Carbohydrates Carbohydrate chemistry Definition classification with examples sources

Carbohydrates

• Carbohydrate chemistry: • Definition, classification with examples, sources of important carbohydrates • Components and bonds present in maltose, lactose, sucrose, trehalose

• Criteria for reducing and non reducing sugars. Briefly the structures of starch, glycogen • Mucopolysaccharides: Composition and significance of hyaluronic acid, chondroitin sulfate, heparin

Definition • Carbohydrates are polyhydroxy aldehydes or ketones or • Compounds, which yield such derivatives on hydrolysis

– Abundant organic molecules – Important constituent of our food

Functions

1. Most abundant dietary source of energy for the body (50 -60%) Ø storage form is glycogen 2. Precursors for many organic compounds: fatty acids (fats), glycerol, amino acids etc.

3. Structural components– Cellulose in plants, – Exoskeleton in insects, – Cell wall of micro organisms, seed husks etc.

4. Components of extra cellular matrix mucopolysaccharides 5. Ribose and deoxyribose in the synthesis of nucleotides, nucleic acids, coenzymes etc.

6. as glycoprotein and glycolipids • participate in • the structure of cell membrane, nervous tissues • other cellular functions such as adhesion and recognition of molecules

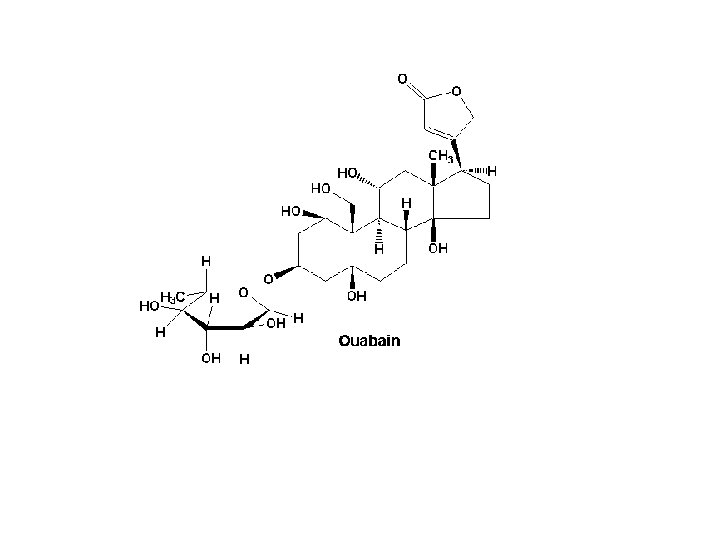
7. Used In the synthesis of • Drugs Streptomycin, Ouabain, digitoxin etc. • Conjugation reactions etc.

Chemistry

• Carbohydrates are » Primarily made up of carbon, hydrogen and oxygen hydrates of carbon : Cn(H 2 O)n » Ratio of carbon, hydrogen, oxygen is 1: 2: 1

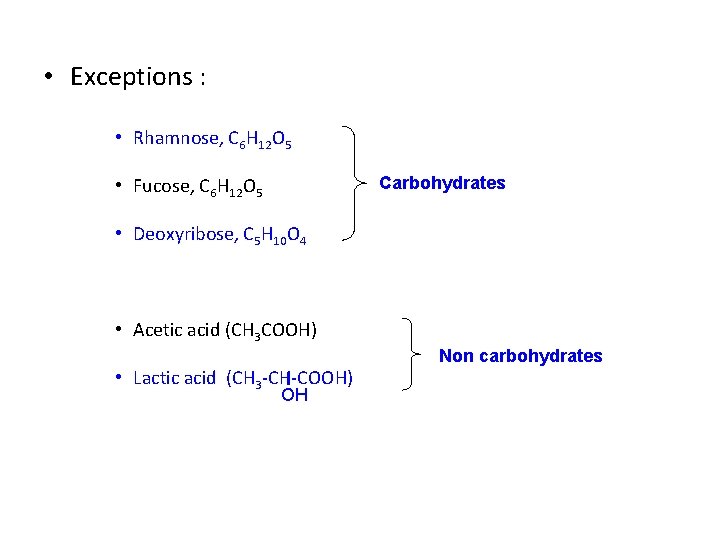
• Exceptions : • Rhamnose, C 6 H 12 O 5 Carbohydrates • Fucose, C 6 H 12 O 5 • Deoxyribose, C 5 H 10 O 4 • Acetic acid (CH 3 COOH) • Lactic acid (CH 3 -CH-COOH) OH Non carbohydrates

Classification of carbohydrates

• Classified based on the number of monosaccharide units in them into 1. Monosaccharides 2. Disaccharides & Oligosaccharides 3. Polysaccharides

1. Monosaccharides

– The simplest of carbohydrates – Contain one sugar unit (monomer) & cannot be hydrolyzed into smaller carbohydrate molecules

– Generally they are sweet to taste – Crystalline – Soluble in water

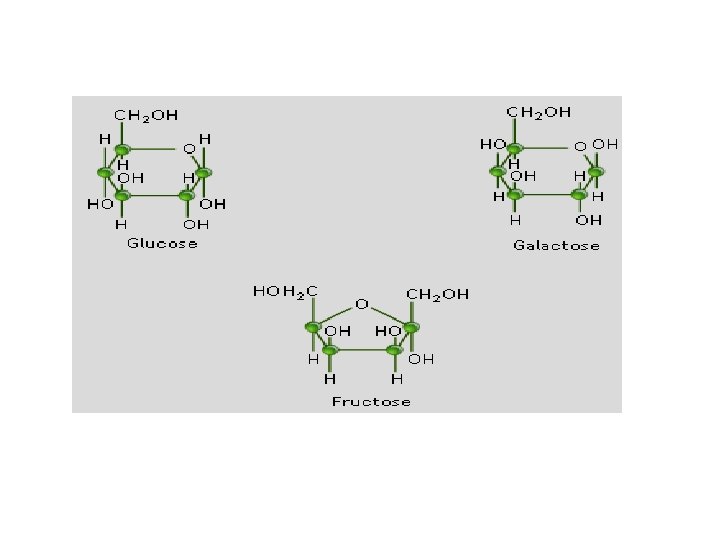
• General formula is Cn. H 2 n. On – Eg. • Glucose, Fructose, Galactose, Mannose • Ribose, Deoxyribose • Erythrose, Xylose • Glyceraldehyde, DHA etc. » Sources:

• Monosaccharides are further classified

a. Based on number of carbon atoms present • • • b. Trioses Tetroses Pentoses Hexoses Heptoses Based on functional group • • Aldoses ketoses

Trioses Eg. • Aldotriose Glyceraldehyde • Ketotriose Dihydroxyacetone

Tetroses Eg. • Aldotetrose Erythrose, Threose • Ketotetrose Erythrulose

Pentoses Eg. • Aldopentose Ribose; Arabinose, Xylose (glycoproteins, gums) , Lyxose (heart muscle) • Ketopentose Ribulose, Xylulose (uronic acid pathway)

Hexoses Eg. • Aldohexoses Glucose, Galactose, Mannose etc. • Ketohexose Fructose: present in fruits, honey etc.

Heptoses Eg. • Aldoheptose Glucoheptose • Ketoheptose Sedoheptulose: an intermediate of HMP shunt

2. Disaccharides

◦ Contain two monosaccharide units joined by glycosidic bond or ◦ Yield two monosaccharide units upon hydrolysis

• General formula: Cn. H 2 n-2 On-1 – Eg. Lactose, Maltose, Isomaltose, Sucrose, Trehalose, etc. » Sources:

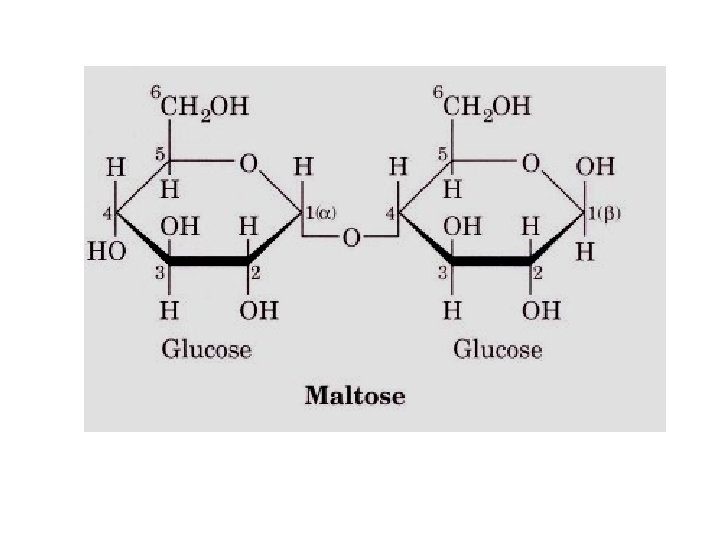
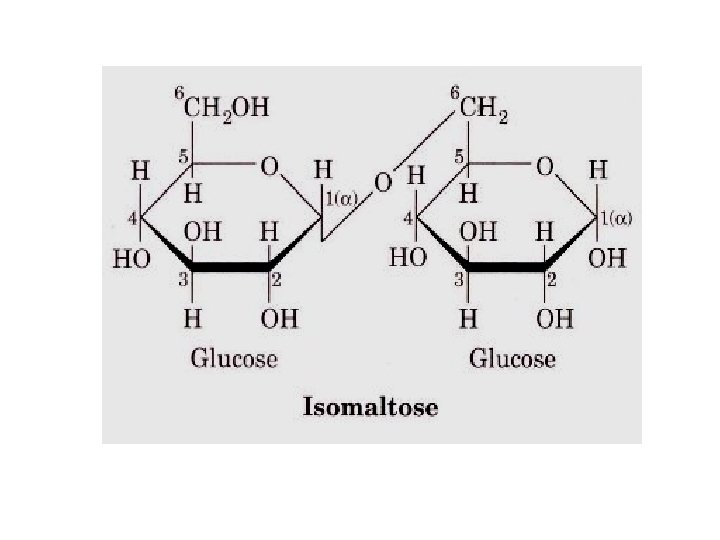

Classified further into • a. Reducing disaccharides • b. Can donate an electron • Eg. Lactose, Maltose, Isomaltose etc. Non-reducing disaccharides – Cannot donate an electron » Eg. Sucrose, Trehalose

3. Oligosaccharides

– Yield 3 -10 monosaccharide units upon hydrolysis • Tri, tetra, penta saccharides & so on » Eg. Maltotriose, Raffinose etc.

• Raffinose is a trisaccharide composed of Galactose, Fructose and Glucose • found in beans, cabbage, Brussels sprouts, broccoli, asparagus, other vegetables, and whole grains. » Raffinose : hydrolyzed to galactose and sucrose by αgalactosidase (not found in humans)

4. Polysaccharides

– Contains more than 10 monosaccharide units joined by glycosidic bonds – Generally high molecular weight molecules – Made up of Glu, Gal, Fructose, modified or derived monosaccharides etc.

• Tasteless – Eg. • Starch, Glycogen, Dextrin, Dextran, Inulin, Chitin • Heparin, Hyaluronic acid, Dermatan sulphate etc.


• Further classified into

1. Homopolysaccharides: • Starch, Glycogen, Dextrin, Dextran, Inulin, Cellulose, Chitin etc.

2. Heteropolysaccharides • Hyaluronic acid, Heparin, Chondroitin sulfate, Dermatan sulfate, Keratan sulfate etc.

Isomerism of monosaccharides

Isomers • Compounds which have the same structural/molecular formulae but differ in spatial configuration

– Optical isomerism (dextro, + (d) & levo, - (l) – Enantiomers- Mirror images of each other (D & L forms) – Aldose-Ketose isomerism – Epimerism – Anomerism (α & β)

Epimers

• Epimers are stereoisomers, which are different from one another only in configuration with respect to a specific single carbon atom

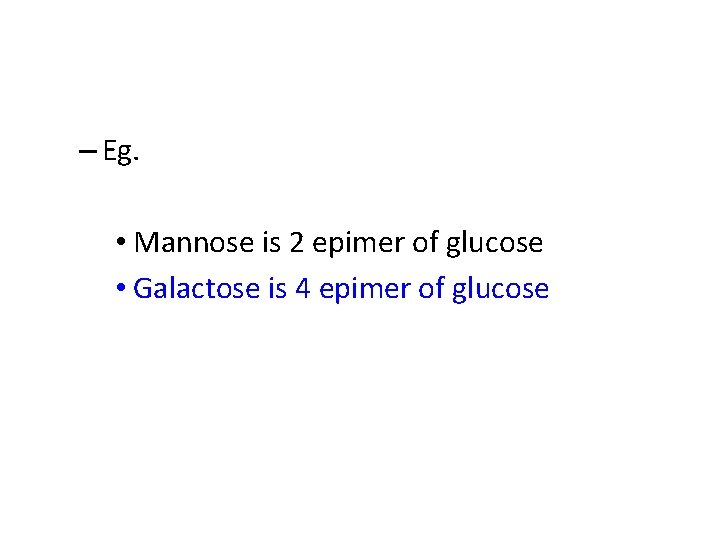
– Eg. • Mannose is 2 epimer of glucose • Galactose is 4 epimer of glucose

Disaccharides

• MALTOSE: • two glucose units are held by α-1, 4 glycosidic bond – Maltose is a reducing sugar

• Sources: – hydrolysis of starch by amylase, germinating seeds and malt • Gives sunflower phenylhydrazine shaped crystals with

• Lactose : sugar found in milk – It is composed of β-glucose and β-galactose linked by β-1, 4 glycosidic bond – A reducing sugar--> forms hedge-hog shaped crystals with phenylhydrazine

• Isomaltose : a disaccharide produced during the digestion of starch by amylase – It has α-1, 6 glycosidic bond between two glucose units – It is digested by enzyme isomaltase (1, 6 glucosidase)

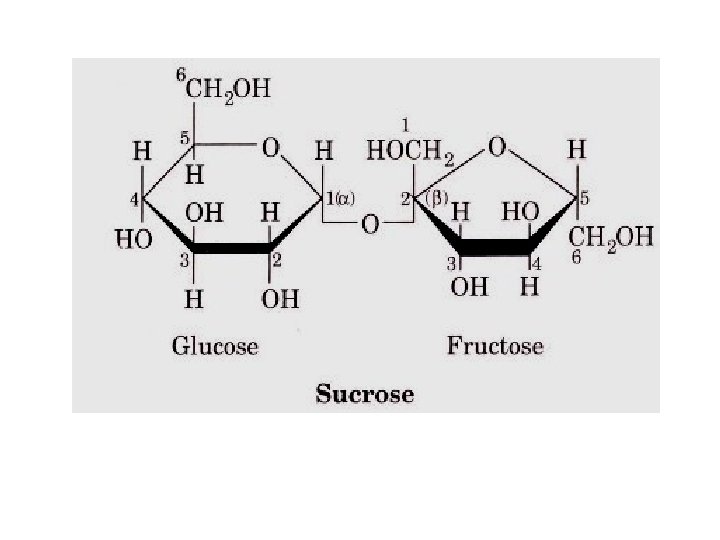
• Sucrose: • Sucrose is made up of α-D-glucose and β-Dfructose – linkage between C 1 of α-glucose and C 2 of β-fructose (α 1 --> β 2 ) – Hence sucrose is non reducing sugar – Present in cane sugar, beet sugar, fruits etc.

Oligosaccharides • Maltotriose : – made up of 3 molecules of glucose units – linked by α-1, 4 glycosidic bonds – produced during hydrolysis of starch
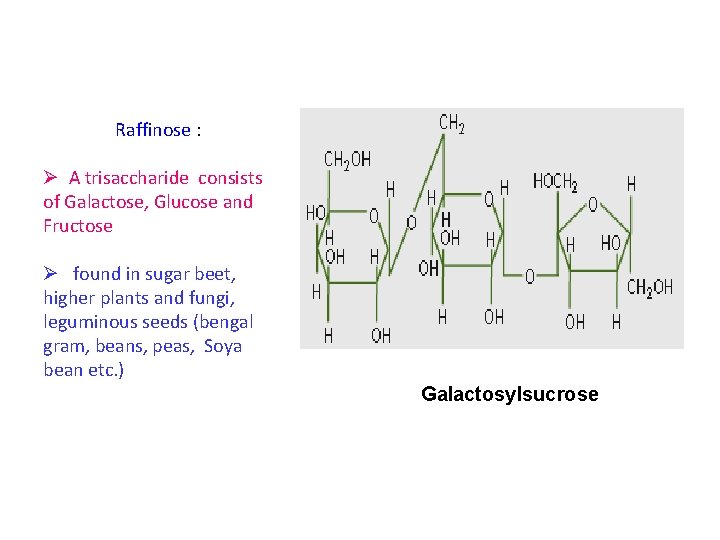
Raffinose : Ø A trisaccharide consists of Galactose, Glucose and Fructose Ø found in sugar beet, higher plants and fungi, leguminous seeds (bengal gram, beans, peas, Soya bean etc. ) Galactosylsucrose

Polysaccharides

• Macromolecules, high molecular weight • Polysaccharides -->consists of more than 10 monosaccharides • 100 to millions of monosaccharides in the chain

– Linear or branched & both

– Serve two important purposes 1. Storage (reserve food in both plant and animals): eg 2. Structural - » forms insoluble structural components like cell wall, membranes, cell coat and extracellular matrix » Cellulose, chitin etc. – Generally non-reducing in nature

Homopolysaccharide • Made up of – Only one type of monosaccharide units or – one type of monosaccharide derivative

– Glucans • Eg. – Starch, glycogen, cellulose – Chitin (N-acetyl glucosamine) – Fructosan • Eg. Inulin

Starch

– Source : potato, root tubers, rice, cereals, other food grains – glucose units are held by α-1, 4 and α-1, 6 bonds

• It is made up of two components 1. Amylose – water soluble, – contributes about 15 -20% 2. Amylopectin – water insoluble – contributes about 80 - 85%;

Amylose

• contains 200 -1000 glucose units • linked by α-1, 4 • Helical, unbranched chain • MW ranges 10 -100 KDa

– Gives blue color with iodine – Heating disrupts the coils and blue color disappears – On cooling blue color reappears

Amylopectin

• branched polysaccharide, has both α-1, 4 & α 1, 6 linkages (@ branch point) • Contains 3 -6 thousands of glucose units, and looks like a branched tree • MW ranges between 50 -1000 KDa

• Branches composed of about 25 -30 glucose units • Absorbs water and swells to form a paste • It gives violet / purple color with iodine

Dextrins

• Obtained by partial hydrolysis of starch • Shows some reducing property compared to starch • Low MW compared to amylopectin of starch – Also present in honey, toasted bread, plant leaves etc.

Glycogen

• Animal starch, highly branched polysaccharide • Both α 1 4 and α 1 6 Glycosidic bonds • High molecular weight : 1000 -5000 KDa • About 6000 -30, 000 glucose units • Water insoluble
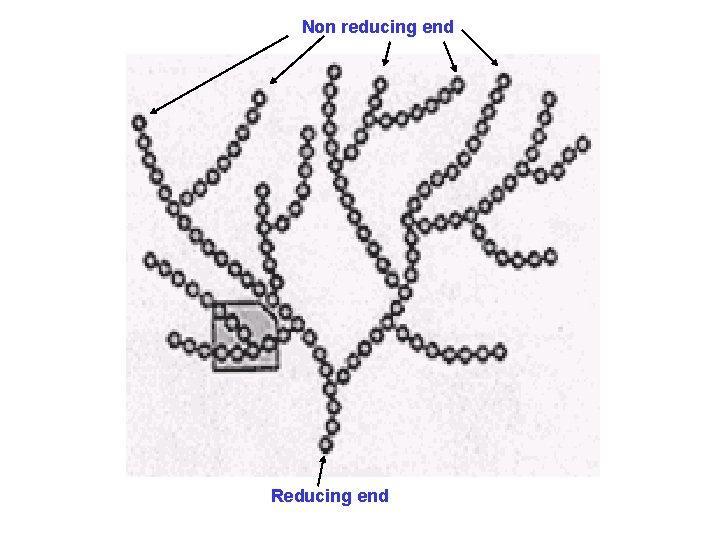
Non reducing end Reducing end

• Glycogen is profusely branched, (more branches than amylopectin) • Branches contains 10 -15 residues of glucose • Gives violet / purple color with iodine

Cellulose
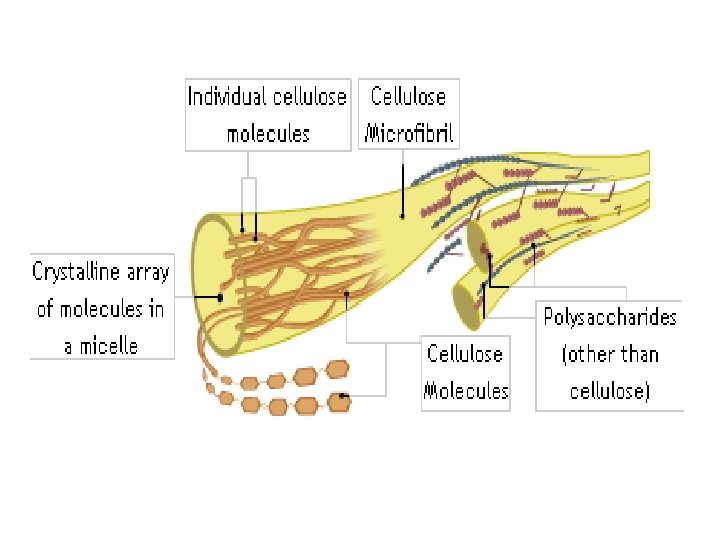

• It is the most abundant polysaccharide in the plant cell wall – Eg. Cotton fibers, wood etc. • unbranched, linked by β-1, 4 linkages

• In ruminants, herbivorous animals certain microorganisms in the gut provides the enzyme for digestion of cellulose • In humans, it provides roughage and adds bulk to the feces

• Cellulose, hemicellulose and other fibers in our diet has many beneficial effects – Holds lots of water molecules and helps in easy passage of stool – Increases peristaltic movement in the bowel – Increases bile acid & cholesterol excretion – Prevents intestinal diverticulosis, colon cancer etc – Has satiety value & Improves glucose tolerance

Dextran

• A complex, branched polysaccharide of glucose • MW varies from 3 -2000 Kda • The straight chain consists of α-1, 6 bond • Branches begin with α-1, 3 linkage

Used during microsurgery to decrease vascular thrombosis (antithrombotic , anti-platelet) Dextrans are potent osmotic agents, used to treat hypovolemia (as plasma volume expander)

Used to reduce blood viscosity • It is used in some eye drops as lubricant • Used in intravenous fluid to solubilize other factors: eg. Iron

Heteropolysaccharides

• Also called mucopolysaccharides, glycosaminoglycans • Sulfate groups and carboxyl groups contributes to acidic nature of molecules (acid mucopolysaccharides)

• made up of more than one type of monosaccharide units or their derivatives • Eg. • Glycosaminoglycans: » » » Hyaluronic acid, Heparin, Chondroitin sulfate, Dermatan sulfate, Keratan sulfate

• The extracellular space of tissues (CT-cartilage, skin, blood vessels, tendons etc. ) consists of collagen and elastin fibers embedded in a matrix or ground substance – The ground substance is predominantly composed of GAG’s

1. Hyaluronic acid • Composition: • Made up of repeating units of N-Acetyl Glucosamine and Glucuronic acid • Not sulfated

• Significance: – Present in synovial fluid, vitreous humor, umbilical cord, skin, cartilage and loose connective tissue – Acts as lubricant, shock absorber etc.

2. Chondroitin sulfate • Composition: – Made up of- repeating units of N-Acetyl Galactosamine and Glucuronic acid – sulfated on C-4 and C-6 of N-Ac-Galactosamine

– Significance: • Found in cartilages, bone, cornea, tendon, ligaments etc. • In cartilage bind collagen and hold fibers in a tight strong network

3. Keratan sulfate • Composition: – Made up of repeating units of N-Acetyl. Glucosamine & Galactose; C-6 of either sugar may be sulfated – No uronic acids • Significance : Found in connective tissue of skin, cartilage, cornea, intervertebral discs etc.

4. Heparin • Composition: – made up of repeating units of Glucosamine (sulfated) & Iduronic acid (sulfated or non sulfated) • & small amounts of N-Ac-Glucosamine & Glucuronic acid

• Significance: – Found in- mast cells that lines arteries in skin, liver and intestinal mucosa – Natural anticoagulant due to its antithrombin activity – prevents intravascular blood coagulation

5. Dermatan sulfate • Composition: – It is made up of repeating units of N-acetyl galactosamine and iduronic acid; sulfated • Significane: – Widely distributed in- skin, arterial walls etc.

Glycoproteins and Proteoglycans
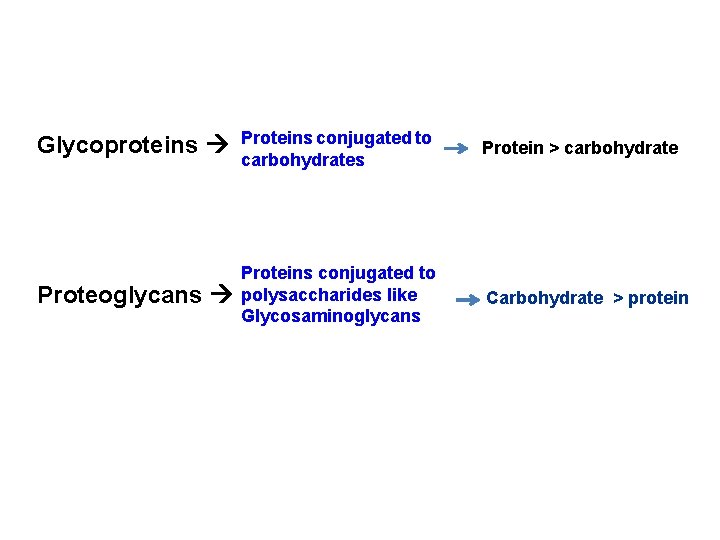
Glycoproteins Proteins conjugated to carbohydrates Protein > carbohydrate Proteoglycans Proteins conjugated to polysaccharides like Glycosaminoglycans Carbohydrate > protein

Functions of proteoglycans (GAG’s)

1. Bind to large amounts of cations and water produce a gel like matrix § Gel like matrix is responsible for high viscosity, elasticity and lubricating property 2. Acts as lubricant and shock absorber synovial fluid

3. Maintain water-electrolyte balance 4. Restrict movement of large particles 5. An anticoagulant- e. g. heparin 6. Support cellular and fibrous components of tissues 7. Mediate cell to cell interaction

Functions of glycoproteins

1. They serve as biological labels for binding of certain proteins 2. Cell to cell recognition and adhesion
3. Govern the rate of degradation of certain hormones, proteins and blood cells 4. Mediate the flow of information between two cells or cell and extracellular matrix

Blood group substances Blood group antigenic determinants contains carbohydrates

- Slides: 115