Carbohydrates Andy Howard Biochemistry Lectures Spring 2019 Tuesday

Carbohydrates Andy Howard Biochemistry Lectures, Spring 2019 Tuesday 26 March 2019 42 Mo

Carbohydrates are more than just energy sources n n n We’ll introduce names carbohydrate monomers and oligomers Carbohydrates can be cyclic or derivatized, and they can be oligomerized and polymerized. Glycoconjugates are complexes involving sugar and peptide oligomers or polymers 03/26/2019 Carbohydrates p. 2 of 75

What we’ll discuss n Carbohydrates n n n n Names & depictions Cyclic Forms Sugar Derivatives Acetals & ketals Glycosides Oligosaccharides 03/26/2019 Carbohydrates Polysaccharides n n n Storage Structural Glycoconjugates n n n Proteoglycans Peptidoglycans Glycoproteins p. 3 of 75
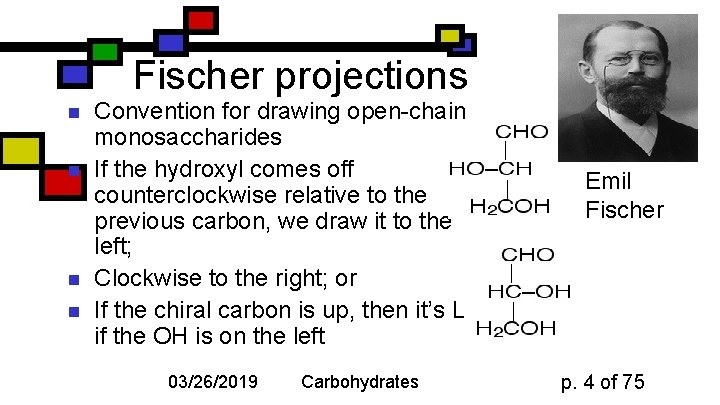
Fischer projections n n Convention for drawing open-chain monosaccharides If the hydroxyl comes off counterclockwise relative to the previous carbon, we draw it to the left; Clockwise to the right; or If the chiral carbon is up, then it’s L if the OH is on the left 03/26/2019 Carbohydrates Emil Fischer p. 4 of 75

Cyclic sugars n n n Aldoses with ≥ 4 carbons & ketoses with ≥ 5 carbons can readily interconvert between the open-chain forms we have drawn and fivemembered(furanose) or six-membered (pyranose) ring forms in which the carbonyl oxygen becomes part of the ring Remember that 5 - and 6 -atom rings form with little or no strain; 4 - and 3 - are much harder There are no C=O bonds in the ring forms 03/26/2019 Carbohydrates p. 5 of 75
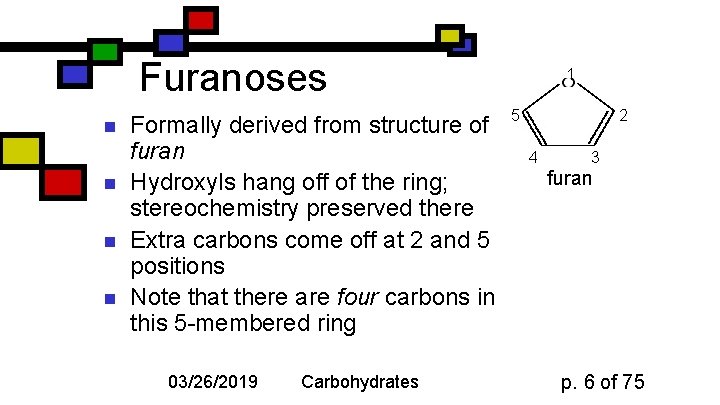
Furanoses n n Formally derived from structure of furan Hydroxyls hang off of the ring; stereochemistry preserved there Extra carbons come off at 2 and 5 positions Note that there are four carbons in this 5 -membered ring 03/26/2019 Carbohydrates 1 5 2 4 3 furan p. 6 of 75
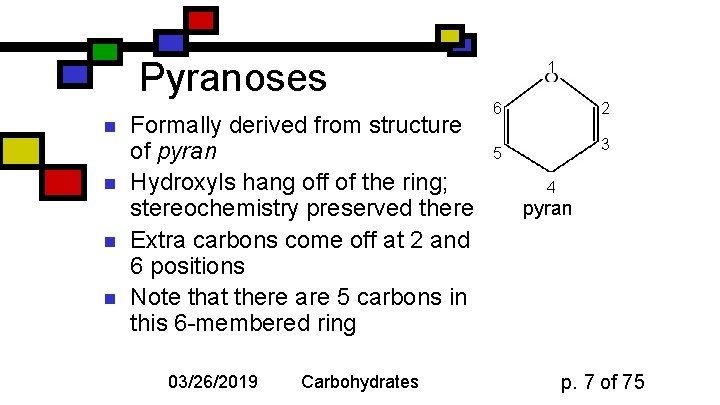
Pyranoses n n Formally derived from structure of pyran Hydroxyls hang off of the ring; stereochemistry preserved there Extra carbons come off at 2 and 6 positions Note that there are 5 carbons in this 6 -membered ring 03/26/2019 Carbohydrates 1 6 2 3 5 4 pyran p. 7 of 75
How do we cyclize a sugar? n n n Formation of an internal hemiacetal or hemiketal (see a few slides from here) by conversion of the terminal hydroxyl to a ring oxygen while the carbonyl becomes a hydroxyl Not a net oxidation or reduction; in fact it’s a true isomerization. The molecular formula for the cyclized form is the same as the open chain form 03/26/2019 Carbohydrates p. 8 of 75
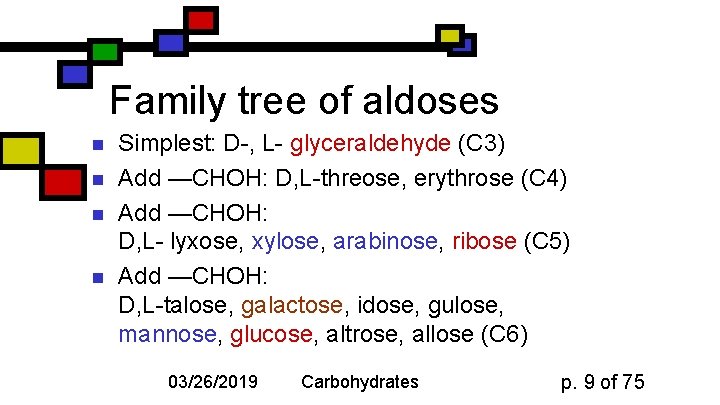
Family tree of aldoses n n Simplest: D-, L- glyceraldehyde (C 3) Add —CHOH: D, L-threose, erythrose (C 4) Add —CHOH: D, L- lyxose, xylose, arabinose, ribose (C 5) Add —CHOH: D, L-talose, galactose, idose, gulose, mannose, glucose, altrose, allose (C 6) 03/26/2019 Carbohydrates p. 9 of 75
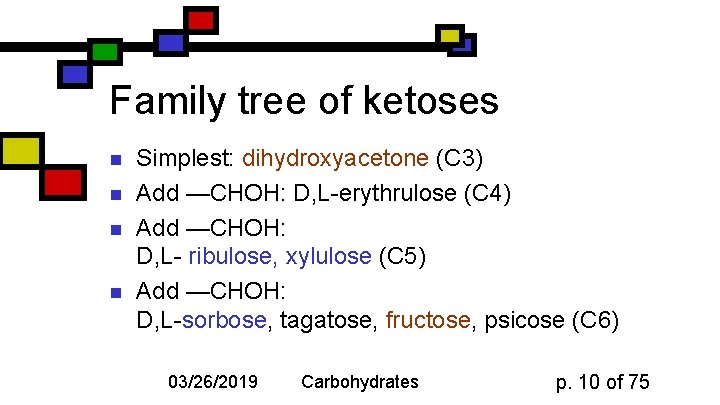
Family tree of ketoses n n Simplest: dihydroxyacetone (C 3) Add —CHOH: D, L-erythrulose (C 4) Add —CHOH: D, L- ribulose, xylulose (C 5) Add —CHOH: D, L-sorbose, tagatose, fructose, psicose (C 6) 03/26/2019 Carbohydrates p. 10 of 75

Relative significance (? ) Glucose, fructose, galactose, mannose, ribose, glyceraldehyde, xylose, arabinose 03/26/2019 Carbohydrates p. 11 of 75
Haworth projections n …provide a way of keeping track the chiral centers in a cyclic sugar, as the Fischer projections enable for straight-chain sugars (cf. fig. 16. 9) 03/26/2019 Carbohydrates Sir Walter Haworth p. 12 of 75

The anomeric carbon n n In any cyclic sugar (monosaccharide, or single unit of an oligosaccharide, or polysaccharide) there is one carbon that has covalent bonds to two different oxygen atoms We describe this carbon as the anomeric carbon 03/26/2019 Carbohydrates O C O p. 13 of 75
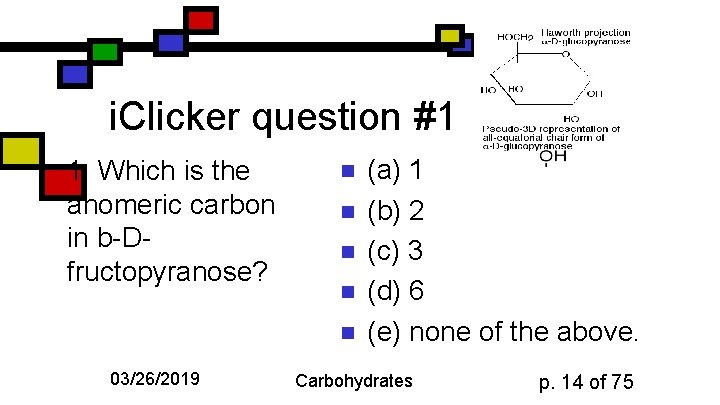
i. Clicker question #1 1. Which is the anomeric carbon in b-Dfructopyranose? n n n 03/26/2019 (a) 1 (b) 2 (c) 3 (d) 6 (e) none of the above. Carbohydrates p. 14 of 75
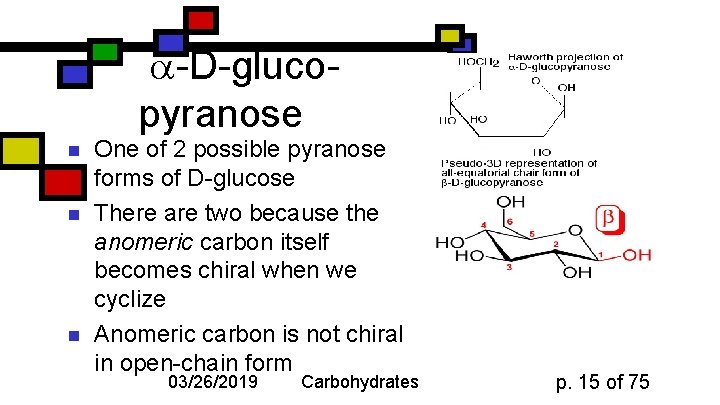
-D-glucopyranose n n n One of 2 possible pyranose forms of D-glucose There are two because the anomeric carbon itself becomes chiral when we cyclize Anomeric carbon is not chiral in open-chain form 03/26/2019 Carbohydrates p. 15 of 75
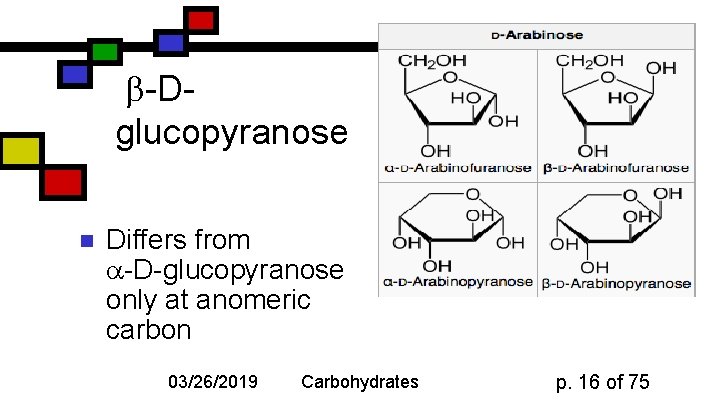
-Dglucopyranose n Differs from -D-glucopyranose only at anomeric carbon 03/26/2019 Carbohydrates p. 16 of 75

Why is glucose special? n n All aldohexoses are fairly similar Glucose is the only aldohexose that, when built into the pyranose form, can be drawn with all its bulky substituents (–OH, –CH 2 OH) in equatorial positions This minimizes steric clashes This matters enough that some hexoses are actually marginally more stable as furanoses, e. g. fructose 03/26/2019 Carbohydrates p. 17 of 75

Count carefully! n n n It’s tempting to think that hexoses are pyranoses and pentoses are furanoses; But that’s not always true The ring always contains an oxygen, so even a pentose can form a pyranose In solution: pyranose, furanose, open-chain forms are all present Percentages depend on the sugar 03/26/2019 Carbohydrates p. 18 of 75
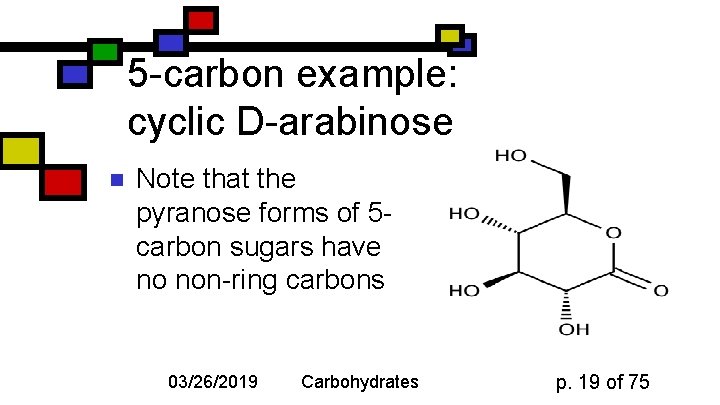
5 -carbon example: cyclic D-arabinose n Note that the pyranose forms of 5 carbon sugars have no non-ring carbons 03/26/2019 Carbohydrates p. 19 of 75
Substituted monosaccharides (cf. § 16. 2) n n Substitutions on the various positions retain some sugar-like character Some substituted monosaccharides are building blocks of polysaccharides 03/26/2019 Carbohydrates p. 20 of 75
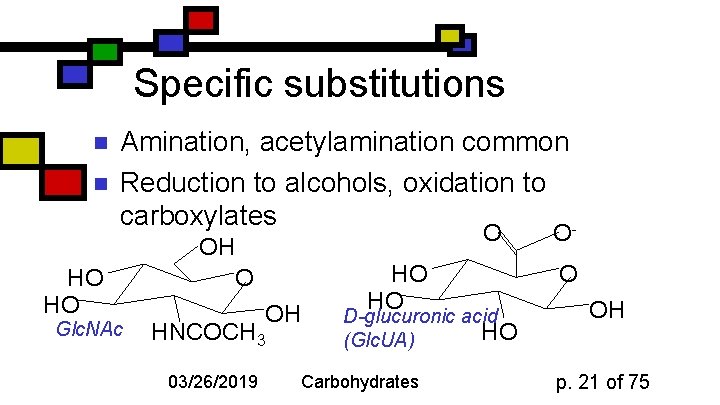
Specific substitutions n n Amination, acetylamination common Reduction to alcohols, oxidation to carboxylates O OH HO HO Glc. NAc O OH HNCOCH 3 03/26/2019 HO HO D-glucuronic acid HO (Glc. UA) Carbohydrates O O OH p. 21 of 75
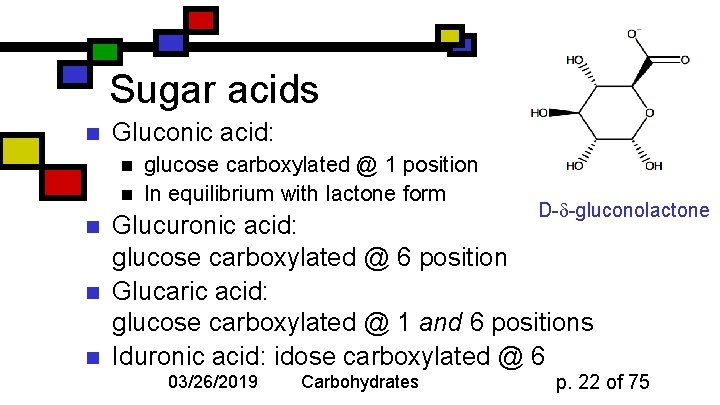
Sugar acids n Gluconic acid: n n n glucose carboxylated @ 1 position In equilibrium with lactone form D- -gluconolactone Glucuronic acid: glucose carboxylated @ 6 position Glucaric acid: glucose carboxylated @ 1 and 6 positions Iduronic acid: idose carboxylated @ 6 03/26/2019 Carbohydrates p. 22 of 75
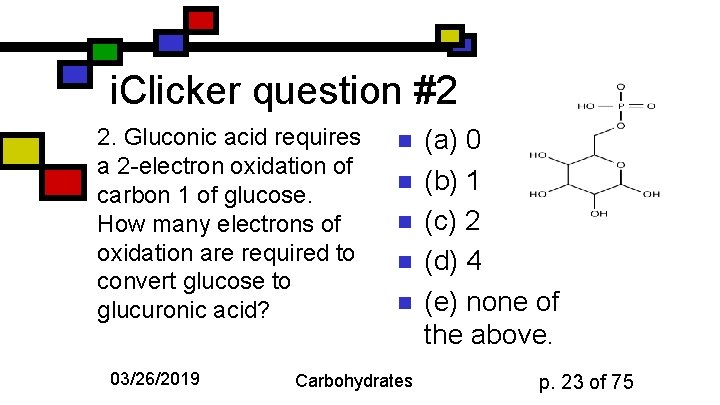
i. Clicker question #2 2. Gluconic acid requires a 2 -electron oxidation of carbon 1 of glucose. How many electrons of oxidation are required to convert glucose to glucuronic acid? 03/26/2019 n n n Carbohydrates (a) 0 (b) 1 (c) 2 (d) 4 (e) none of the above. p. 23 of 75

Sugar alcohols n n n Mild reduction of sugars convert aldehyde or ketone moiety to alcohol Generates an additional asymmetric center in ketoses with > 3 carbons These remain in open-chain forms Smallest: glycerol Sorbitol, myo-inositol, ribitol are important 03/26/2019 Carbohydrates p. 24 of 75
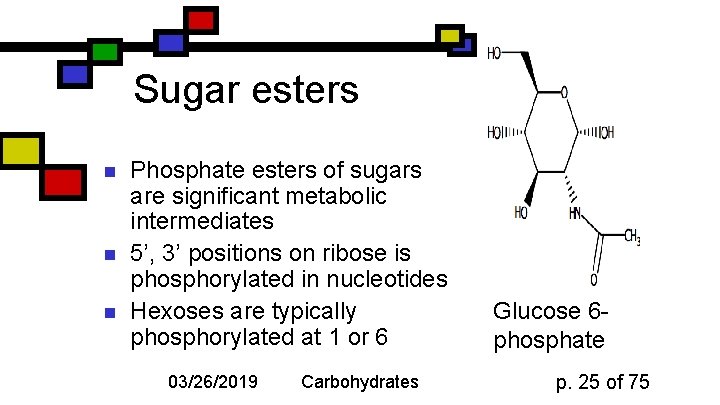
Sugar esters n n n Phosphate esters of sugars are significant metabolic intermediates 5’, 3’ positions on ribose is phosphorylated in nucleotides Hexoses are typically phosphorylated at 1 or 6 03/26/2019 Carbohydrates Glucose 6 phosphate p. 25 of 75
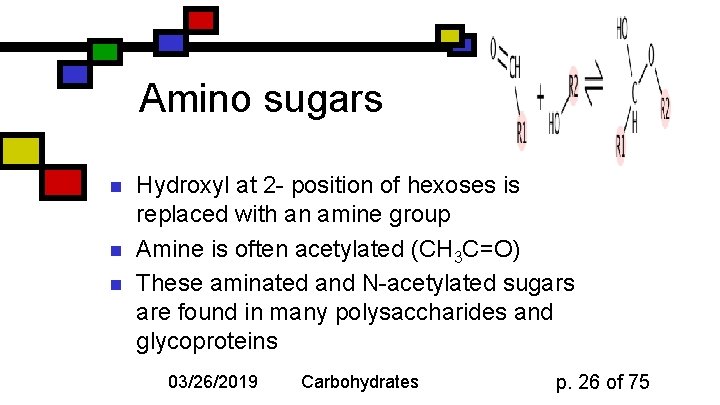
Amino sugars n n n Hydroxyl at 2 - position of hexoses is replaced with an amine group Amine is often acetylated (CH 3 C=O) These aminated and N-acetylated sugars are found in many polysaccharides and glycoproteins 03/26/2019 Carbohydrates p. 26 of 75
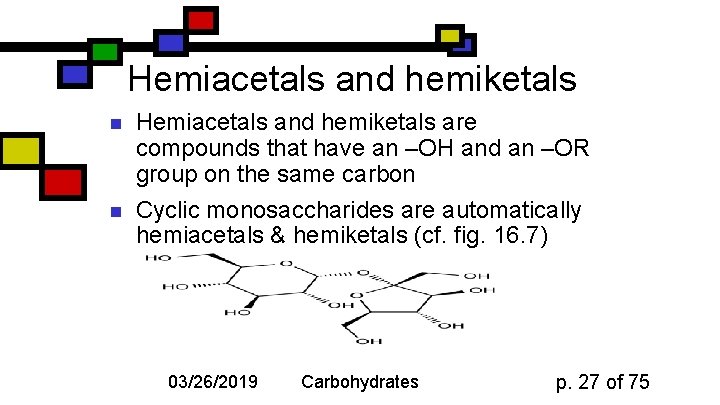
Hemiacetals and hemiketals n n Hemiacetals and hemiketals are compounds that have an –OH and an –OR group on the same carbon Cyclic monosaccharides are automatically hemiacetals & hemiketals (cf. fig. 16. 7) 03/26/2019 Carbohydrates p. 27 of 75
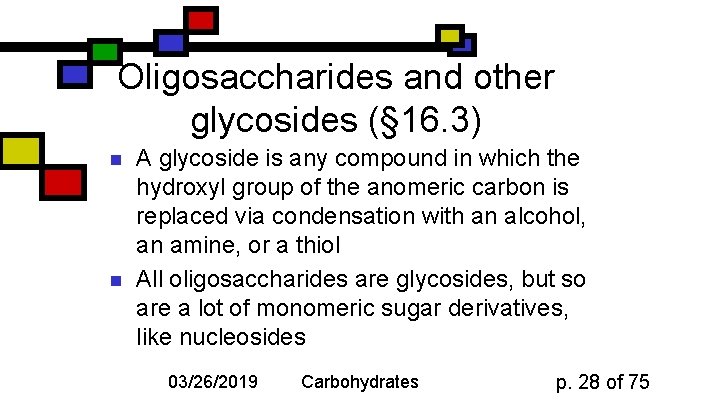
Oligosaccharides and other glycosides (§ 16. 3) n n A glycoside is any compound in which the hydroxyl group of the anomeric carbon is replaced via condensation with an alcohol, an amine, or a thiol All oligosaccharides are glycosides, but so are a lot of monomeric sugar derivatives, like nucleosides 03/26/2019 Carbohydrates p. 28 of 75
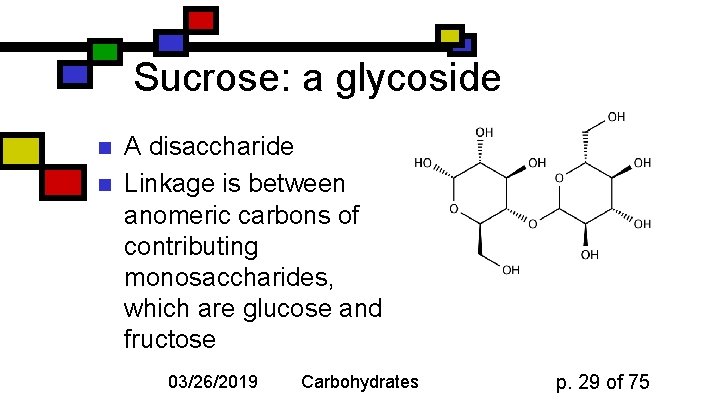
Sucrose: a glycoside n n A disaccharide Linkage is between anomeric carbons of contributing monosaccharides, which are glucose and fructose 03/26/2019 Carbohydrates p. 29 of 75
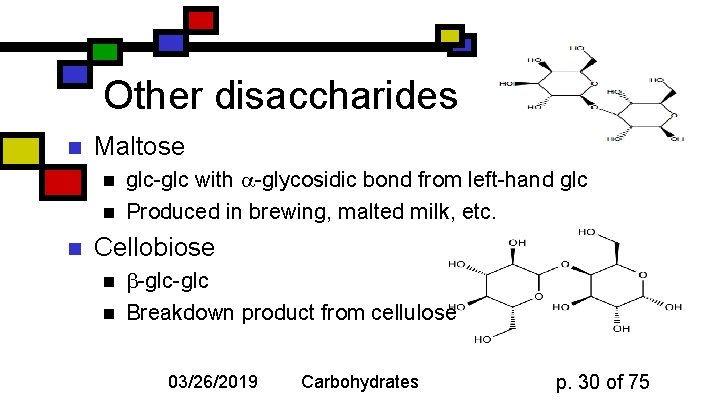
Other disaccharides n Maltose n n n glc-glc with -glycosidic bond from left-hand glc Produced in brewing, malted milk, etc. Cellobiose n n -glc Breakdown product from cellulose 03/26/2019 Carbohydrates p. 30 of 75

Lactose: another disaccharide n Lactose: -gal-glc n n Milk sugar Lactose intolerance caused by absence of enzyme capable of hydrolyzing this glycoside 03/26/2019 Carbohydrates p. 31 of 75

Reducing sugars n n Sugars that can undergo ring-opening to form the open-chain aldehyde compounds that can be oxidized to carboxylic acids We describe those as reducing sugars because they can reduce metal ions or amino acids in the presence of base 03/26/2019 Carbohydrates p. 32 of 75
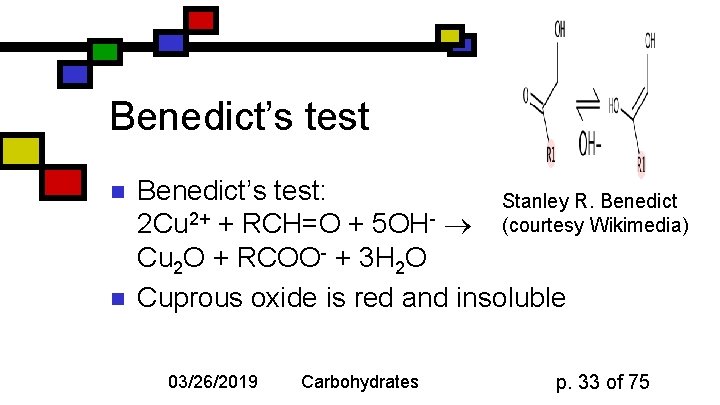
Benedict’s test n n Benedict’s test: Stanley R. Benedict 2 Cu 2+ + RCH=O + 5 OH- (courtesy Wikimedia) Cu 2 O + RCOO- + 3 H 2 O Cuprous oxide is red and insoluble 03/26/2019 Carbohydrates p. 33 of 75
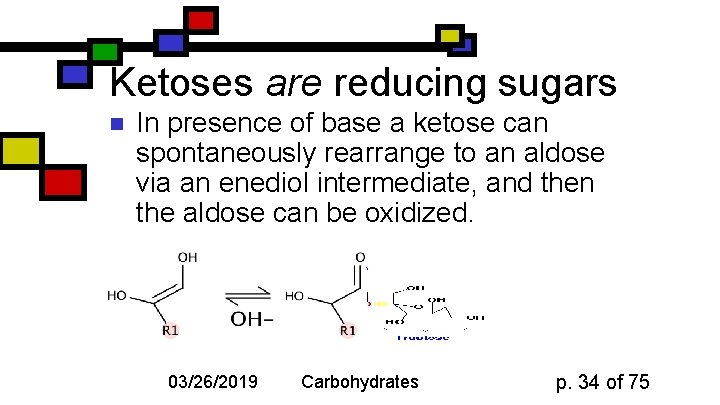
Ketoses are reducing sugars n In presence of base a ketose can spontaneously rearrange to an aldose via an enediol intermediate, and then the aldose can be oxidized. 03/26/2019 Carbohydrates p. 34 of 75
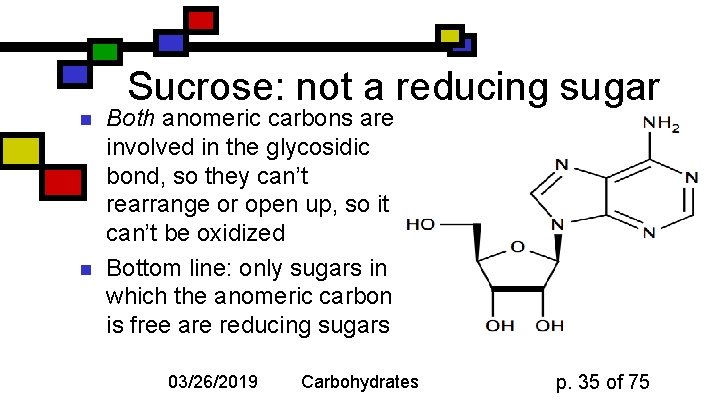
Sucrose: not a reducing sugar n n Both anomeric carbons are involved in the glycosidic bond, so they can’t rearrange or open up, so it can’t be oxidized Bottom line: only sugars in which the anomeric carbon is free are reducing sugars 03/26/2019 Carbohydrates p. 35 of 75

Why does this matter? n n Partly historical: this cuprate reaction was one of the first well-characterized tools for characterizing these otherwise very similar compounds But it also gives us a convenient way of distinguishing among types of glycosidic arrangements, even if we never really use Cu 2+ ions in experiments 03/26/2019 Carbohydrates p. 36 of 75

Reducing & nonreducing ends n n Typically, oligo and polysaccharides have a reducing end a nonreducing end Non-reducing end is the sugar moiety whose anomeric carbon is involved in the glycosidic bond Reducing end is sugar whose anomeric carbon is free to open up and oxidize Enzymatic lengthening and degradation of polysaccharides occurs at nonreducing end or ends 03/26/2019 Carbohydrates p. 37 of 75
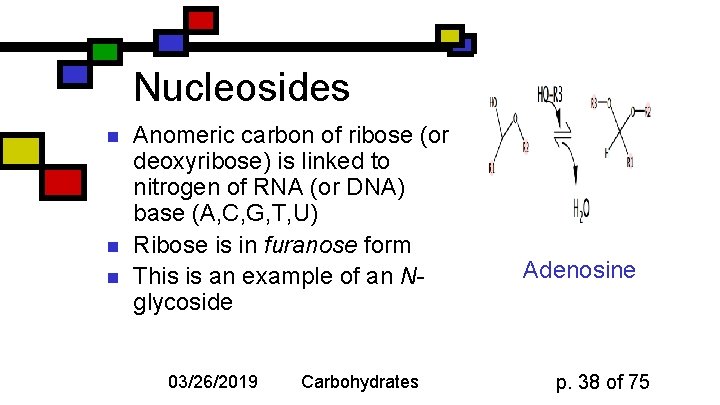
Nucleosides n n n Anomeric carbon of ribose (or deoxyribose) is linked to nitrogen of RNA (or DNA) base (A, C, G, T, U) Ribose is in furanose form This is an example of an Nglycoside 03/26/2019 Carbohydrates Adenosine p. 38 of 75
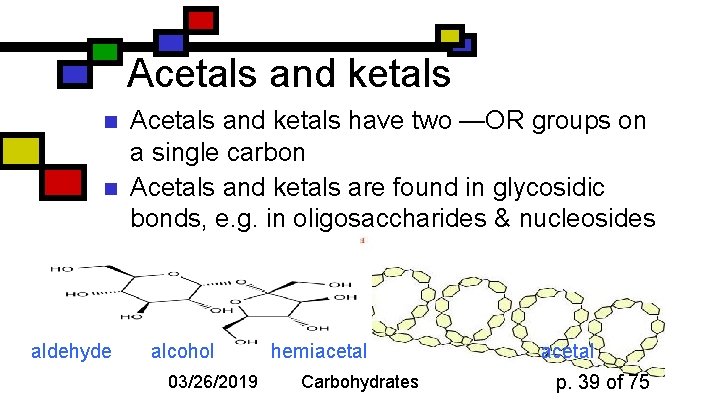
Acetals and ketals n n aldehyde Acetals and ketals have two —OR groups on a single carbon Acetals and ketals are found in glycosidic bonds, e. g. in oligosaccharides & nucleosides alcohol 03/26/2019 hemiacetal Carbohydrates acetal p. 39 of 75

Polysaccharides (§ 16. 4) n n n Homoglycans: all building blocks same Heteroglycans: more than one kind of building block No equivalent of genetic code for carbohydrates, so long ones will be heterogeneous in length and branching, and maybe even in monomer identity 03/26/2019 Carbohydrates p. 40 of 75
Polysaccharides: homoglycans n Storage homoglycans (all monomers are glucose (Glc)) n n n Starch: amylose ( (1 4)Glc) , amylopectin Glycogen Structural homoglycans n n n Cellulose ( (1 4)Glc) Chitin ( (1 4)Glc. NAc) Others 03/26/2019 Carbohydrates p. 41 of 75

Polysaccharides: heteroglycans Found in glycosaminoglycans (disaccharide units) n Hyaluronic acid: Glc. UA, Glc. NAc) ( (1 3, 4)) n Glycosylations of proteins n 03/26/2019 Carbohydrates p. 42 of 75

Storage polysaccharides n n Sources of glucose for energy and carbon Long-chain polymers of glucose n Starch (amylose and amylopectin) n n in plants, it’s stored in 3 -100 µm granules Typically 70% amylopectin, but it varies with the plant from which it’s taken Glycogen: animals, some bacteria Branches found in all but amylose 03/26/2019 Carbohydrates p. 43 of 75
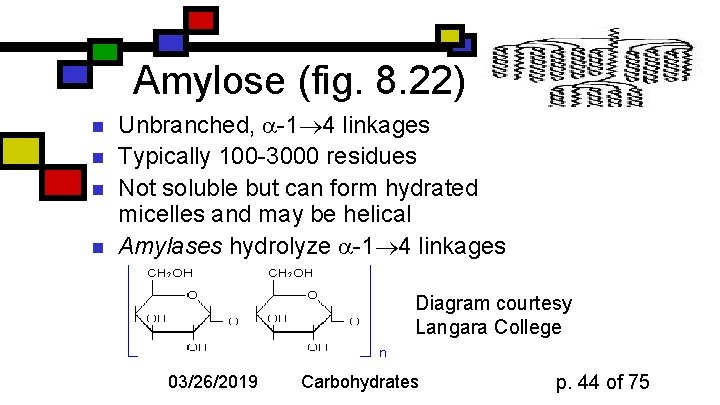
Amylose (fig. 8. 22) n n Unbranched, -1 4 linkages Typically 100 -3000 residues Not soluble but can form hydrated micelles and may be helical Amylases hydrolyze -1 4 linkages Diagram courtesy Langara College 03/26/2019 Carbohydrates p. 44 of 75

Amylopectin n n Mostly -1 4 linkages; 4% -1 6 Each sidechain has 15 -25 glucose moieties -1 6 linkages broken down by debranching enzymes Usually 300 -50000 total glucose units per amylopectin molecule One reducing end, many nonreducing ends 03/26/2019 Carbohydrates p. 45 of 75

Glycogen n n Principal storage form of glucose in human liver; some in muscle Branched ( -1 4 + a few -1 6) More branches (~10%) Often larger than starch: 50000 glucose One reducing end, many nonreducing ends 03/26/2019 Carbohydrates p. 46 of 75
Preview: glycogen metabolism n n Broken down to G-1 -P units Built up from G-6 -P G-1 -P UDP-Glucose units 03/26/2019 Carbohydrates p. 47 of 75
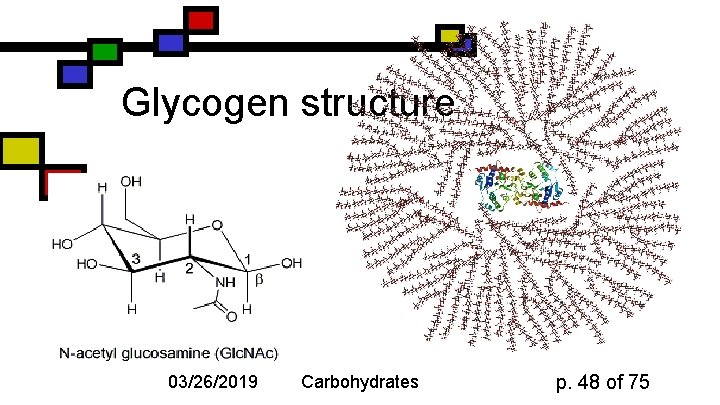
Glycogen structure 03/26/2019 Carbohydrates p. 48 of 75
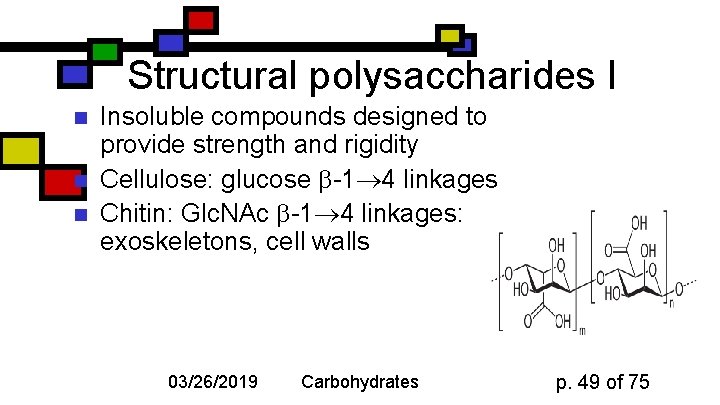
Structural polysaccharides I n n n Insoluble compounds designed to provide strength and rigidity Cellulose: glucose -1 4 linkages Chitin: Glc. NAc -1 4 linkages: exoskeletons, cell walls 03/26/2019 Carbohydrates p. 49 of 75

Cellulose: details n n n Rigid, flat structure: each glucose is upside down relative to its nearest neighbors 300 -15000 glucose units Found in plant cell walls Resistant to most glucosidases Cellulases found in termites, ruminant gut bacteria 03/26/2019 Carbohydrates p. 50 of 75
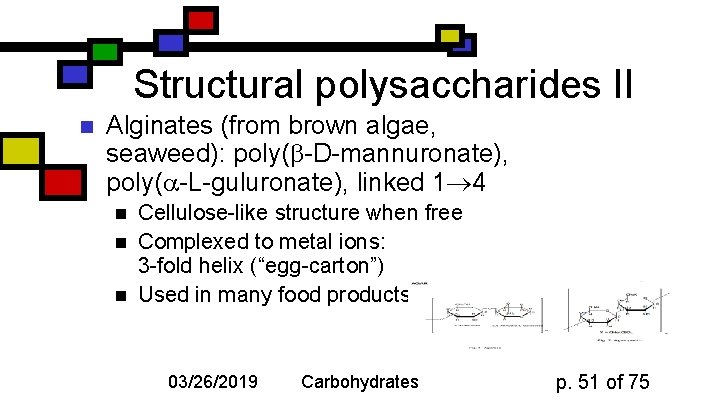
Structural polysaccharides II n Alginates (from brown algae, seaweed): poly( -D-mannuronate), poly( -L-guluronate), linked 1 4 n n n Cellulose-like structure when free Complexed to metal ions: 3 -fold helix (“egg-carton”) Used in many food products 03/26/2019 Carbohydrates p. 51 of 75
Structural polysaccharides III n Agarose: alternating D-galactose, 3, 6 -anhydro-L-gal, with some 6 -methyl-D-gal side chains n n Forms gels: hold huge amounts of H 2 O Can be processed to use in the lab for gel exclusion chromatography Found in red algae as part of agar, which also contains agaropectin Glycosaminoglycans: see next section 03/26/2019 Carbohydrates p. 52 of 75

Components of agar 03/26/2019 Carbohydrates p. 53 of 75

Sugar Complexes n n Sugars form complexes with proteins, peptides and lipids What we call these complexes depends on how long the saccharides are and how long the peptides are 03/26/2019 Carbohydrates p. 54 of 75
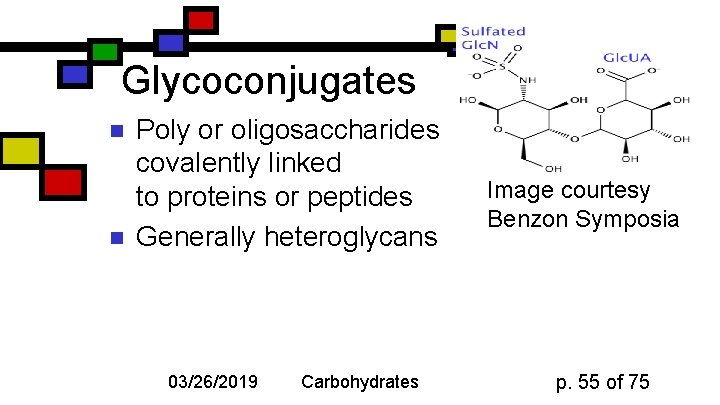
Glycoconjugates n n Poly or oligosaccharides covalently linked to proteins or peptides Generally heteroglycans 03/26/2019 Carbohydrates Image courtesy Benzon Symposia p. 55 of 75

Categories of glycoconjugates n n n Proteoglycans (protein+glycosaminoglycans) Peptidoglycans (peptide+polysaccharide) Glycoproteins (protein+oligosaccharide) 03/26/2019 Carbohydrates p. 56 of 75
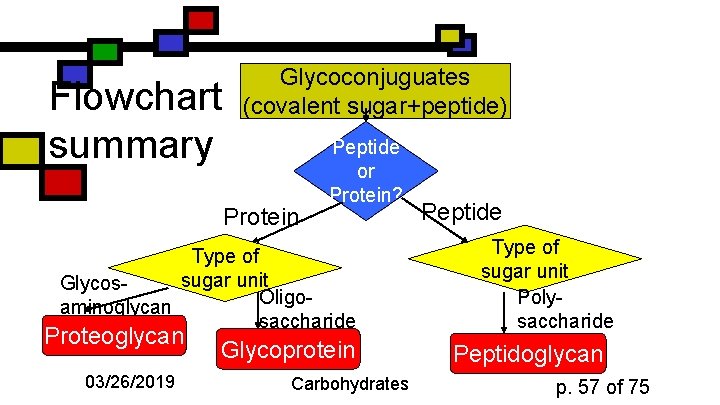
Flowchart summary Glycoconjuguates (covalent sugar+peptide) Protein Peptide or Protein? Type of sugar unit Glycos. Oligoaminoglycan saccharide Proteoglycan 03/26/2019 Glycoprotein Carbohydrates Peptide Type of sugar unit Polysaccharide Peptidoglycan p. 57 of 75

Proteoglycans: Glycosaminoglycans n n n Unbranched heteroglycans of repeating disaccharides One component is Gal. N, Glc. N, Gal. NAc, or Glc. NAc Other component: an alduronic acid like Glc. UA —OH or —NH 2 often sulfated Found in cartilage, joint fluid 03/26/2019 Carbohydrates p. 58 of 75
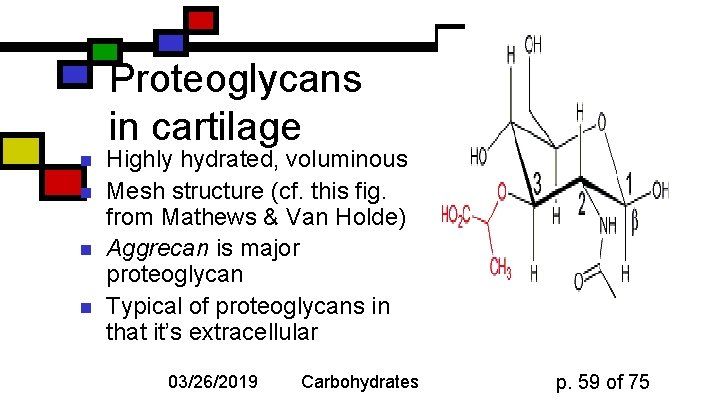
Proteoglycans in cartilage n n Highly hydrated, voluminous Mesh structure (cf. this fig. from Mathews & Van Holde) Aggrecan is major proteoglycan Typical of proteoglycans in that it’s extracellular 03/26/2019 Carbohydrates p. 59 of 75
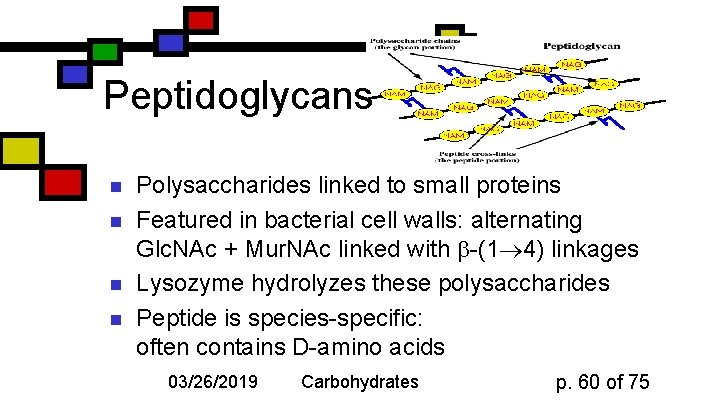
Peptidoglycans n n Polysaccharides linked to small proteins Featured in bacterial cell walls: alternating Glc. NAc + Mur. NAc linked with -(1 4) linkages Lysozyme hydrolyzes these polysaccharides Peptide is species-specific: often contains D-amino acids 03/26/2019 Carbohydrates p. 60 of 75

Peptidoglycans in bacteria n n n Gram-negative: thin peptidoglycan layer separates 2 phospholipid bilayer membranes Gram-positive: only one bilayer, with thicker peptidoglycan cell wall outside it Gram stain binds to thick wall, not thin layer 03/26/2019 Carbohydrates p. 61 of 75
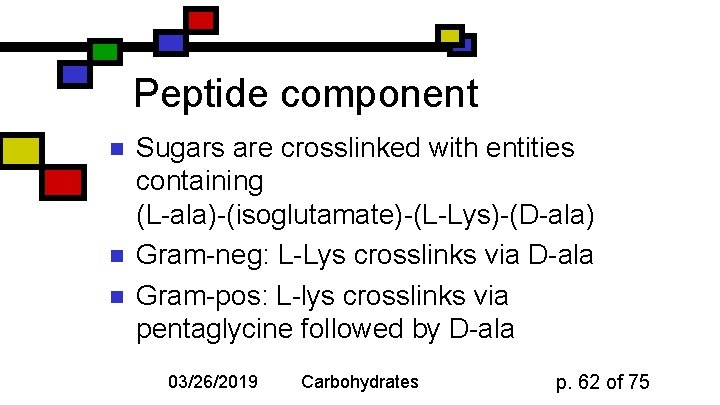
Peptide component n n n Sugars are crosslinked with entities containing (L-ala)-(isoglutamate)-(L-Lys)-(D-ala) Gram-neg: L-Lys crosslinks via D-ala Gram-pos: L-lys crosslinks via pentaglycine followed by D-ala 03/26/2019 Carbohydrates p. 62 of 75

Gram-negative bacteria: the periplasmic space n n n Periplasmic space: space between the 2 cell membranes, inside that peptidoglycan layer Peptidoglycan is attached to outer membrane via 57 -residue hydrophobic proteins Outer membrane has a set of lipopolysaccharides attached to it; these sway outward from membrane 03/26/2019 Carbohydrates p. 63 of 75
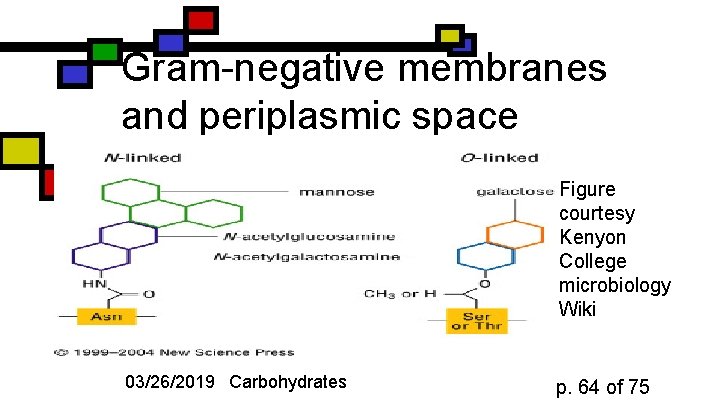
Gram-negative membranes and periplasmic space Figure courtesy Kenyon College microbiology Wiki 03/26/2019 Carbohydrates p. 64 of 75

Glycoproteins (§ 16. 5) n n n 1 -30 carbohydrate moieties per protein Proteins can be enzymes, hormones, structural proteins, transport proteins Microheterogeneity: same protein, different sugar combinations Eight sugars common in eukaryotes PTM glycosylation much more common in eukaryotes than prokaryotes 03/26/2019 Carbohydrates p. 65 of 75

i. Clicker question #3 3. Molecule Z consists of a 300 -amino acid polypeptide with three oligosaccharide units attached to it. Which type of glycoconjugate is this? 03/26/2019 n n (a) proteoglycan (b) peptidoglycan (c) glycoprotein (d) not enough information to tell Carbohydrates p. 66 of 75

Diversity in glycoproteins n n n Variety of sugar monomers or glycosidic linkages Linkages always at C-1 on one sugar but can be C-2, 3, 4, 6 on the other one Up to 4 branches But: not all the specific glycosyltransferases you would need to get all this diversity exist in any one organism 03/26/2019 Carbohydrates p. 67 of 75
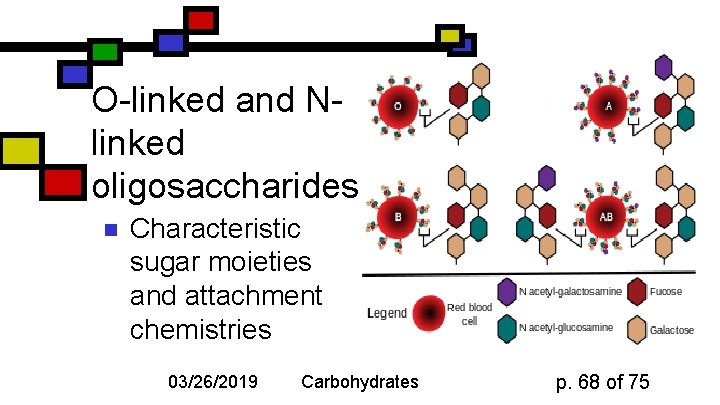
O-linked and Nlinked oligosaccharides n Characteristic sugar moieties and attachment chemistries 03/26/2019 Carbohydrates p. 68 of 75

O-linked oligosaccharides n n Gal. NAc to Ser or Thr; often with gal or sialic acid on Gal. NAc 5 -hydroxylysines on collagen are joined to D-Gal Some proteoglycans joined via Gal-Xyl-Ser Single Glc. NAc on ser or thr 03/26/2019 Carbohydrates p. 69 of 75

Human ABO Blood Groups n n n Based on H antigen: cell-surface oligosaccharide that links to a small protein (or to a ceramide) Invariant part of H antigen structure is Fuc-Gal. NAc-Gal Fucose is 6 -deoxygalactose 03/26/2019 Carbohydrates p. 70 of 75

ABO blood groups, continued n n A antigen attaches another Gal. NAc to the first Gal B antigen attaches another Gal to 1 st Gal 03/26/2019 Carbohydrates p. 71 of 75

Enzymology and Heritance n n Fucosyltransferase on chromosome 19 codes for invariant part Single gene on chr 9 codes for: n n A Gal. NAc transferase in the A case A Gal transferase in the B case A nonfunctional frameshift in O case AB people have 1 A on one copy of chr 9 and 1 B on the other copy of chr 9 03/26/2019 Carbohydrates p. 72 of 75

Incompatibilities n n n O blood can be donated to anyone AB people can receive any blood People with O blood will raise Ig. M antibodies against A or B blood People with B blood will raise Ig. M antibodies against A blood People with A blood will raise Ig. M antibodies against B blood 03/26/2019 Carbohydrates p. 73 of 75

Distributions of blood types n n Note that O is the most common in general, but its prevalence varies worldwide People with O blood are slightly more prone to infection by Vibrio cholerae 03/26/2019 Carbohydrates p. 74 of 75

N-linked oligosaccharides n n Generally linked to Asn Types: n n n High-mannose Complex (Sialic acid, …) Hybrid (Gal, Gal. NAc, Man) 03/26/2019 Carbohydrates Diagram courtesy Oregon State U. p. 75 of 75
- Slides: 75