Carbohydrates and Lipids Carbohydrates Simple and complex sugar

Carbohydrates and Lipids

Carbohydrates • Simple and complex sugar molecules • Biological uses: • • Energy source Building/structural material Cell communication Energy Storage • Made up of only three different elements: • Carbon • Hydrogen • Oxygen

The Simplest Sugars • Monosaccharide – Simplest form of a carbohydrate, consisting of a single sugar unit. • Used as building blocks for more complex carbohydrates • Generally have a ratio of 1 Carbon, 2 Hydrogen and 1 Oxygen (CH 2 O)n (n= #of carbon atoms) • Monosaccharides that contain three (triose), five (pentose) and six (hexose) are the most common in living organisms.
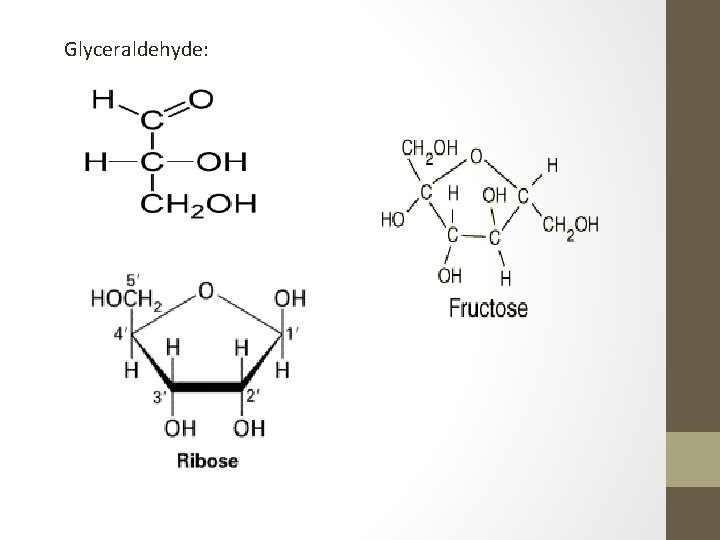
Glyceraldehyde:
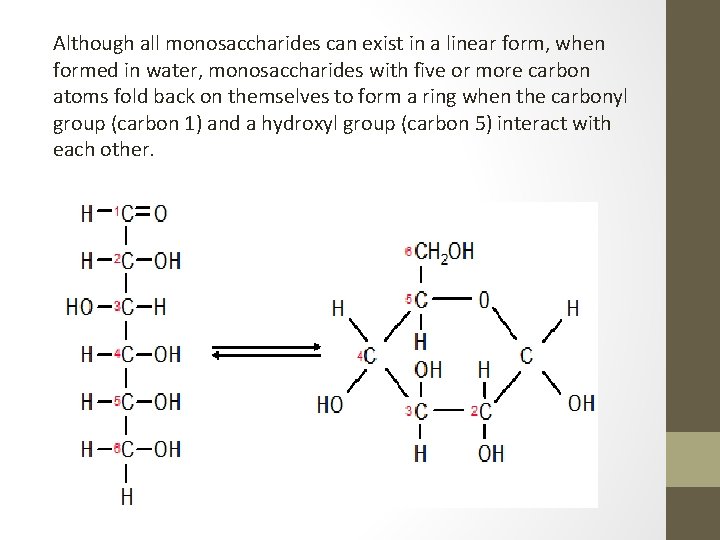
Although all monosaccharides can exist in a linear form, when formed in water, monosaccharides with five or more carbon atoms fold back on themselves to form a ring when the carbonyl group (carbon 1) and a hydroxyl group (carbon 5) interact with each other.
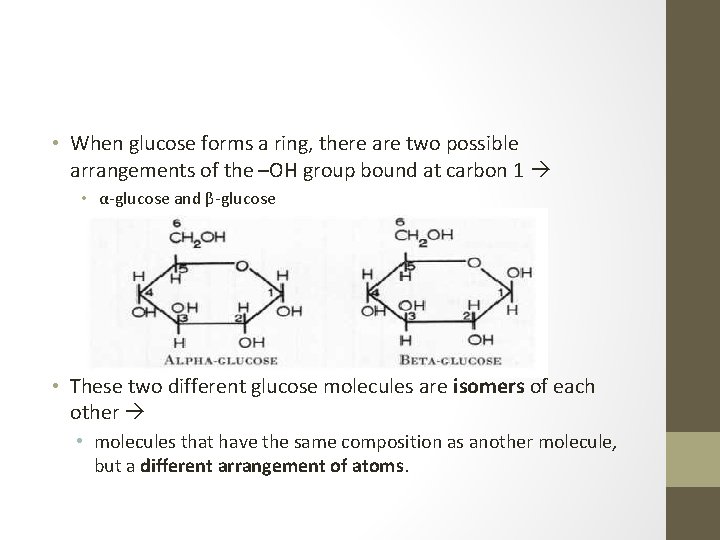
• When glucose forms a ring, there are two possible arrangements of the –OH group bound at carbon 1 • α-glucose and β-glucose • These two different glucose molecules are isomers of each other • molecules that have the same composition as another molecule, but a different arrangement of atoms.

• Monomer a small molecule that can bind chemically to other molecules • Polymer a large molecule that is formed when monomers link together chemically in a chain. • Polymerization a process in which small subunits (either identical or variable) are linked to form a large molecule
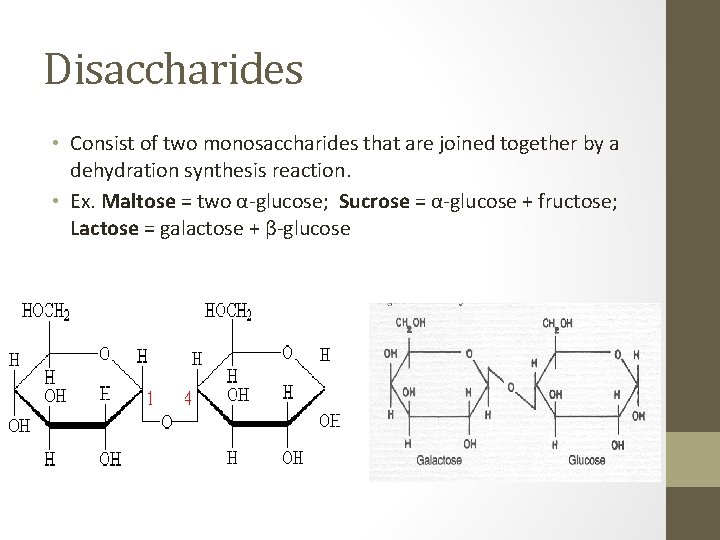
Disaccharides • Consist of two monosaccharides that are joined together by a dehydration synthesis reaction. • Ex. Maltose = two α-glucose; Sucrose = α-glucose + fructose; Lactose = galactose + β-glucose
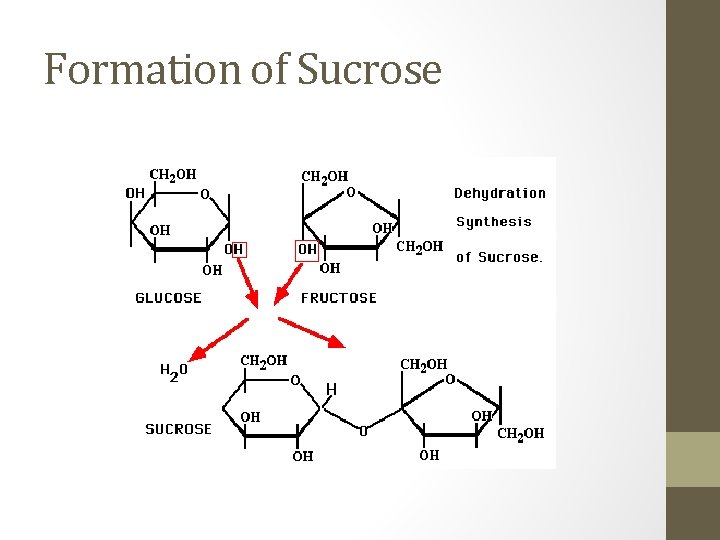
Formation of Sucrose

• Bonds which link monosaccharides into larger carbohydrates are called: Glycosidic bonds. • Linkages are designated as either α or β depending on the orientation of the –OH functional group bonded to the 1 carbon. • Ex. Maltose and sucrose have an α-linkage, lactose has a β-linkage • Like monosaccarides, disaccharides have polar functional groups that make them very hydrophilic; therefore they are easily dissolved in water.

Complex Carbohydrates • Polysaccharides Hundreds to thousands of monosaccharides can link together to form a complex carbohydrate. • Starch and glycogen are important for energy storage. Others, like cellulose and chitin, are essential for structural support. • Glycogen and cellulose are the most common polysaccharides. They assemble from hundreds to thousands of glucose units. • Polysaccharides are very polar and therefore very hydrophilic. However, since they are such large molecules, they attract water but do not dissolve.

Cellulose • Cellulose molecules are long and straight with many polar –OH groups. • These two features enable many cellulose molecules to form side by side and form hundreds and thousands of H-bonds, which gives cellulose its strength.
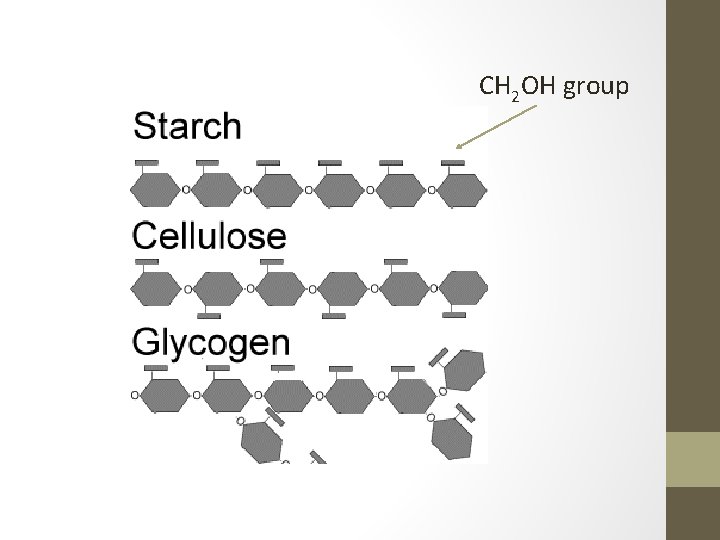
CH 2 OH group

Lipids • A non-polar compound that is made mostly of carbon and hydrogen. • Because they are non-polar, they do not dissolve in water (hydrophobic). This contributes to their ability to form cell membranes… • Other functions include: being stored by cells to be used as an energy source, hormones that regulate cellular activities, and vitamins. • Five main categories of lipids: • • • Fatty acids Fats Phospholipids Steroids Waxes

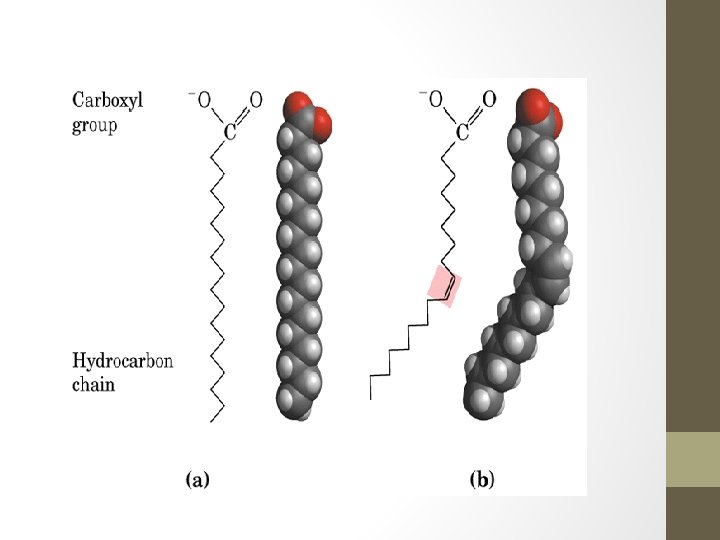
Fatty Acids • A molecule that consists of a carboxyl (-COOH) functional group and a hydrocarbon chain. • Fatty acids contain 4 or more carbons in the chain. • Most common forms of fatty acids have even-numbered chains of 14 to 22 carbons. • The longer the chain the progressively less water soluble they get

Fatty Acids • If the hydrocarbon chain binds the maximum possible number of hydrogen atoms and all the carbons are linked by single bonds, the fatty acid is said to be saturated. • Unsaturated fatty acids have one of more carbons that are double bonded (one double bond = monounsaturated; two or more double bonds = polyunsaturated. The double bond creates a kink in the molecule which causes it to bend.

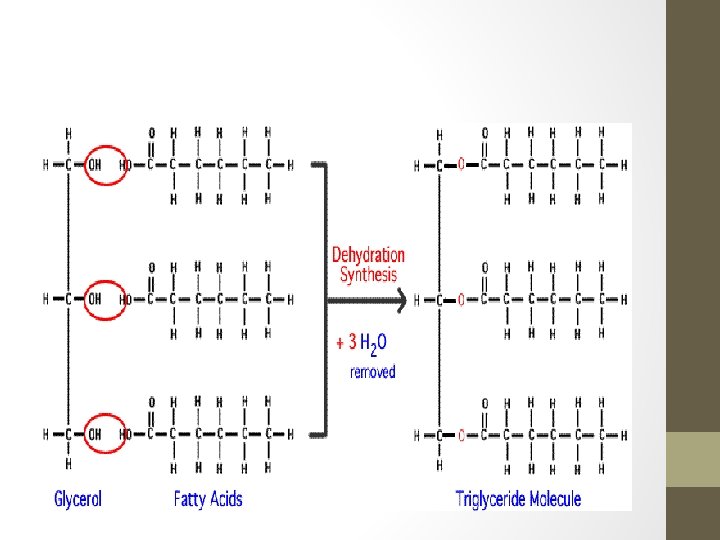
Fats • Made of two types of molecules fatty acid and glycerol • 1 -3 fatty acid chains are joined to a single glycerol molecule through a dehydration synthesis reaction between the –OH functional groups on the glycerol and the carboxyl group on the fatty acid. • If three fatty acids are joined to one glycerol molecule the fat is called a triglyceride

Fats • Fats obtained from animals, (ex. butter or lard) usually only contain saturated fatty acid chains, so they are called saturated fats. • Fats derived from plants, (ex. olive oil) usually contain more unsaturated and polyunsaturated fatty acid chains, so they are called unsaturated fats. These are generally referred to as oils. • Unsaturated fats are liquids at room temperatures and saturated fats are solid. • Unsaturated fats have shorter chains than saturated fats Longer chains = less fluid as length increases.

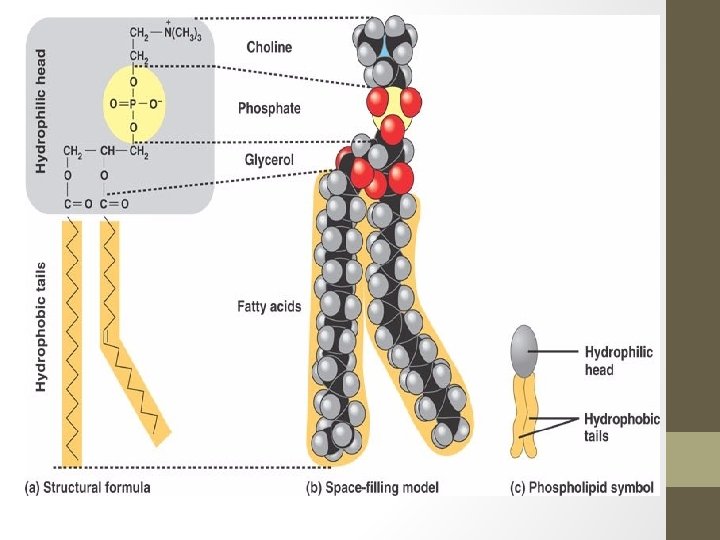
Phospholipids • Consists of two fatty acids and a phosphate group bound to a glycerol • These are the primary lipids of a cell membrane • Like in a fat molecule, glycerol is the backbone, however, it has a charged phosphate group which usually binds to another polar or charged unit (eg. Choline). • Contains both a hydrophobic portion (fatty acid tails), and a hydrophilic portion (head). Molecules that contain both are called amphipathic molecules. • Because of this polar head and non-polar tail, these molecules will form a phospholipid bilayer when in water with heads pointing towards the water and tails pointing away.

Steroids • Structures are based on a framework of four carbon rings. • Small differences in the side groups attached to the rings distinguish one from another. • The most abundant are sterols. They have a single polar –OH group at one end of the ring framework, and a complex, non-polar hydrocarbon chain at the other. • Sterols are almost completely non-polar (hydrophobic), the –OH gives the one end a slight polar (hydrophilic) property

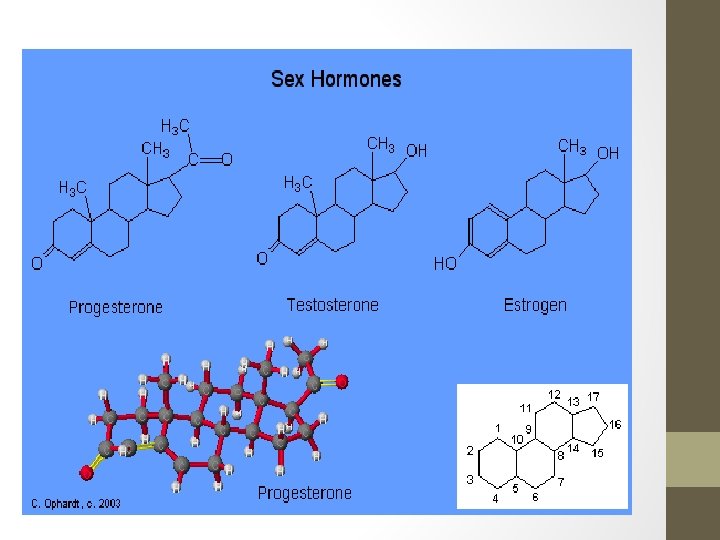
Steroids • Cholesterol is an important component of the plasma membrane surrounding a cell and it also can convert into a number of different compounds such as vitamin D. • Sex hormones, such as testosterone, estrogen and progesterone are also steroids. • Anabolic steroids mimic the male sex hormone testosterone.

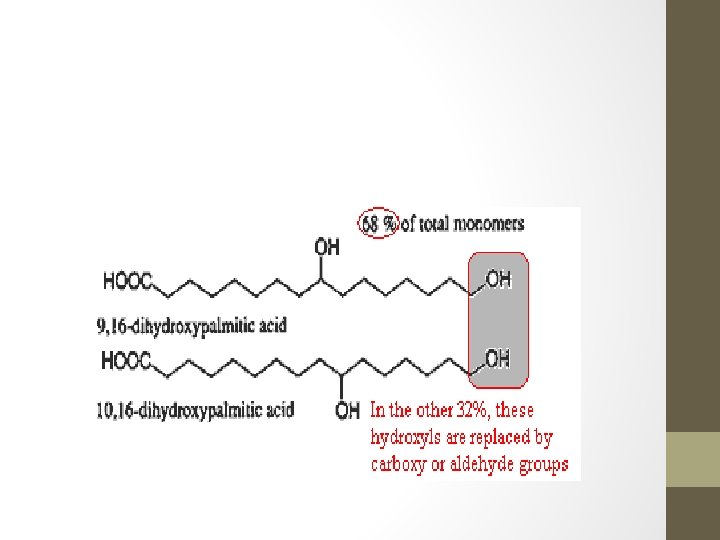
Waxes • Formed when long fatty acid chains are joined to alcohols or carbon rings. • Extremely non-polar, hydrophobic and soft solids over a wide range of temperatures. • Makes them ideal for flexible, water-resistant coatings on various plant or animal parts. • Ex. Cutin • produced by certain plant cells to produce a water-resistant coating on leaves, stems and fruits. This enables the plant to conserve water, and acts as a barrier to infection and disease. • Birds produce a waxy material to keep their feathers dry and bees produce beeswax to make their honeycombs.
- Slides: 28