Carbohydrates 2 3 1 Carbohydrates A Organic compounds

Carbohydrates (2 -3)

1. Carbohydrates A. Organic compounds composed of Carbon, Hydrogen and Oxygen. C-6, H-12, O-6 B. Carbo = carbon; hydro = water 1. Ratio of 2 hydrogen's to 1 oxygen C. Carbohydrates exist as subunits called monosaccharide, disaccharide and polysaccharide.
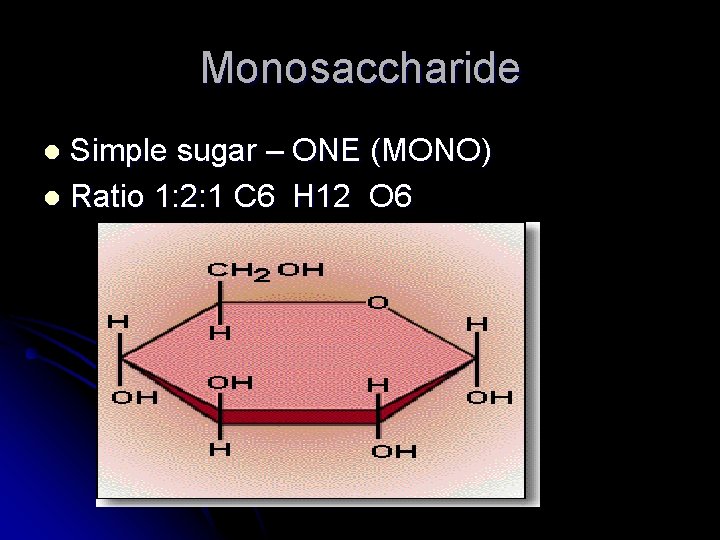
Monosaccharide Simple sugar – ONE (MONO) l Ratio 1: 2: 1 C 6 H 12 O 6 l

C. General formula Six carbon monosaccharide C 6 H 12 O 6 D. Most common monosaccharide 1. glucose = blood sugar 2. fructose = fruit sugar 3. lactose = milk sugar

E. When different compounds have the same number of atoms they are called Isomers. 1. Isomers will have the same chemical formula but a different molecular structure and different properties.
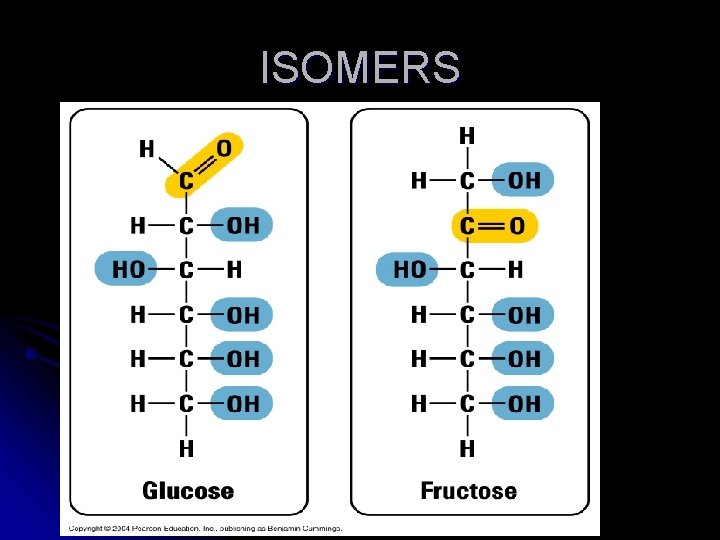
ISOMERS
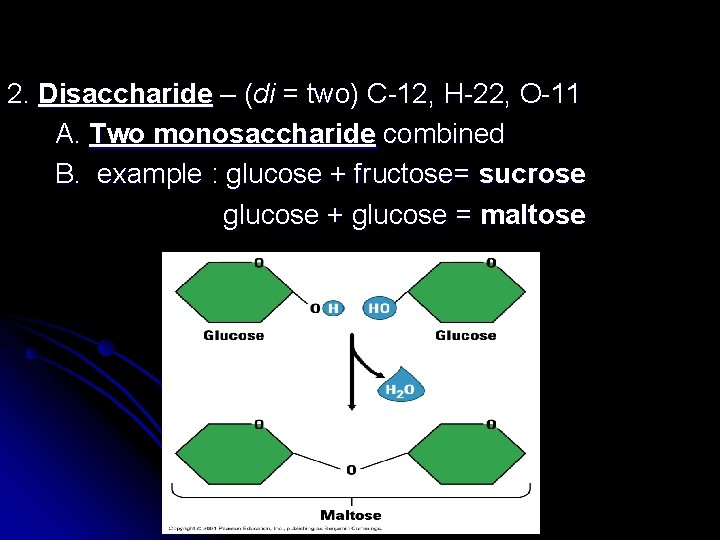
2. Disaccharide – (di = two) C-12, H-22, O-11 A. Two monosaccharide combined B. example : glucose + fructose= sucrose glucose + glucose = maltose

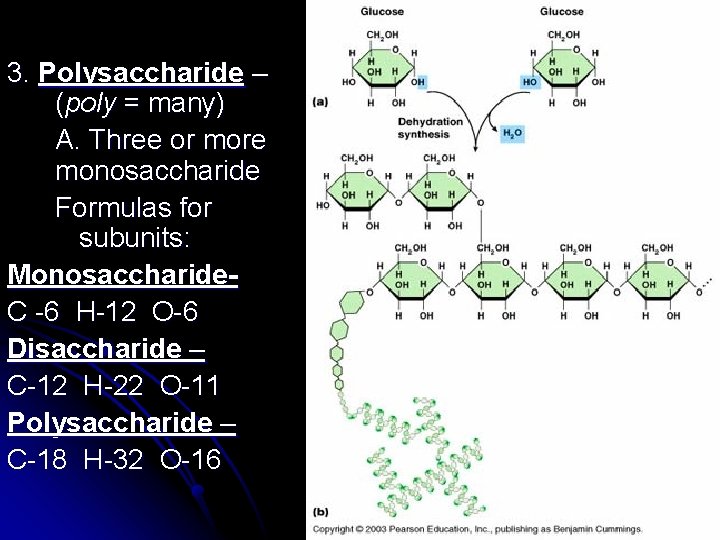
3. Polysaccharide – (poly = many) A. Three or more monosaccharide Formulas for subunits: Monosaccharide. C -6 H-12 O-6 Disaccharide – C-12 H-22 O-11 Polysaccharide – C-18 H-32 O-16

5. Glycogen - Polysaccharide A. Contains hundreds of glucose molecules strung together. B. Glucose is stored in the liver and muscles as glycogen and ready for use as energy. 6. Starch - Cellulose - Polysaccharide A. Starch produced in plants give plants energy and Cellulose found in plants gives strength to plant cells wall.

- Slides: 11