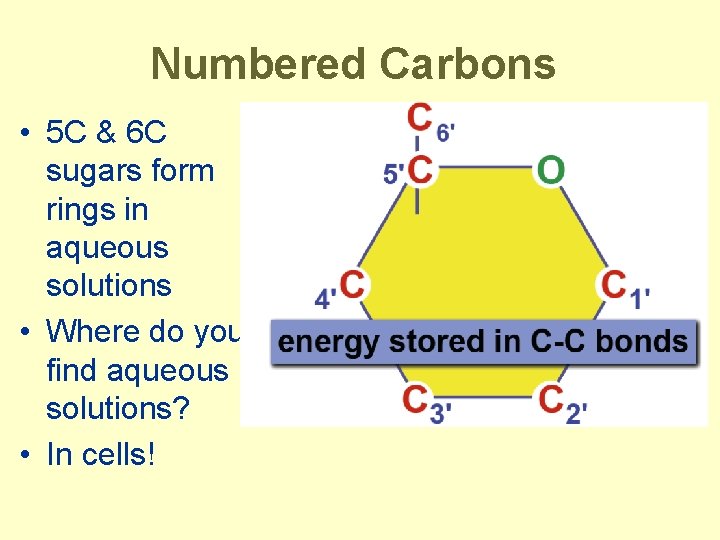
Numbered Carbons • 5 C & 6 C sugars form rings in aqueous solutions • Where do you find aqueous solutions? • In cells!
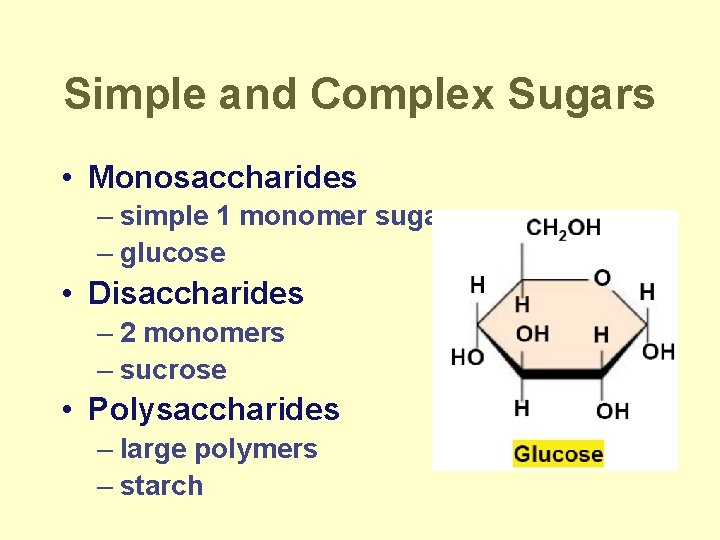
Simple and Complex Sugars • Monosaccharides – simple 1 monomer sugars – glucose • Disaccharides – 2 monomers – sucrose • Polysaccharides – large polymers – starch
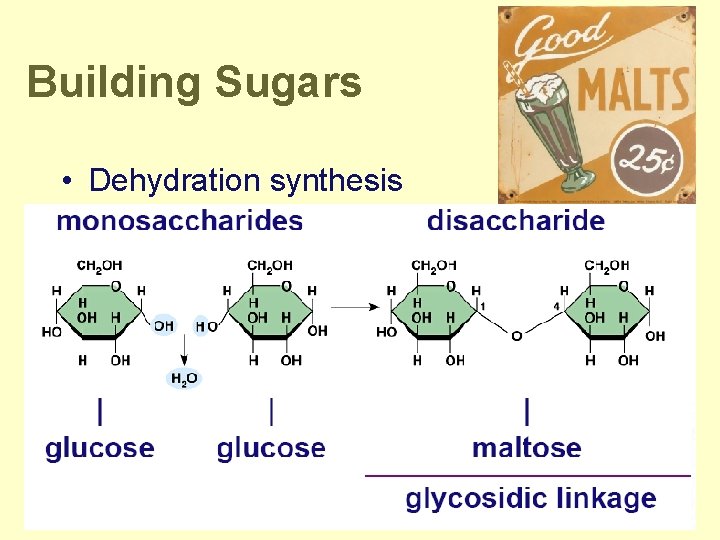
Building Sugars • Dehydration synthesis
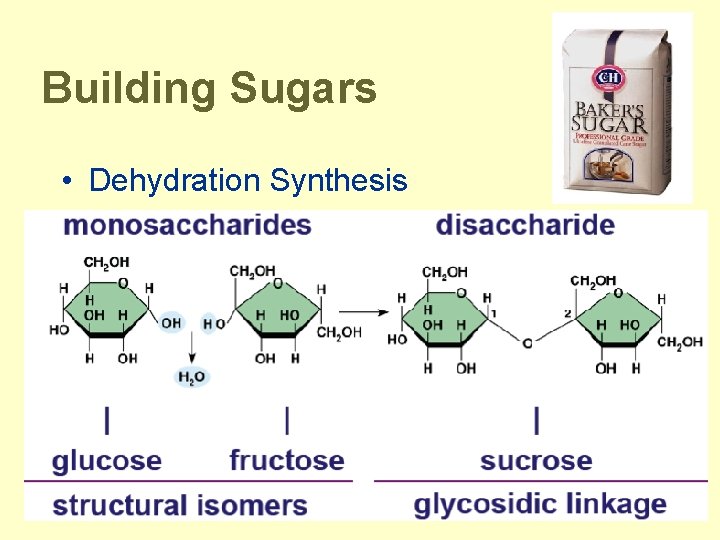
Building Sugars • Dehydration Synthesis

Polysaccharides • Polymers of sugars – costs little energy to build – easily reversible = release energy • Function: – energy storage • starch (plants) • glycogen (animals) – structure = building materials • cellulose (plants) • chitin (arthropods & fungi)

Branched vs. Linear Polysaccharides • What does branching do?
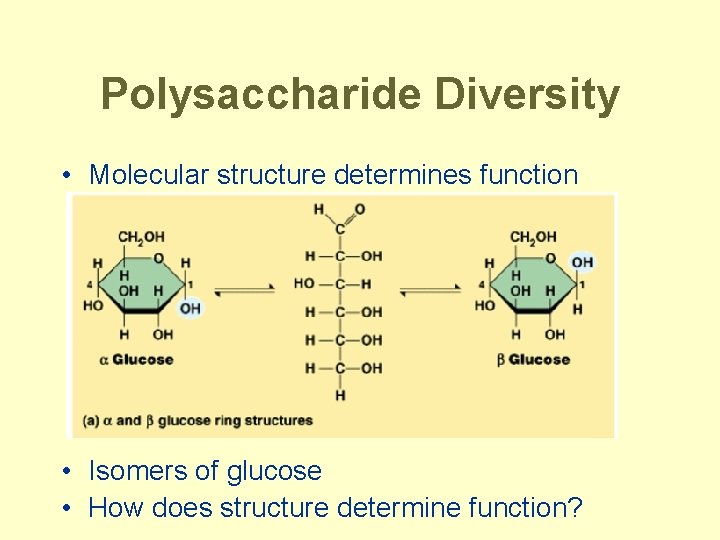
Polysaccharide Diversity • Molecular structure determines function • Isomers of glucose • How does structure determine function?
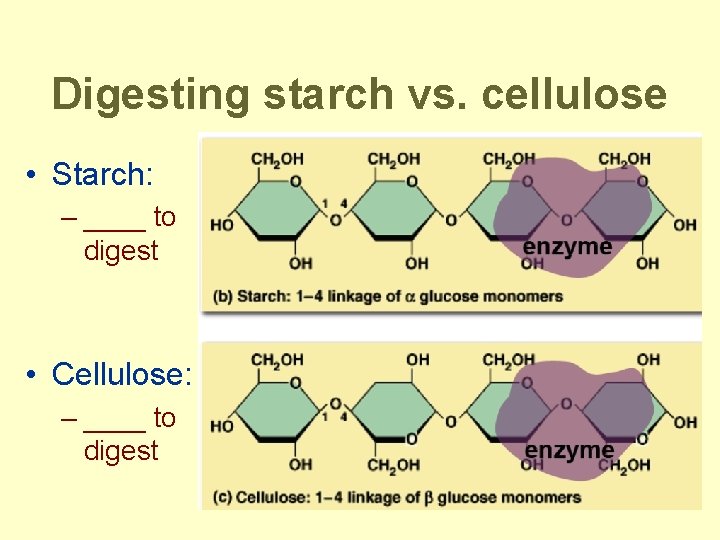
Digesting starch vs. cellulose • Starch: – ____ to digest • Cellulose: – ____ to digest
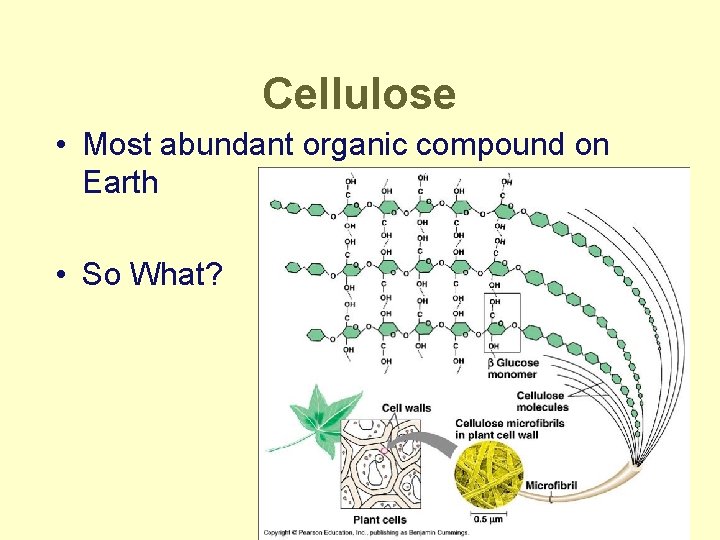
Cellulose • Most abundant organic compound on Earth • So What?

• Cow – can digest cellulose well; no need to eat supplemental sugars • Gorilla – can’t digest cellulose well; must supplement with sugar source, like fruit

Macromolecules Lipids

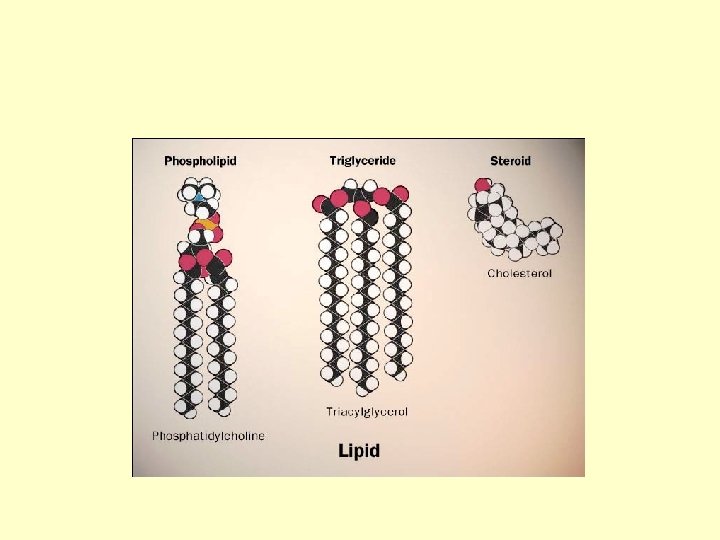
Lipids • Lipids are composed of C, H, O – long hydrocarbon chain • Diverse group – fats – phospholipids – steroids • Do not form polymers – big molecules made of smaller subunits – not a continuing chain
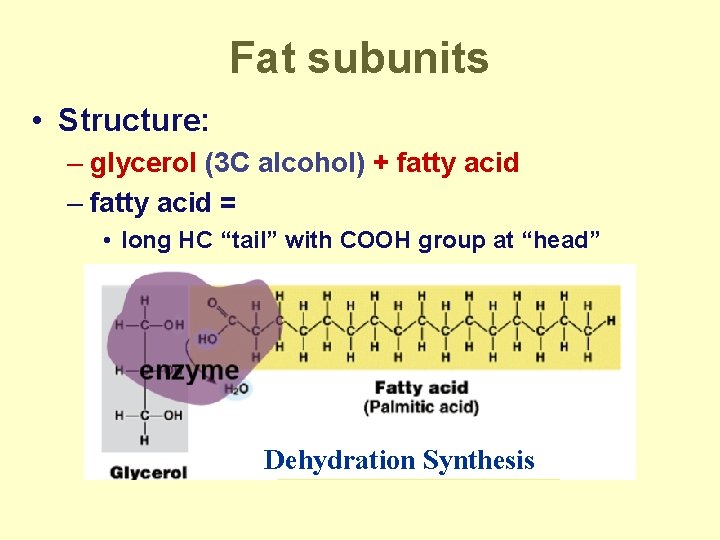
Fat subunits • Structure: – glycerol (3 C alcohol) + fatty acid – fatty acid = • long HC “tail” with COOH group at “head” Dehydration Synthesis
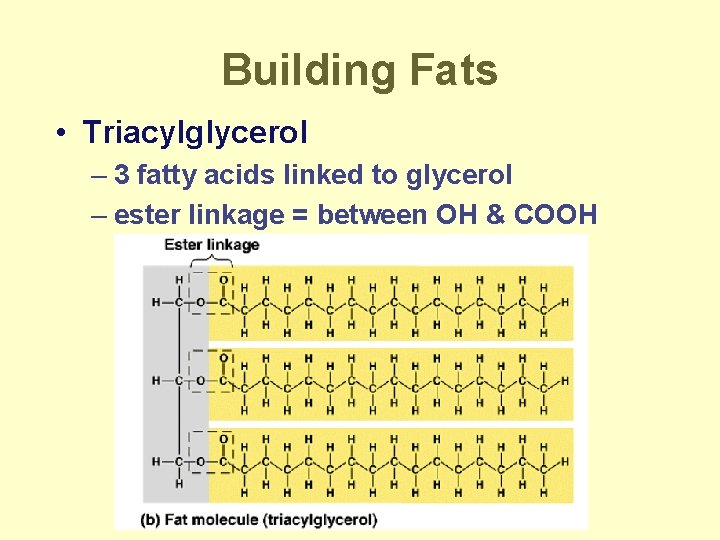
Building Fats • Triacylglycerol – 3 fatty acids linked to glycerol – ester linkage = between OH & COOH
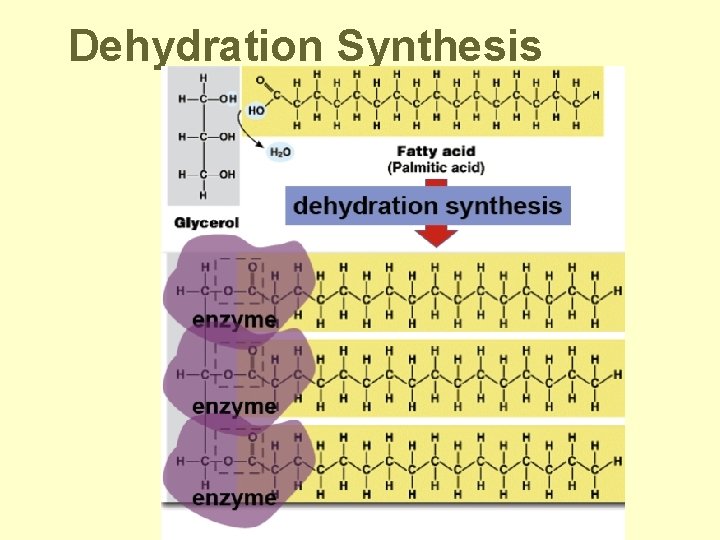
Dehydration Synthesis
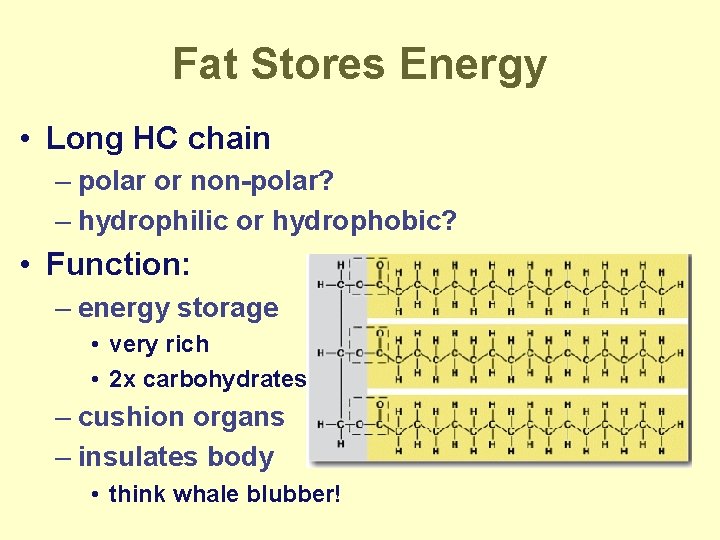
Fat Stores Energy • Long HC chain – polar or non-polar? – hydrophilic or hydrophobic? • Function: – energy storage • very rich • 2 x carbohydrates – cushion organs – insulates body • think whale blubber!
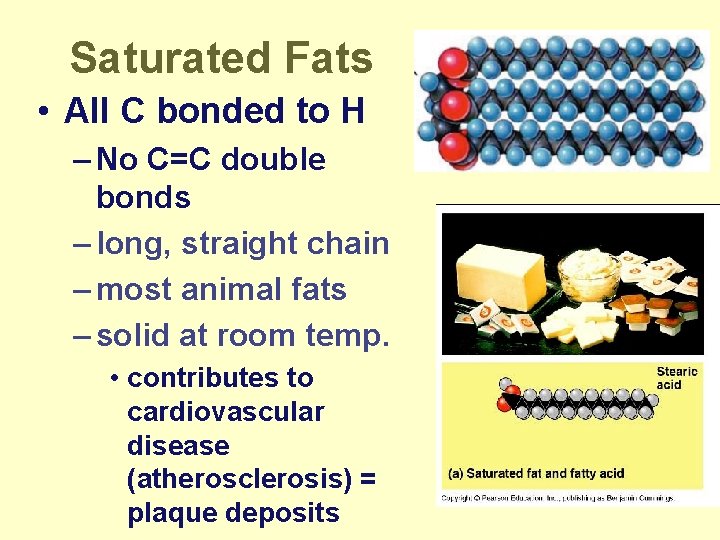
Saturated Fats • All C bonded to H – No C=C double bonds – long, straight chain – most animal fats – solid at room temp. • contributes to cardiovascular disease (atherosclerosis) = plaque deposits

Unsaturated Fats • C=C double bonds in the fatty acids – plant & fish fats – vegetable oils – liquid at room temperature • the kinks made by double bonded C prevent the molecules from packing tightly together
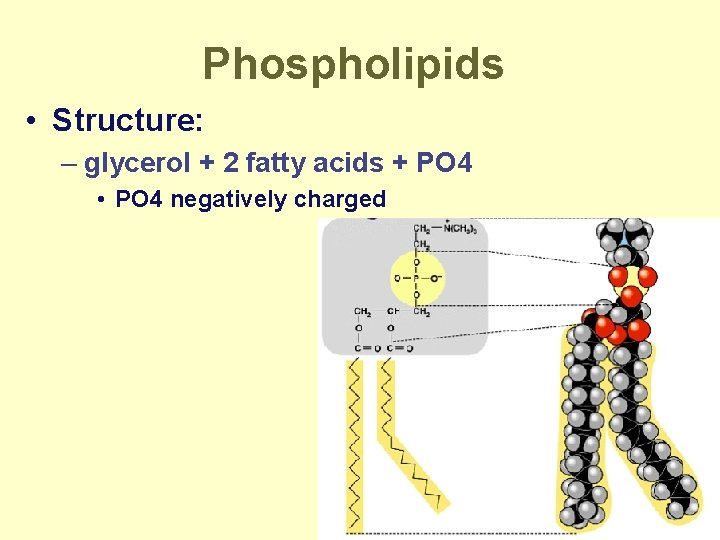
Phospholipids • Structure: – glycerol + 2 fatty acids + PO 4 • PO 4 negatively charged
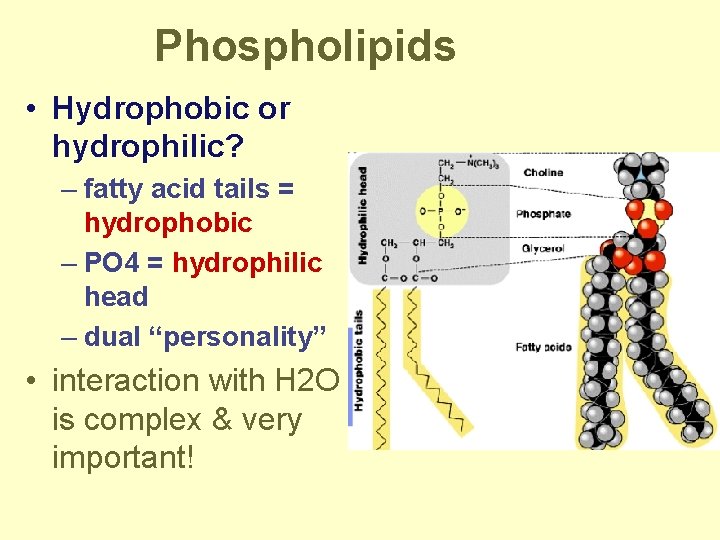
Phospholipids • Hydrophobic or hydrophilic? – fatty acid tails = hydrophobic – PO 4 = hydrophilic head – dual “personality” • interaction with H 2 O is complex & very important!
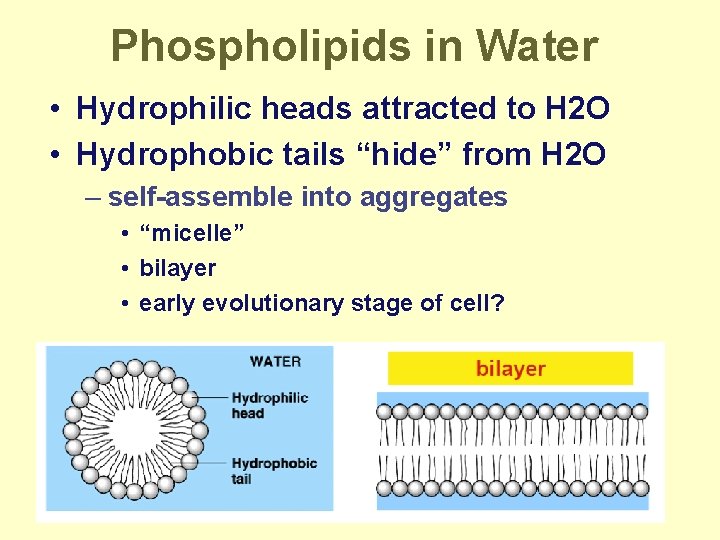
Phospholipids in Water • Hydrophilic heads attracted to H 2 O • Hydrophobic tails “hide” from H 2 O – self-assemble into aggregates • “micelle” • bilayer • early evolutionary stage of cell?

So…why is this important? • Phospholipids create a barrier in water – define outside vs. inside – cell membranes
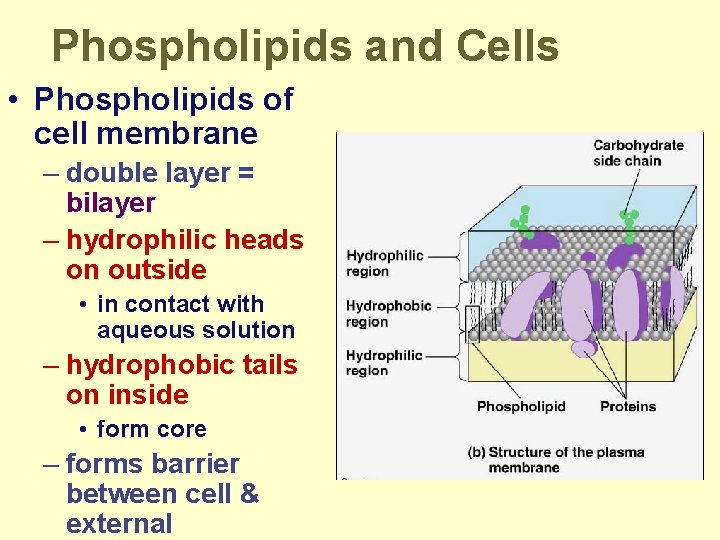
Phospholipids and Cells • Phospholipids of cell membrane – double layer = bilayer – hydrophilic heads on outside • in contact with aqueous solution – hydrophobic tails on inside • form core – forms barrier between cell & external
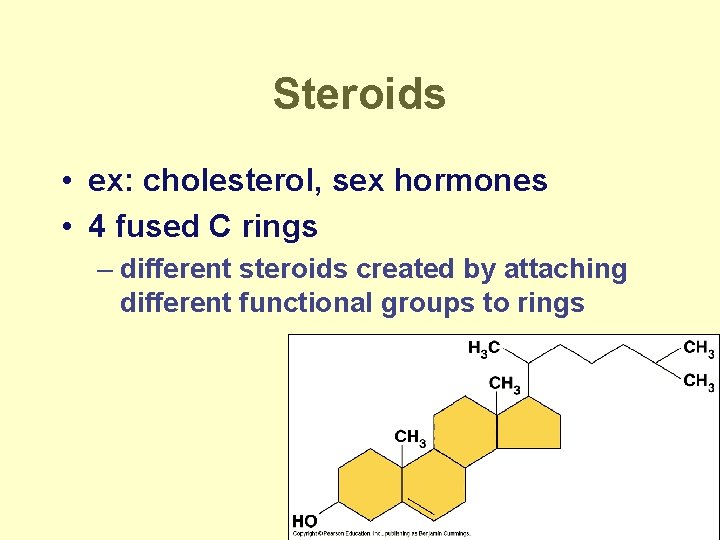
Steroids • ex: cholesterol, sex hormones • 4 fused C rings – different steroids created by attaching different functional groups to rings
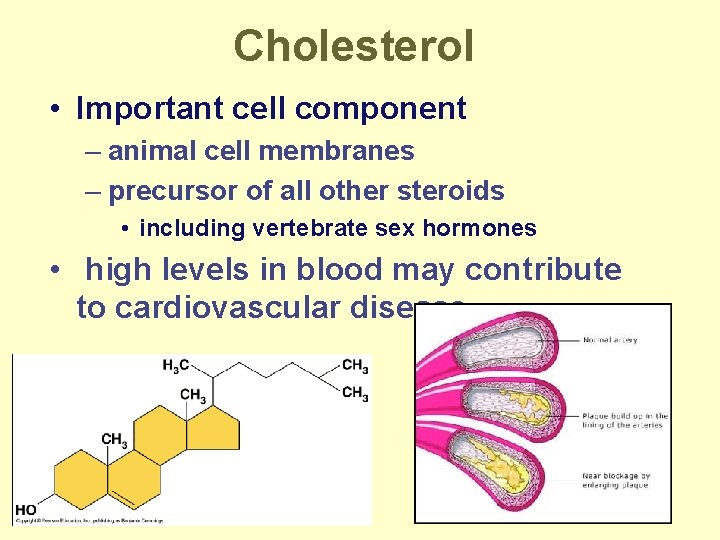
Cholesterol • Important cell component – animal cell membranes – precursor of all other steroids • including vertebrate sex hormones • high levels in blood may contribute to cardiovascular disease
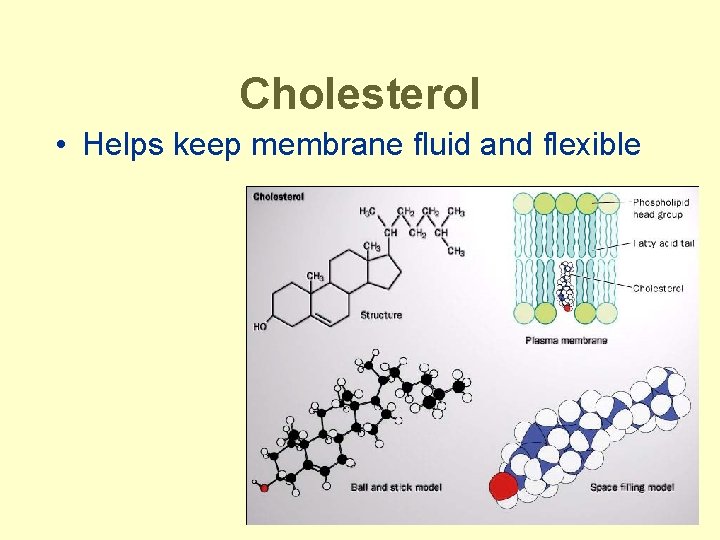
Cholesterol • Helps keep membrane fluid and flexible
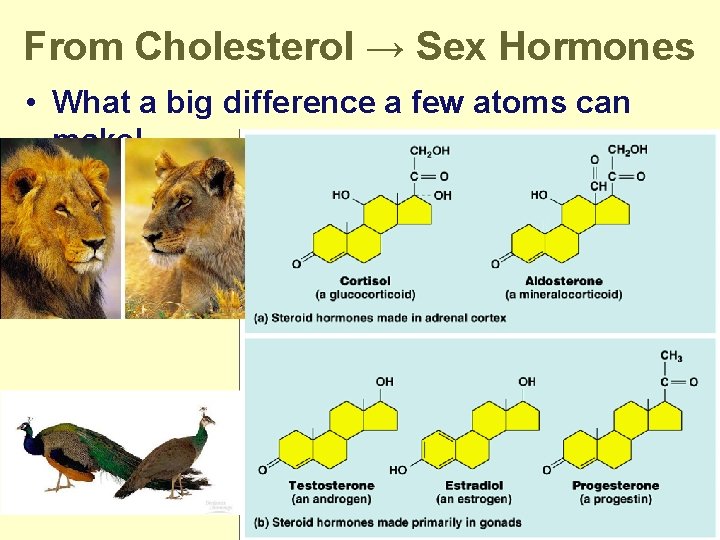
From Cholesterol → Sex Hormones • What a big difference a few atoms can make!
- Slides: 31